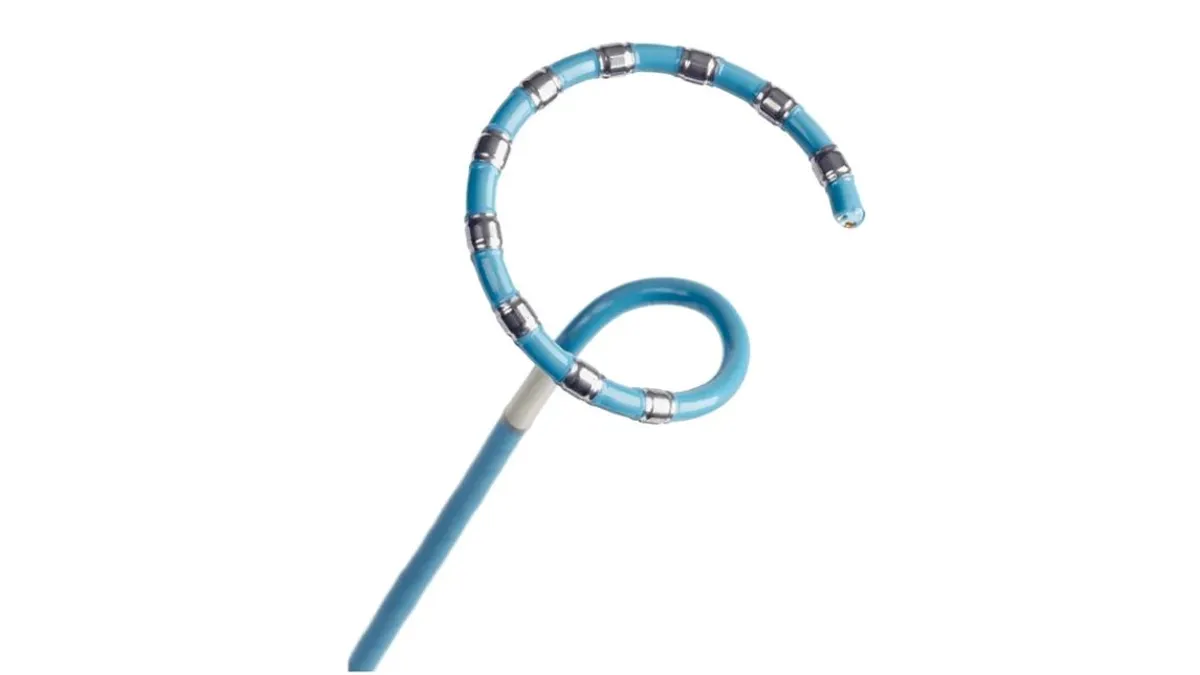
Boston Scientific shared pivotal trial results on Wednesday that showed promising results for its drug-eluting balloon in treating patients with repeat blockages.
The company’s Agent drug-coated balloon performed better than an uncoated balloon in procedures to reopen blocked arteries at one year, according to data presented at the Transcatheter Cardiovascular Therapeutics (TCT) 2023 conference. Boston Scientific was the study sponsor.
Stents are often used to treat patients with coronary artery disease, but sometimes the stented part of the artery can become blocked or narrowed by scar tissue. About 10% of patients who undergo coronary angioplasty end up needing restenosis, said Robert Yeh, section chief of interventional cardiology at the Beth Israel Deaconess Medical Center and the study’s lead investigator. Yeh is also a paid consultant of Boston Scientific.
When this happens, treatment options in the U.S. include balloon angioplasty, putting in another stent layer, or a rare option of using radiation to prevent the artery from blocking up again, Yeh told MedTech Dive.
“No matter how we treat these patients, once you've blocked up your artery one time, you are at a much, much higher risk for blocking it up again, and going down this pathway of procedure after procedure, and potentially stent after stent,” Yeh said. “We really are looking for more options for these patients, particularly ones that don't require us to leave additional layers of metal in the artery.”
Boston Scientific hopes its Agent balloon, which is coated in the drug paclitaxel, can prevent future blockages in these patients. Paclitaxel inhibits the cell growth inside the stent. No drug-coated balloons are currently approved in the U.S. for coronary in-stent restenosis, but they are a first-line treatment in Europe, where Agent has been available as a CE-marked device since 2014.
Paclitaxel has come under scrutiny after the Food and Drug Administration required warnings be added to device labeling in response to a 2018 analysis that linked drug-coated balloons and stents to an increased risk of death. The agency recently walked back its concerns after a new meta-analysis found no increased risk.
Yeh said the device is designed with a low-dose formulation of paclitaxel, and it is designed so the drug stays on the balloon, so very little travels downstream. He added there were no safety signals in the study.
Trial results
Boston Scientific shared results of a randomized, controlled trial, which found that its drug-coated balloon had a lower rate of target lesion failure (17.9%) after 12 months than an uncoated balloon (28.7%), meeting the study’s primary endpoint. Target lesion failure included myocardial infarction, the need for a revascularization procedure, or cardiac mortality.
Individually, procedures with the Agent balloon had no definitive cases of clotting with the stent, compared to 3.9% of patients who underwent procedures with the uncoated balloon. The Agent balloon also demonstrated a 51% reduction in target lesion revascularization and a 49% reduction in myocardial infarction.
Yeh said that the drug-coated balloon was expected to reduce rates of restenosis, “and that’s exactly what it did.” But the reduction in heart attacks was more of a surprise.
“We don't think of … restenosis as being the kind of disease that leads to a substantial number of heart attacks. But in this particular case, actually, there were a number of patients who had heart attacks during follow-up, and that rate was cut in half,” Yeh said. “It speaks to the very high-risk population that was enrolled in the study, as well as the really strong benefits of the device for reducing recurrent events.”
The study included the first 480 of 600 patients enrolled across 40 U.S. sites. About half of the patients had a prior heart attack, three quarters had multivessel disease, 44% of the participants had multiple stent layers, and 51% were diabetic. Three quarters of the patients were men, Yeh added.
Future adoption
The results hold promise for uptake of Boston Scientific’s Agent balloon, which has recieved a breakthrough designation by the FDA.
Trial Chair Ajay Kirtane, director of the Columbia Interventional Cardiovascular Care program, said in an emailed statement that it was “gratifying to see these results which will hopefully facilitate the first approval of a dedicated coronary drug-coated balloon in the United States – fulfilling a huge unmet need for patients.”
The procedure is not difficult, but requires careful attention, Yeh said. It requires “meticulous attention” to vessel preparation, ideally is facilitated by intravascular imaging, and requires precise delivery of the balloon to the right location for the drug application.
For in-stent restenosis, Yeh said he thinks it could be “rapidly taken up as a potential first-line therapy for the device,” depending on when the device is approved, insurance reimbursement and cost.
“There's so much clinical enthusiasm, apart from those issues, that I suspect that for in-stent restenosis, it's going to be used significantly,” Yeh added.
In a September investor call, Boston Scientific President of Interventional Cardiology Therapies Lance Bates said in-stent restenosis could be a $500 million market. Longer-term, the company could also pursue small-vessel procedures where a drug-coated balloon is used instead of a stent.