Dive Brief:
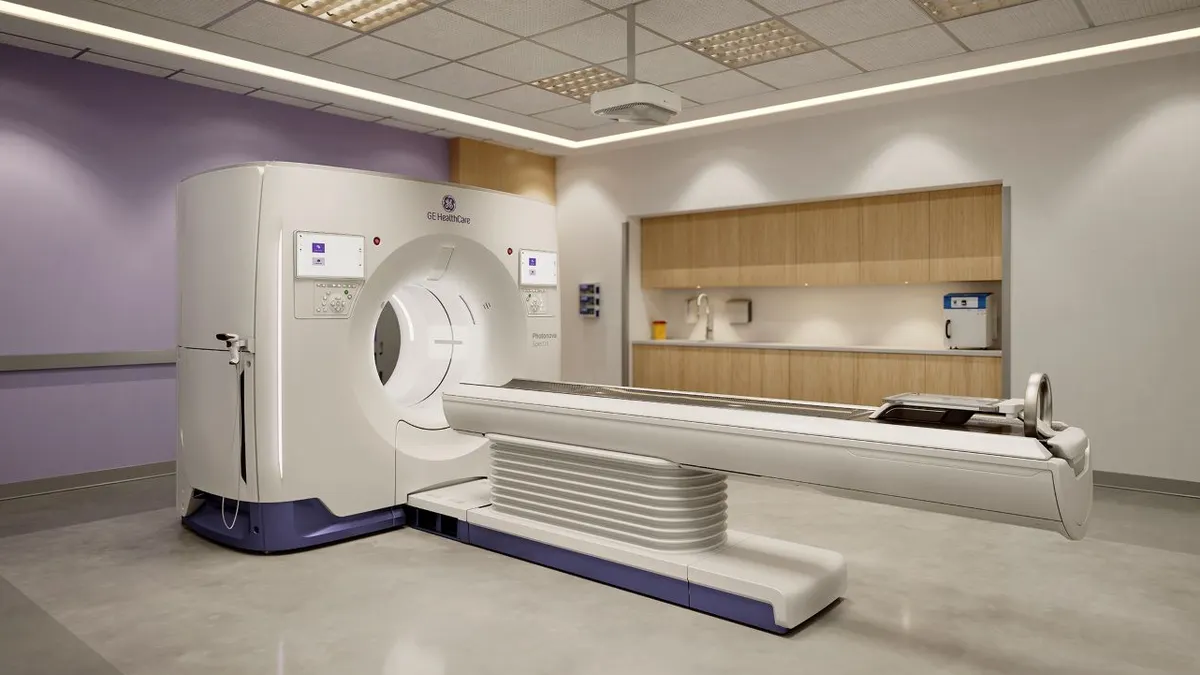
- GE HealthCare has received 510(k) clearance for its spectral photon-counting CT technology, the company said Monday.
- The Food and Drug Administration clearance positions GE HealthCare to compete with Siemens Healthineers’ Naeotom Alpha, the first photon-counting CT machine to reach the U.S. market.
- Citi Research analysts said in a note to investors that GE HealthCare’s Photonova Spectra is differentiated by its use of a detector designed to produce high-contrast spectral images with detailed visualization.
Dive Insight:
Standard CT detectors convert X-rays into light to enable detection by light sensors. Information is lost in the conversion. Photon-counting CT technology eliminates the intermediate step. The imaging devices feature detectors that count X-ray photons and individually measure their energy, retaining information that could be valuable to physicians and delivering a sharper, higher-contrast image.
Siemens Healthineers received 510(k) clearance for Naeotom Alpha in 2021 and expanded the product range in 2025.
GE HealthCare acquired Prismatic Sensors for photon-counting CT technology in 2020. Citi Research analysts said the 510(k) clearance of Photonova followed years of anticipation.
The FDA nod positions GE HealthCare to show whether its claims about the advantages of its technology translate into market share. Photonova is the first photon-counting CT technology to use “deep silicon,” a material that GE HealthCare acquired from Prismatic. GE HealthCare is pitching the sensor technology as a way to empower physicians to clearly distinguish between materials such as iodine, calcium and fat.
Citi Research analysts said the sensor differentiates Photonova, explaining that it enables detailed neurological, oncological, musculoskeletal, thoracic and cardiac imaging. The analysts added that GE HealthCare’s work to ensure Photonova can be easily installed with minimal changes to imaging rooms could reduce barriers to entry and accelerate onboarding.
Siemens Healthineers is defending its business. Discussing photon counting at the J.P. Morgan Healthcare Conference in January, Siemens Healthineers CEO Bernd Montag said his company is “miles ahead in turning this into a business and making a difference for patients.” The company’s photon-counting CT had generated 1 billion euros in orders and 700 million euros in revenue, Montag said.
Citi Research analysts expect Photonova orders to ramp throughout 2026 and contribute to GE HealthCare sales next year. The forecast is aligned with comments GE HealthCare CEO Peter Arduini made on an earnings call last month. Arduini said the order cycle is typically six to nine months, meaning that, while some sales will be generated in 2026, 2027 will be the key year for new imaging products including Photonova.