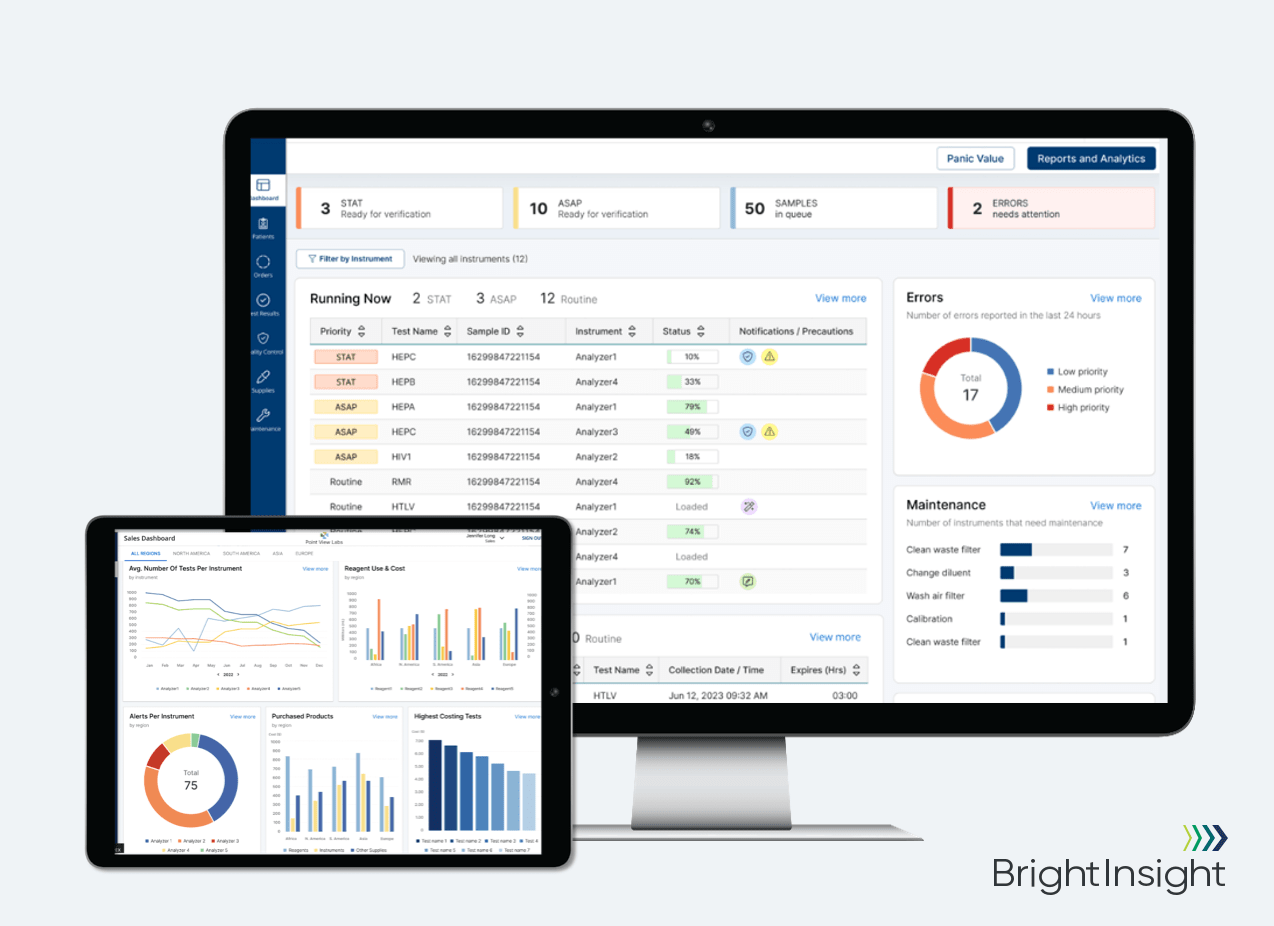
BrightInsight, Inc., provider of the leading global platform for biopharma and medtech regulated digital health solutions, today announced the launch of its digital Connected Diagnostics Platform at the 2022 AACC Annual Scientific Meeting and Clinical Lab Expo. This comprehensive, end-to-end platform is a one-stop shop for in vitro diagnostics manufacturers that streamlines workflows, accelerates revenue growth and drives innovation.
The Connected Diagnostics Platform is four powerful tools in one, and includes a Proxy Agent, Analytics Dashboards, Integration Middleware and Workflow Portals (for the IVD manufacturer and the Lab), all running on the compliant and secure BrightInsight Platform. BrightInsight eliminates the need for multiple products and partners, enabling accelerated time to market and minimizing risk with our best-in-class cybersecurity, privacy compliance and global scalability.
“The BrightInsight Connected Diagnostics Platform is a one-stop solution for in vitro diagnostics manufacturers to maximize workflow and visibility, manage devices more efficiently and reduce costs,” said Kal Patel, M.D., CEO and Co-Founder. “Our four-in-one platform provides unprecedented access to Real-World Data and actionable insights, enabling greater product innovation, new revenue streams and streamlined workflows. BrightInsight is the healthcare industry’s trusted digital partner with a proven track record of helping biopharma and medtech companies build transformational digital solutions at scale.”
The global in vitro diagnostics market is projected to grow nearly 50 percent from $71 billion in 2022 to $107 billion in 2029. And since 60 to 70 percent of healthcare decisions are influenced by IVD test results, it is critical for IVD manufacturers to manage their devices and data securely and reliably. The Connected Diagnostics Platform is designed to meet the demands of IVD manufacturers.
BrightInsight welcomes Stephen N. Oesterle, M.D. to the BrightInsight Advisory Council. Dr. Oesterle is a renowned expert in digital and medtech who serves on the Board of Baxter International, Paragon 28, Peija Medical and Sigilon Therapeutics, Inc. Prior to advising public equity and operating companies in the healthcare industry, from 2002 to 2015, Dr. Oesterle was a member of the Executive Committee of Medtronic plc, serving as Senior Vice President of Medicine and Technology.
“I’m delighted to join the BrightInsight Advisory Council as the company introduces its latest digital platform innovation designed specifically to enable in vitro diagnostics manufacturers to accelerate time to market for digital solutions that will improve workflows. BrightInsight was a pioneer and is now a leading digital platform for medtech and biopharma, building regulated digital solutions that will enable greater data insights and analysis, to drive improved outcomes,” said Dr. Oesterle.
Dr. Oesterle joins the group of esteemed healthcare, technology and business thought leaders working with the BrightInsight Leadership Team to advance the company’s vision to transform patient outcomes globally through the power of digital technology.
BrightInsight provides the leading global platform for biopharma and medtech regulated digital health solutions. When speed matters, we help companies accelerate time to market for regulated digital health offerings across therapeutic areas, including apps, healthcare provider interfaces, analytics dashboards, algorithms, medical devices, connected combination products, diagnostics and Software as a Medical Device (SaMD). BrightInsight replaces the need for lengthy and complex ‘build from scratch’ implementations by offering configurable software solutions and a proven platform built on Google Cloud under a Quality Management System to support global security, privacy and regulatory requirements. When building digital health products on the BrightInsight Platform, compliance is future-proofed as intended use changes scale across geographies.
For more information, visit BrightInsight’s website, Blog, Twitter, and LinkedIn pages.