FDA
-
Michelle Tarver faces challenges as new CDRH leader. But patient groups, industry are optimistic.
Patient advocates are hoping for change under new director Michelle Tarver, while industry groups look to build on former director Jeff Shuren’s leadership.
By Elise Reuter • Oct. 31, 2024 -
Deep Dive
AI could be a game changer, but healthcare needs to be ‘exceedingly careful’
Artificial intelligence tools could help solve workforce challenges. Implementation, however, can be difficult, pushing organizations to consider less risky administrative and back-office tasks first.
By Emily Olsen • Oct. 29, 2024 -
 Explore the Trendline➔
Explore the Trendline➔
 Sarah Silbiger via Getty Images
Sarah Silbiger via Getty Images Trendline
TrendlineMedical device safety in spotlight after high profile recalls
From Philips’ massive recall of respiratory devices to ongoing health risks with breast implants, medical devices tied to patient harm have put a focus on product safety.
By MedTech Dive staff -

 Retrieved from Hologic on May 23, 2024
Retrieved from Hologic on May 23, 2024
FDA advises clinicians to stop using Hologic radiographic markers
Hologic has recalled the Biozorb devices after 188 reports of adverse events in patients who received the implants in breast tissue.
By Susan Kelly • Oct. 28, 2024 -
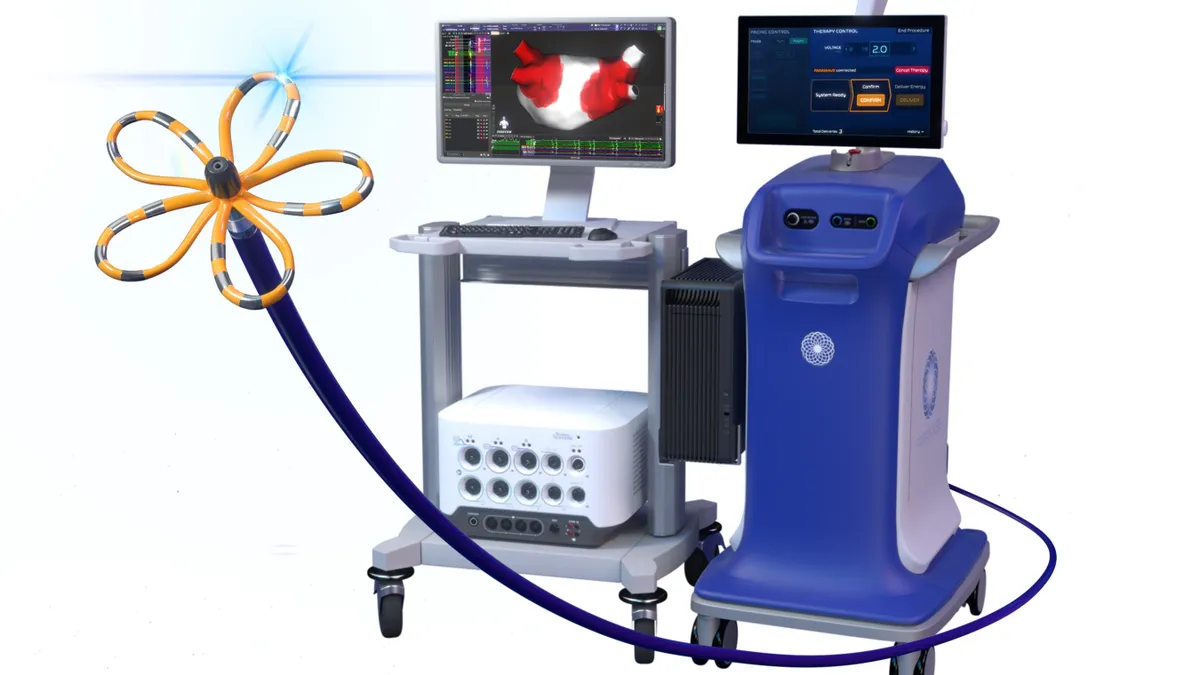
Medtronic wins FDA approval for Affera mapping and ablation system
Affera combines mapping technology with a catheter capable of performing radiofrequency and pulsed field ablation.
By Nick Paul Taylor • Oct. 25, 2024 -
The Medtech Conference
Medtech Conference recap: New CDRH leader details approach; AI and LDTs in focus
Catch up on our recent coverage of Advamed’s The Medtech Conference.
By Elise Reuter • Oct. 23, 2024 -

 Retrieved from iRhythm on October 22, 2024
Retrieved from iRhythm on October 22, 2024
iRhythm’s Zio AT design changes win FDA clearance
The agency cleared one of two 510(k) submissions iRhythm filed for the heart monitor after receiving a warning letter from the agency last year.
By Susan Kelly • Oct. 23, 2024 -
Tarver named new director of FDA’s device center
Michelle Tarver, who will officially replace longtime CDRH leader Jeff Shuren, emphasized the agency’s focus on patients in comments last week.
By Elise Reuter • Oct. 22, 2024 -
The Medtech Conference
That’s a wrap: 5 takeaways from The Medtech Conference
The conference — Advamed’s largest — featured an appearance by the device center’s new acting director and sessions on AI, clinical trial diversity and the FDA’s contentious LDT rule.
By Elise Reuter • Oct. 22, 2024 -
Deep Dive
5 steps to navigate the FDA’s new lab developed test rule
Laboratories face a series of upcoming deadlines to comply with stricter FDA oversight of in-house tests. Here are five strategies labs can take to be ready.
By Susan Kelly • Oct. 21, 2024 -
Boston Scientific wins FDA approval for Farapulse cardiac mapping
Stifel analyst Rick Wise said Boston Scientific can now offer “one-stop-shopping” with a pulsed field ablation catheter and integrated mapping system to treat atrial fibrillation.
By Susan Kelly • Oct. 21, 2024 -
Boston Scientific blood-blocking agent tied to additional 2 deaths, 8 injuries
After a February recall, Boston Scientific has warned physicians about new safety risks for Obsidio Embolic, which is now connected to a total of 15 injuries and four deaths.
By Nick Paul Taylor • Oct. 21, 2024 -
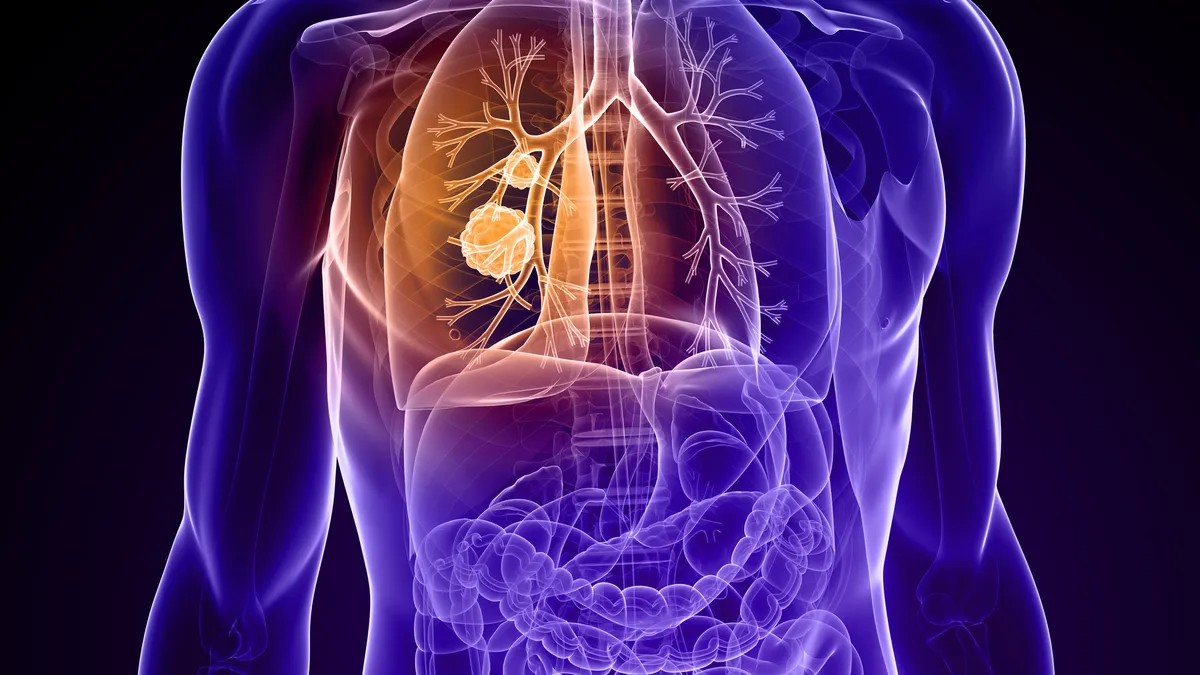
Novocure wins FDA approval for electric field device in lung cancer
Alongside immunotherapy or chemo, the wearable device helped patients live longer in a trial, though the results are somewhat controversial.
By Jonathan Gardner • Oct. 18, 2024 -
The Medtech Conference
Tarver, acting CDRH director, sets tone for future of the device center
Former CDRH leader Jeff Shuren told an audience at The Medtech Conference that Michelle Tarver “will make an excellent center director, and quite frankly, the best is yet to come.”
By Elise Reuter • Oct. 18, 2024 -
FDA prioritizes guidance on lab developed tests in 2025 plan
The Center for Devices and Radiological Health intends to develop final guidance on its enforcement discretion policy regarding special controls for LDTs, among other topics.
By Nick Paul Taylor • Oct. 18, 2024 -
FDA labels Cue’s recall of COVID tests as Class II, revokes EUAs
Cue Health began the recall of nearly 250,000 COVID-19 test kits in May, shortly after the FDA advised people at home and healthcare professionals not to use the products.
By Nick Paul Taylor • Oct. 17, 2024 -
The Medtech Conference
Legacy medical devices keep regulators up at night
The FDA’s Suzanne Schwartz said at The Medtech Conference that addressing legacy devices is a “work in progress” and a problem regulators and industry need to work on together.
By Elise Reuter • Oct. 17, 2024 -
Health officials outline industry role in AI oversight in JAMA article
FDA Commissioner Robert Califf and two colleagues warned that the “scale of effort” needed to repeatedly evaluate AI models “could be beyond any current regulatory scheme.”
By Nick Paul Taylor • Oct. 16, 2024 -
The Medtech Conference
FDA, Health Canada give sneak peek into future AI regs
Canada’s device agency plans to issue guidance on machine learning, and the FDA plans to release a draft guidance next year on lifecycle management and premarket submissions for AI devices.
By Elise Reuter • Oct. 16, 2024 -
Medtronic to evaluate Affera in ventricular tachycardia
The FDA approved an early feasibility study of Medtronic’s Affera mapping and ablation system and Sphere-9 catheter in patients with ventricular tachycardia, an abnormal heart rhythm.
By Susan Kelly • Oct. 15, 2024 -
The Medtech Conference
4 trends to watch at Advamed’s The Medtech Conference
As medtech companies, experts and regulators gather in Toronto, FDA leadership changes and the upcoming U.S. presidential election loom over the industry.
By Elise Reuter • Oct. 14, 2024 -

 Retrieved from Aerial Lens on October 10, 2024
Retrieved from Aerial Lens on October 10, 2024
Baxter ups IV fluid allocations amid supply shortages
As hospitals around the U.S. report IV fluid shortages, Baxter has increased supply allocations for direct customers and distributors. It plans on returning to 90% to 100% allocation of certain supplies by the end of 2024.
By Ricky Zipp • Oct. 10, 2024 -
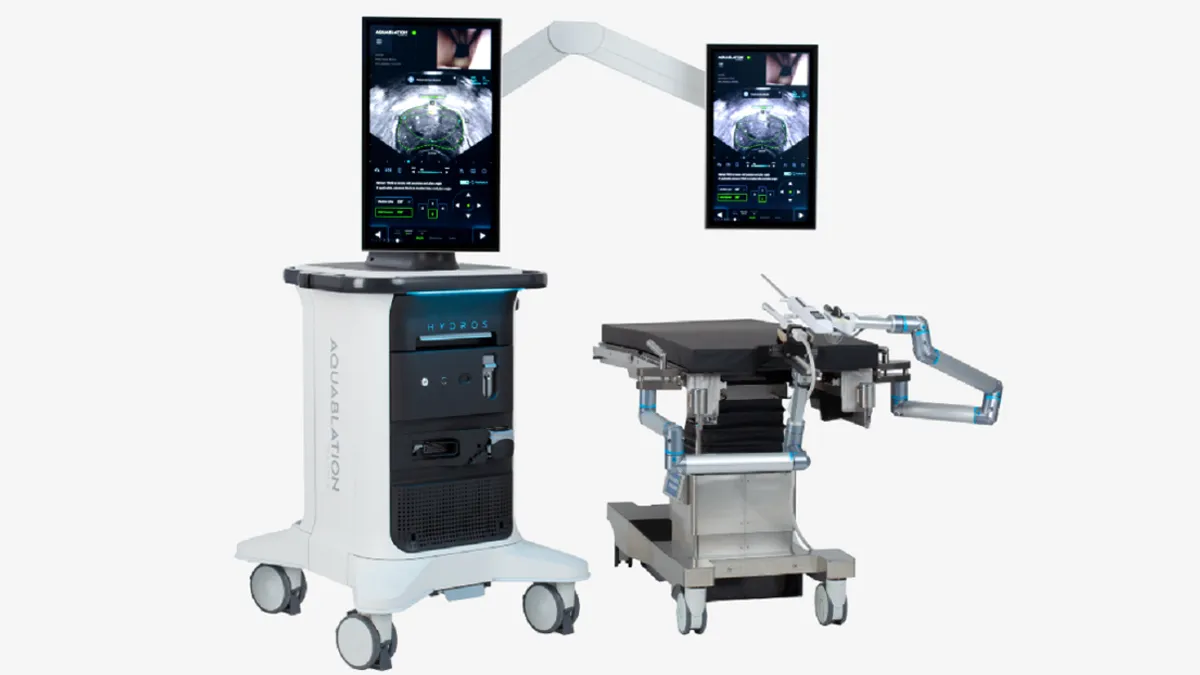
Procept secures FDA approval to study surgical robot in prostate cancer
The trial will compare the therapy to radical prostatectomy and could unlock a market that analysts value at $500 million.
By Nick Paul Taylor • Oct. 9, 2024 -
Mercury Medical recalls emergency resuscitators
Healthcare professionals use the devices to provide newborns and infants with emergency breathing support.
By Nick Paul Taylor • Oct. 8, 2024 -
Exact Sciences wins FDA approval for Cologuard Plus colorectal cancer test
Exact Sciences expects to launch the updated test, which has higher specificity than its predecessor, with Medicare coverage and guideline inclusion in 2025.
By Nick Paul Taylor • Oct. 7, 2024 -
Elucid wins FDA 510(k) nod for heart plaque image analysis software
The software turns coronary CT angiography images into 3D models that quantify and classify plaque morphology to improve predictions of heart attack and stroke risk.
By Nick Paul Taylor • Oct. 4, 2024