Dive Brief:
-
Polares Medical has raised $40 million to pursue a fast-growing cardiovascular opportunity targeted by Abbott, Boston Scientific, Edwards Lifesciences and Medtronic.
-
The Swiss company, which has U.S. operations in Palo Alto, California, said in a statement posted Thursday it will use the money to finish pilot clinical studies of a transcatheter mitral valve hemi-replacement device in mitral regurgitation patients.
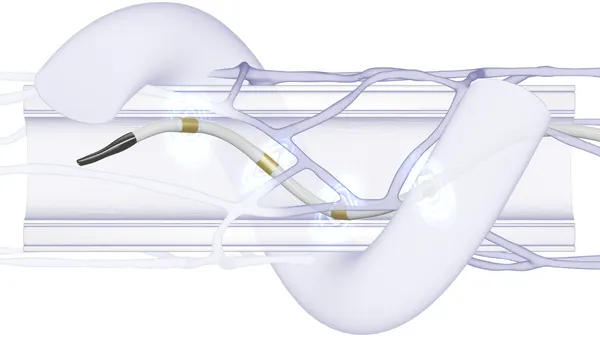
- The focus puts Polares up against a clutch of other medtech companies that are seeking to build on the success of Abbott’s transcatheter mitral valve repair device MitraClip.
Dive Insight:
Medtech companies have bet billions of dollars on the mitral regurgitation market over the past 10 years or so. Abbott made an early move by acquiring Evalve in 2009, setting it up to turn mitral valve repair device MitraClip into a key growth driver a decade later. Boston Scientific, Edwards, Medtronic and Abbott itself have all subsequently made significant mitral valve repair and replacement acquisitions.
Polares has persuaded investors it can carve out a niche in that competitive landscape. A syndicate of U.S. and European investors collectively put up $40 million to run clinical trials on both sides of the Atlantic. The financing comes two years after Polares raised $25 million to bring its technology to clinical evaluation.
The new funding will support development of a prosthetic implant that replaces the posterior leaflet, a flap of the mitral valve that when functioning correctly helps stop blood flowing backward through the heart. By inserting a prosthetic posterior flap, Polares believes it can improve the interaction with the native anterior leaflet, thereby stopping blood from flowing the wrong way.
Polares’ device grew out of the work of Michael Lesh, a cardiologist and serial entrepreneur behind startups bought by companies including Johnson & Johnson. Lesh founded Middle Peak Medical to go after mitral regurgitation in 2011. Symetis bought the startup in 2017, only to spin the technology out to create Polares when Boston Scientific acquired the rest of its business for $435 million.
Throughout that time, the various owners of the technology have said relatively little about how it is positioned against other devices. But a patent filing outlines the need for a device that directly improves interactions between the leaflets, rather than acts indirectly on them, but “does not disrupt leaflet anatomy via fusion or otherwise.”
Polares is currently testing its device in a 12-subject observational study and said early results have “demonstrated the feasibility” of its approach. If the device comes through larger tests, Polares will position itself to enter a market that is growing at a double-digit rate. Growth could accelerate still further as payers permit the use of valve repair and replacement in additional patient populations.