Dive Brief:
- Philips said Thursday it has received 510(k) clearance for a heart procedure visualization tool it developed with Edwards Lifesciences.
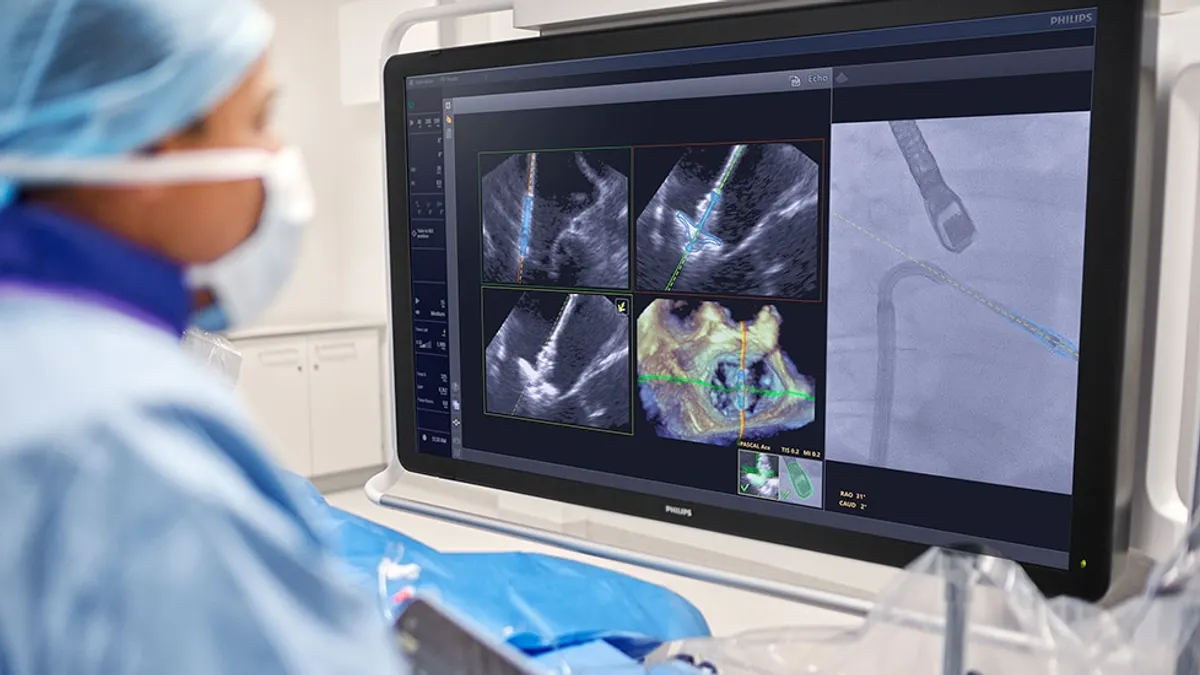
- The system, called EchoNavigator R5.0 with DeviceGuide, uses artificial intelligence to enable surgeons to track and visualize mitral valve repair devices during minimally invasive heart procedures.
- Philips said the system is intended for use with Edwards’ Pascal Ace mitral valve repair system, which competes with Abbott’s MitraClip for the transcatheter edge-to-edge repair market.
Dive Insight:
Physicians use X-ray fluoroscopy and 3D transesophageal echocardiography to visualize the heart and device during TEER procedures. However, the technologies create two separate images. The surgical team must interpret where the device is in the body based on the two images, potentially leading to miscommunication and suboptimal device placement.
Philips designed its software to combine the outputs of the two imaging modalities to continuously show the location of the device in the body. By visualizing the device in the ultrasound image, the companies aim to enable surgeons to more consistently place the repair system in the optimal location.
A paper published this month reported the experiences of physicians who used the system to treat 51 patients. More than two-thirds of users said the software was helpful or very helpful. The 4% of users who said the system was not helpful mainly had device recognition issues in patients with multiple heart implants. Teams said the software improved discussions about optimal trajectory in 85% of cases.
The Food and Drug Administration clearance, which updates a product that originally launched in 2012, comes four months after Philips said it was working with Edwards. Philips had already filed for 510(k) clearance when it disclosed the alliance. The collaboration combined Philips’ imaging and AI capabilities with Edwards’ valvular heart therapy expertise.
Bert van Meurs, chief business leader of image-guided therapy at Philips, said at the company’s capital markets day last month that DeviceGuide is “a concrete example of how we make emerging complex therapies easier to adopt and scale.” Developing such products is only possible by partnering with leading companies in the field, such as Edwards, van Meurs said.