Medical technology companies in 2024 are launching devices that address two especially vexing challenges affecting millions of heart patients: high blood pressure and atrial fibrillation, a form of irregular heartbeat. Company executives anticipate multibillion-dollar markets for these treatments, and they have key developments lined up for this year.
Medtronic, Boston Scientific, Johnson & Johnson and Abbott are tackling AFib with a new technique that uses pulsed field energy to ablate heart tissue and new approaches to prevent blood clots from escaping the heart’s left atrial appendage.
Medtronic is pushing ahead with the introduction of a novel procedure called renal denervation that aims to lower blood pressure.
In the treatment of valve disease, a common heart problem that restricts blood flow, market leader Edwards Lifesciences is working to obtain new approvals and expanded indications.
“It does feel like a time when medtech is getting a resurgence of innovation and a resurgence of new technology for doctors to use,” said BTIG analyst Marie Thibault.
Here are four trends in cardiac care setting the course for medtech companies this year:
1. Heart device heavyweights focus on pulsed field ablation
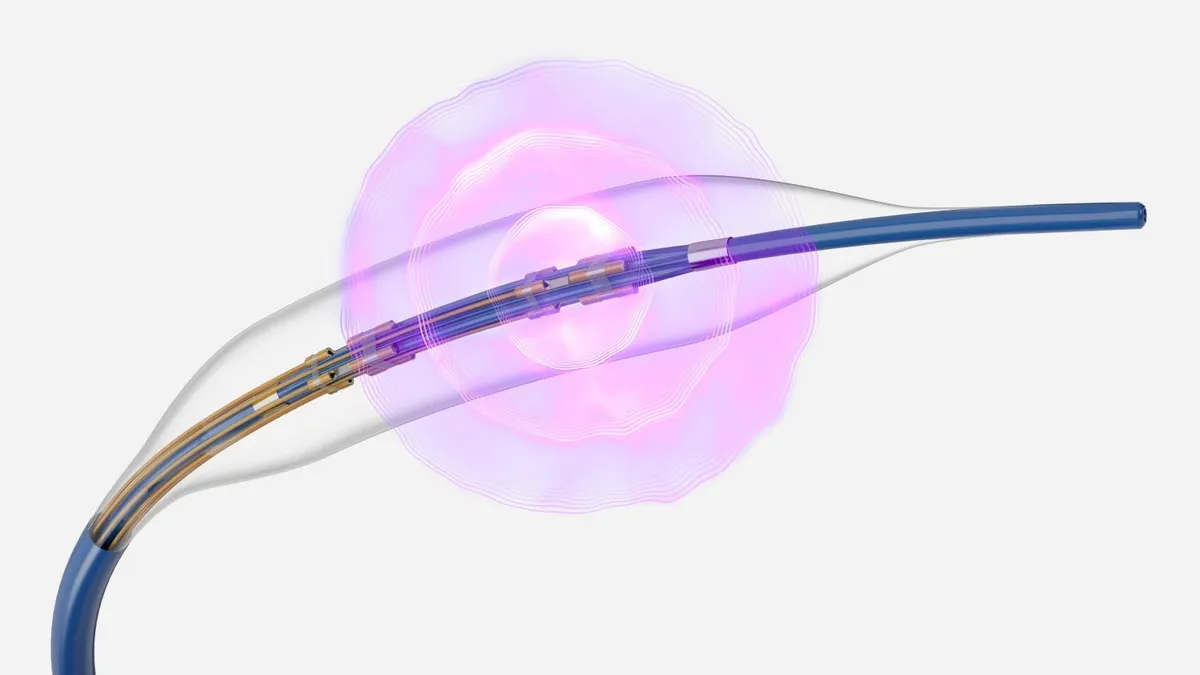
Medtronic, Boston Scientific, J&J and Abbott all are pursuing the emerging market for pulsed field ablation (PFA), a treatment for AFib that is attracting strong interest from heart doctors.
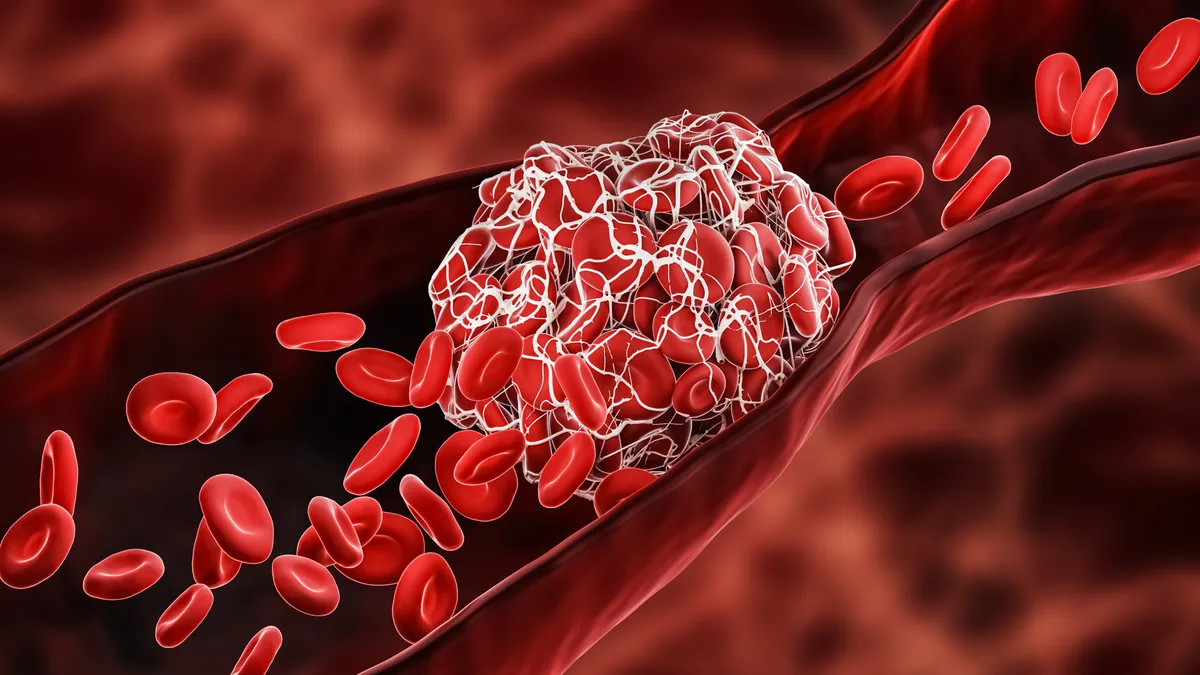
AFib, an irregular heart rhythm that raises the risk for blood clots, is increasing in prevalence as the population ages. PFA, which applies short, high-voltage pulses of energy to cardiac tissue, is thought to be more precise than traditional ablation methods that use thermal energy to combat AFib.
“This is being viewed as probably the best evolution within the electrophysiology space in 15 years,” said Mizuho analyst Anthony Petrone. “There is a lot of anticipation for these new PFA catheters.”
PFA systems from Medtronic and Boston Scientific are already in use in Europe.
“What we see from the data is that it's faster and safer. And so that's enough for doctors to get excited,” said BTIG’s Thibault.
In December, Medtronic secured the first U.S. approval for a PFA system, called PulseSelect. Soon after, Boston Scientific moved up the timeline for launching its Farapulse PFA device in the U.S. to the first quarter of this year.
Petrone said the device industry will have “basically a two-horse race” in PFA for now.
J&J could be third to market in the U.S. The company plans to submit its Varipulse catheter for Food and Drug Administration review in 2024, CEO Joaquin Duato said last week on an earnings call.
“This is being viewed as probably the best evolution within the electrophysiology space in 15 years. There is a lot of anticipation for these new PFA catheters.”

Anthony Petrone
Mizuho analyst
Abbott is now entering the competition as well. The company said this month it expects approval in the first half of the year for a U.S. clinical trial of its Volt PFA system.
Boston Scientific expects the global AFib ablation market to more than double to $11 billion by 2028, with PFA predicted to grow to more than 60% of procedures, from under 5% today.
Both Medtronic and J&J also have AFib devices in the works that integrate pulsed field and radiofrequency energy. Medtronic is working to bring its Affera system to the U.S., while J&J is evaluating a dual- energy approach.
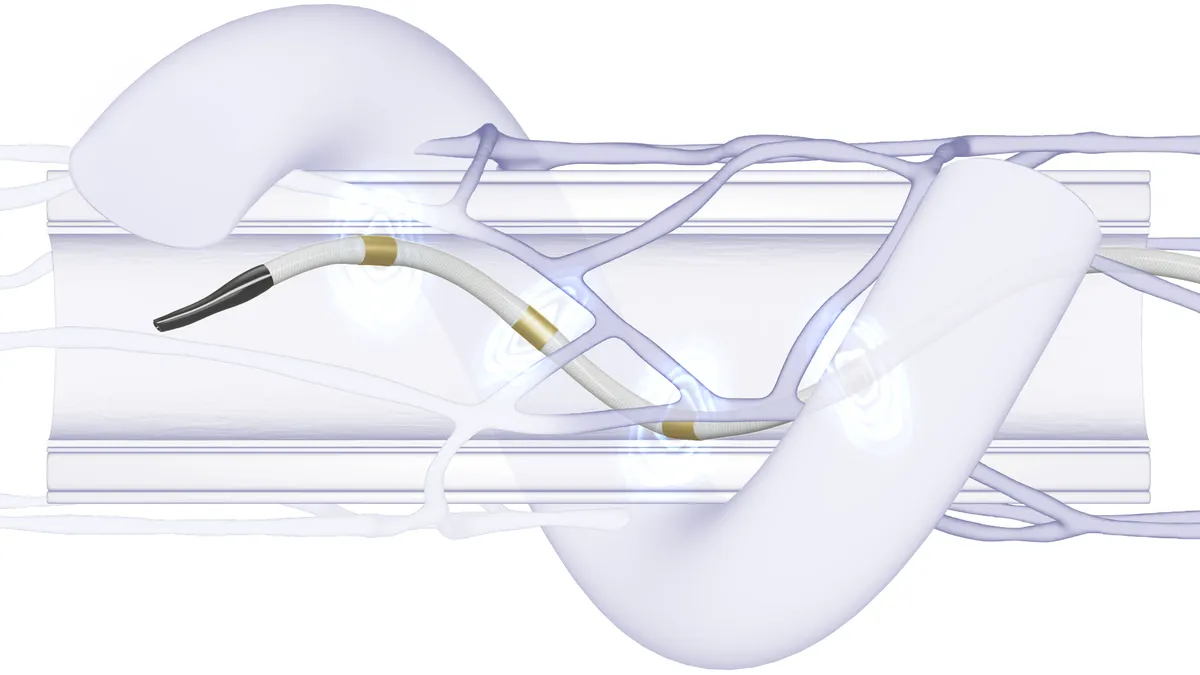
2. Boston Scientific faces challengers in left atrial appendage treatment
Late 2023 saw both Medtronic and J&J announce the acquisition of devices designed to reduce the stroke risk associated with the heart’s left atrial appendage (LAA), a small sac in the upper left chamber. The devices aim to stop clots caused by AFib from entering the bloodstream.
Boston Scientific, whose Watchman implant is the leader in LAA closure, has predicted the market could quadruple to more than $6 billion by 2030. The company promotes the device, which fits into the LAA to permanently close it off, as an alternative to a lifetime of blood thinners for patients with AFib not caused by a valve condition.
Abbott introduced a competing LAA closure system, the Amplatzer Amulet, in 2021.
“What we've seen so far is that Boston’s business continues to withstand the competitive pressure quite well,” said Morningstar analyst Debbie Wang.
BTIG’s Thibault predicted the LAA market will grow by more than 20% this year.
“Boston Scientific's Watchman has been able to keep growing despite the competitive entry from Abbott, and Abbott’s entry has expanded the market for left atrial appendage closure,” she said.
Now, Medtronic has jumped in with the acquisition of the Penditure exclusion system. The device, currently rolling out in the U.S., is expected to compete directly with Atricure’s Atriclip.
J&J, through its acquisition of Laminar, is developing a separate approach that would eliminate the left atrial appendage.
“Left atrial appendage closure and exclusion is a hot space,” said Thibault.
3. Reimbursement is key as Medtronic forges new ground in hypertension
Nearly 93 million U.S. adults have uncontrolled high blood pressure, putting them at risk for heart attack or stroke.
Medtronic is bringing to market one of the first device-based treatments for those patients, after overcoming multiple setbacks during its 14 years in development. The renal denervation procedure aims to lower blood pressure with radiofrequency energy targeted at nerves leading to the kidneys.
“I think that market is going to be smaller than everyone hoped for.”

Jeff Jonas
Gabelli Funds
CEO Geoff Martha in December called the market a multibillion-dollar opportunity.
The technology, however, has its skeptics in the medical community. Medtronic and smaller competitor Recor secured FDA approval in November for renal denervation devices despite heated debate among the agency’s expert advisers about the effectiveness of the treatment. Medtronic’s Symplicity Spyral device missed the primary endpoint in a pivotal trial.
The concerns could factor into payers’ reimbursement decisions, ultimately limiting the pool of patients likely to receive the treatment, according to analysts.
“I think that market is going to be smaller than everyone hoped for,” said Jeff Jonas, a portfolio manager at Gabelli Funds. “There are dozens of blood pressure medicines that are all generic and low cost, and work well. For the most part, that market is pretty well served with drug therapy.”
Private insurers and Medicare could limit reimbursement for renal denervation to people who can’t comply with drug therapy, Jonas added.
“There are people who do have side effects or drug-drug interactions, or just don't take their medicine like they are supposed to. It's going to be a relatively small subset of those patients that get denervation surgery,” he said.
Medtronic is pursuing both local and national Medicare coverage determinations and has been in contact with the largest private payers, Sean Salmon, president of Medtronic’s cardiovascular portfolio, said on an earnings call in November.
“Rest assured, we’re working hard on reimbursement,” he said. “It’s really critical for the ramp of this technology.”
4. Valve leader Edwards targets a wider patient population
Heart valve procedures have been slow to bounce back after the COVID-19 pandemic, when hospitals postponed non-urgent operations, creating backlogs that providers continued to work through in 2023.
“They haven't really gotten the same post-COVID rebound that we've seen with orthopedics or other surgeries,” said Gabelli’s Jonas.
Market leader Edwards has forecast sales growth of 8% to 10% for 2024, below pre-pandemic levels. Medtronic, which competes against Edwards in the $6 billion-plus transcatheter aortic valve replacement (TAVR) market, pegs the segment’s growth in the high-single- to low-double-digits.
“Everybody's wondering if we've run out of TAVR patients, because both Medtronic and Edwards have seen their TAVR products growing now somewhere in the low-double-digits, versus growing at 15 or 16%,” Morningstar’s Wang said.
Edwards is still working to get patients “off the sidelines” after the COVID disruption, Larry Wood, group president of TAVR and surgical structural heart, told analysts on the company’s third-quarter earnings call.
Against this backdrop, the company is pushing ahead with efforts to expand the pool of patients eligible to receive a replacement aortic valve. Data from Edwards’ pivotal trial studying TAVR in patients with severe aortic stenosis without symptoms, expected to be presented at the Transcatheter Cardiovascular Therapeutics conference later this year, will be closely watched, analysts said.
“Many patients are still undiagnosed and untreated,” CEO Bernard Zovighian said in a presentation this month at the J.P. Morgan Healthcare Conference.
Interest in the company’s progress on devices to treat mitral and tricuspid valve regurgitation is also high. Edwards’ Pascal mitral valve repair system was approved in the U.S. in late 2022 and competes against Abbott’s Mitraclip.
“The big question in my mind for 2024 is what kind of headway is Edwards going to make in the U.S. with its Pascal product,” said Wang. “Now that they have approval in the U.S., how quickly can they train up the centers, and how quickly can they get practitioners who are used to using Mitraclip to try and switch over to Pascal?”
Edwards expects to gain FDA approval for the Evoque tricuspid valve in mid-2024.