Dive Brief:
- Abbott’s remote heart failure monitoring system has improved quality of life and reduced heart failure hospitalizations in a clinical trial.
- The data, which were published in The Lancet, come from a trial that randomized 348 patients to be monitored using Abbott’s CardioMEMS device or standard care. The study linked the device to a 44% reduction in heart failure-related hospitalizations.
- Dutch researchers ran the clinical trial to address the lack of evidence on the use of the device in Europe. The study is the third randomized, controlled trial globally to link CardioMEMS to a significant health benefit.
Dive Insight:
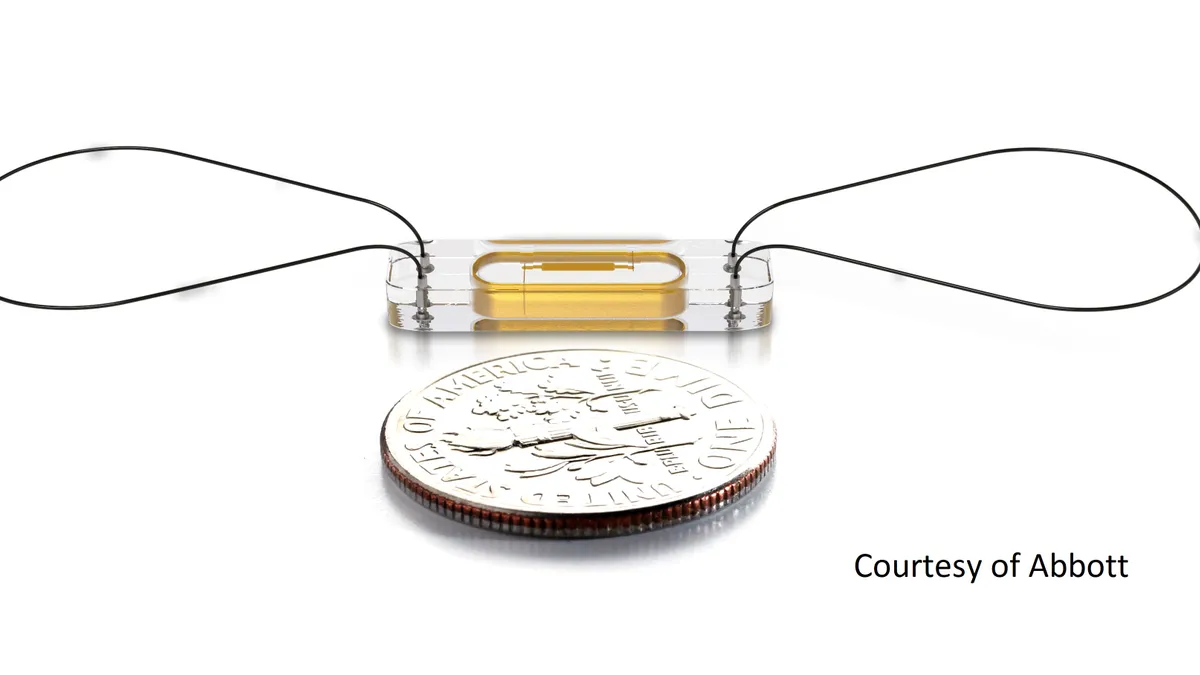
CardioMEMS consists of a paperclip-sized sensor that is implanted in the pulmonary artery. Once in place, the sensor monitors pulmonary artery pressure and sends notifications if changes in the patient’s condition indicate worsening heart failure. The device is designed to detect deterioration before physiological markers change and thereby enable early, proactive adjustments to treatment.
Abbott won U.S. Food and Drug Administration approval for a wireless version of the device in 2014 and has continued to gather data on its safety and efficacy since then. The latest evidence comes from a trial initiated by investigators in the Netherlands.
The evidence, which was also shared at a European Society of Cardiology meeting, shows that patients monitored using Abbott’s CardioMEMS scored significantly higher on the Kansas City Cardiomyopathy Questionnaire (KCCQ) overall summary than their peers in the control group. KCCQ Scores, a measure of the patient's perception of their health status, improved by seven points in the CardioMEMS cohort.
With scores falling slightly in the control group, the study detected a significant difference in the KCCQ results, indicating that CardioMEMS improved the quality of the patients’ lives. Improvements in KCCQ were seen from the first assessment, which took place three months after implantation. Patients in the CardioMEMS cohort were also less likely to be hospitalized with heart failure.
Earlier data on the effectiveness of CardioMEMS primarily came from the U.S.. Running the study at 25 centers in the Netherlands meant the device needed to improve on high-quality standard care, according to J.J. Brugts, principal investigator for the trial and associate professor at Erasmus Medical Center.
“The Dutch health care system is known for its structured approach to heart failure management at outpatient clinics with general access to guideline-directed medical therapies. This approach raises the bar, making the MONITOR-HF results even more meaningful given that patients who were already receiving a high standard of care reported significant improvements in their quality-of-life while using the CardioMEMS monitoring system,” Brugts said in a statement.