Dive Brief:
- Johnson & Johnson’s Impella heart pump failed to lower the risk of major adverse events during complex percutaneous coronary interventions and carried a greater risk of death in a study of patients with severe left ventricular dysfunction and extensive coronary disease. The research was presented this weekend at the American College of Cardiology’s annual meeting and published in the New England Journal of Medicine.
- The results of the CHIP-BCIS3 trial are surprising, said lead study author Divaka Perera, because the premise for mechanical circulatory support is to protect the heart by reducing its workload. "Our findings strongly suggest that we shouldn't be using this device routinely without more evidence of benefit," Perera said in a statement.
- A second study, the STEMI-DTU trial, presented at the meeting and published in JACC, found the pump did not improve outcomes in certain heart attack patients when used to rest the heart before stent procedures, prompting investigator Gregg Stone also to caution against routine use.
Dive Insight:
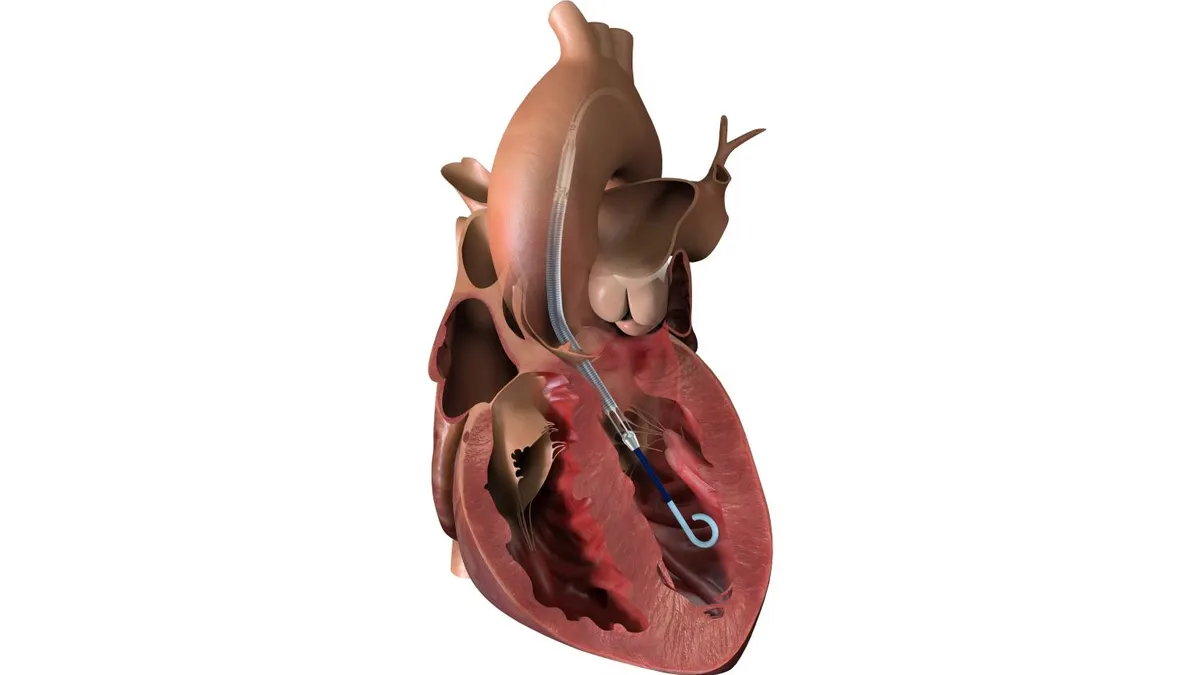
J&J’s Impella pumps, acquired in the $16.6 billion purchase of Abiomed in 2022, have been a strong growth driver for the company’s cardiovascular business, contributing $1.75 billion to sales in 2025.
Impella pumps have Food and Drug Administration approval for use in high-risk PCI, a procedure to open blocked coronary arteries.
Researchers in the 300-person CHIP-BCIS3 trial set out to compare outcomes for patients undergoing PCI with mechanical circulatory support versus without. They analyzed pairs of patients, finding those without a pump fared better in 43% of the comparisons and those who got a pump did better in 36.6%. There was no difference in 20.4% of the pairs.
Forty-seven patients in the pump group died, compared to 33 deaths in the PCI-only group. The higher death rate among patients who received a pump is a strong signal that the treatment is doing harm, Perera said.
The NEJM article noted that use of heart pumps to support high-risk PCI has grown even though the safety and efficacy of the devices had not been compared to PCI without mechanical circulatory support in a randomized trial.
In an editorial accompanying the article, authors Brahmajee Nallamothu and Brett Wanamaker emphasized that the CHIP-BCIS3 data should encourage a more selective approach to the use of mechanical circulatory support in high-risk PCI.
A J&J spokesperson said the CHIP-BCIS3 investigators completed a challenging trial in an advanced heart failure patient population with complex coronary artery disease. “We remain committed to studying this vulnerable population using targeted therapies, shared decision-making, and support strategies that improve patient outcomes,” the spokesperson said in an email.
In the STEMI-DTU trial, researchers tested the hypothesis that a pump could limit heart muscle damage in heart attack patients who don’t have cardiogenic shock, where the body’s organs are not getting enough blood. The study found similar heart muscle damage in the two patient groups: 30.8% with the pump versus 31.9% in the control group. The rate of device-related major bleeding or blood vessel complications at 30 days was 30.8% in the pump group, exceeding the 26.5% performance goal.
“Unfortunately, infarct size, the primary endpoint of the study, was not reduced, and as expected, there were bleeding complications from the device,” said Stone. “Thus, while the microaxial flow pump is life-saving in patients with cardiogenic shock, at the present time we do not recommend its routine use in patients with evolving heart attack without shock."
J&J, in a statement, said the STEMI-DTU study showed that supporting the left ventricle with an Impella CP pump for more than 40 minutes before blood flow is restored neither reduced nor increased heart muscle damage.
“Findings from the trial support future studies on whether pre-PCI treatment with Impella CP opens a window of time that enables delivery of adjunct pharmacotherapies designed to reduce reperfusion injury,” the company said.