Clinical Trials
-
J&J’s Impella heart pump shows no patient benefit in 2 trials
Study investigators cautioned against routine use of the device in complex percutaneous coronary interventions and to treat heart attack patients who don’t have cardiogenic shock.
By Susan Kelly • April 1, 2026 -
Medtronic to study renal denervation combined with PCI
Medtronic detailed plans to further study the blood pressure treatment while releasing additional data from the SPYRAL HTN trials that showed the procedure reduced hypertensive emergencies.
By Susan Kelly • March 31, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Courtesy of Intuitive Surgical
Courtesy of Intuitive Surgical Trendline
TrendlineTop 5 stories from MedTech Dive
From the top medtech trends to watch in 2026 to haphazard layoffs at the Food and Drug Administration and the evolving use of AI in the medtech sector, here is a collection of top stories from MedTech Dive.
By MedTech Dive staff -
Boston Scientific posts positive stroke prevention data for Watchman
The company said it will seek an expanded indication for the left atrial appendage closure device after it met all safety and efficacy goals in the closely watched CHAMPION-AF study.
By Susan Kelly • March 30, 2026 -
Glucotrack plans US trial of implantable glucose monitor this year
Glucotrack plans to file for an investigational device exemption in the second quarter, with hopes of starting a trial in the second half of 2026.
By Elise Reuter • March 27, 2026 -
Thermo Fisher completes $8.9B buyout of Clario
Thermo Fisher plans to create a fast-growing business by integrating Clario’s data capture and management system with its existing capabilities.
By Nick Paul Taylor • March 25, 2026 -
Insulet posts clinical data on fully closed-loop insulin delivery system
The company aims to start a pivotal trial this year, file for 510(k) clearance in 2027 and launch the device in 2028.
By Nick Paul Taylor • March 12, 2026 -
Zimmer Biomet shares smart knee data at AAOS
The orthopedic firm published data showing patients with its Persona IQ implant had lower revision and physical therapy rates than those who received traditional knee implants.
By Elise Reuter • March 6, 2026 -
Sponsored by Infineon
Transforming epilepsy care: How innovative implantable neurotechnology offers new hope
Minimally invasive brain stimulation may be the new frontline treatment for drug-resistant patients.
Feb. 23, 2026 -
Grail’s multi-cancer early detection test misses study goal
Shares in the company lost about half their value in Friday trading after the clinical trial failed to demonstrate a statistically significant reduction in Stage III and IV cancers.
By Susan Kelly • Feb. 20, 2026 -
4 takeaways from the 2026 AF Symposium
Johnson & Johnson, Boston Scientific and Abbott were among the companies showcasing new data on atrial fibrillation treatments, including pulsed field ablation catheters.
By Susan Kelly • Feb. 10, 2026 -
Abbott details Volt, TactiFlex data; Pulse Biosciences’ results shine
Abbott and Pulse released their findings at the AF Symposium that wrapped this weekend in Boston.
By Susan Kelly • Feb. 9, 2026 -
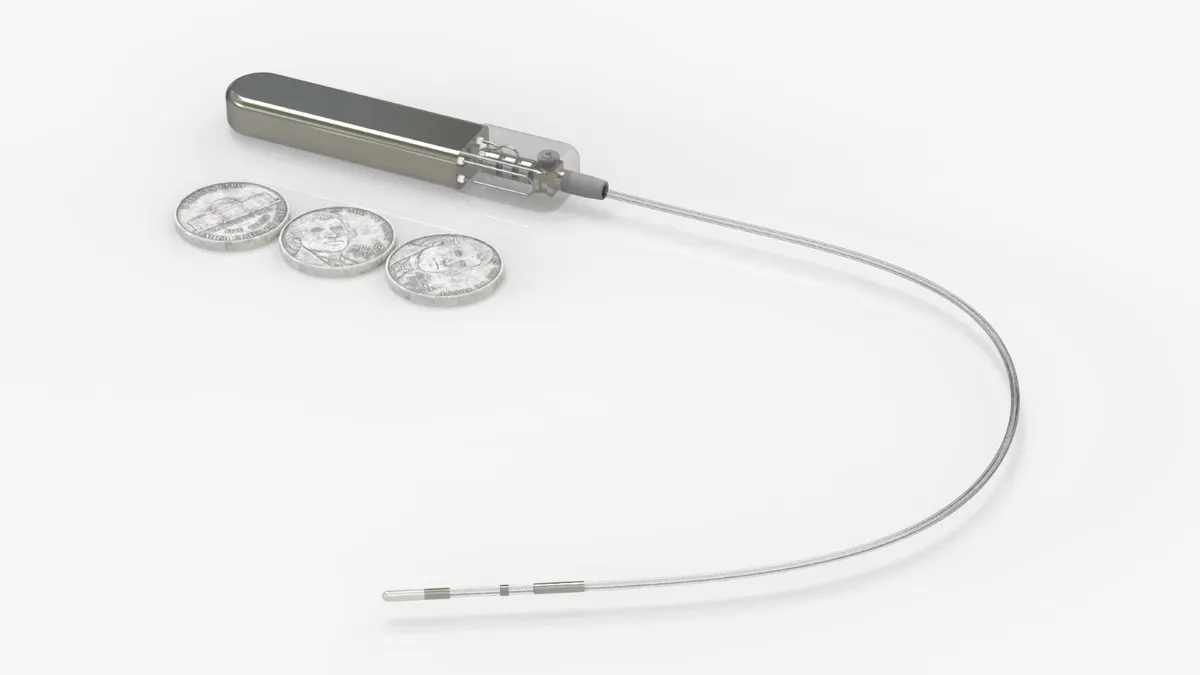
Pulse Biosciences wins FDA approval to begin PFA catheter study
Shorter, nanosecond pulses for treating atrial fibrillation differentiate the company’s approach from existing pulsed field ablation systems on the market.
By Susan Kelly • Dec. 18, 2025 -
 Retrieved from AtriCure on December 12, 2025
Retrieved from AtriCure on December 12, 2025
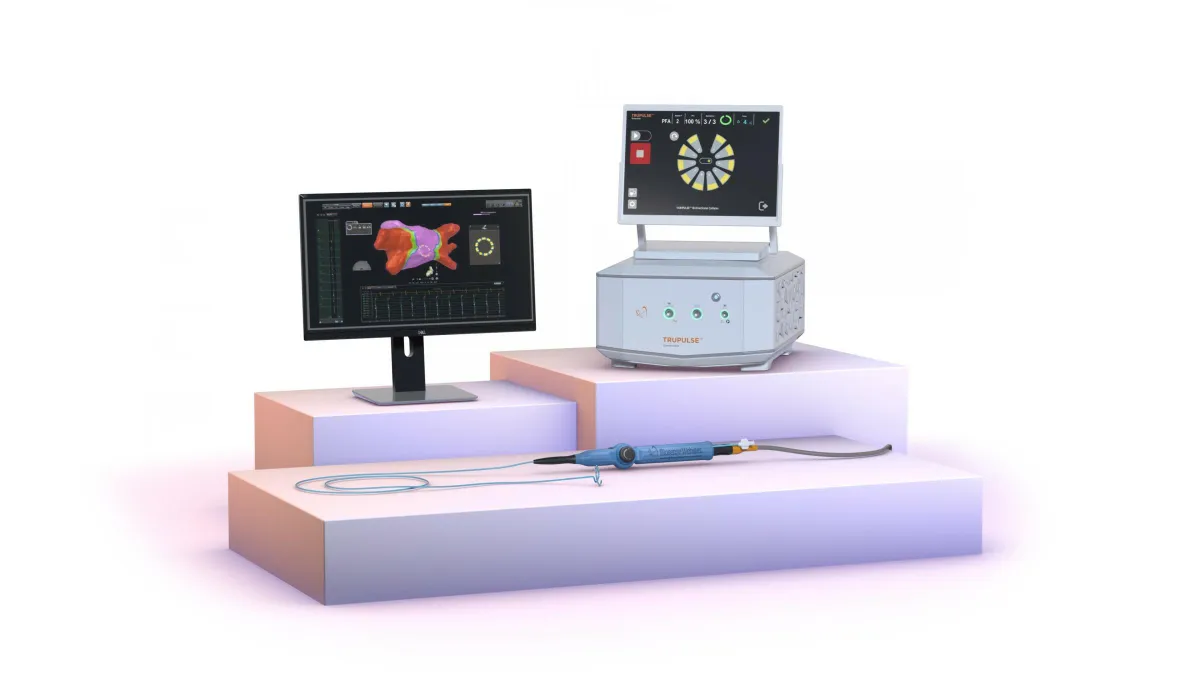
AtriCure treats first patients with dual PFA/RF system
Combining pulsed field ablation and a radiofrequency technique in one platform is expected to reduce procedure times.
By Susan Kelly • Dec. 12, 2025 -
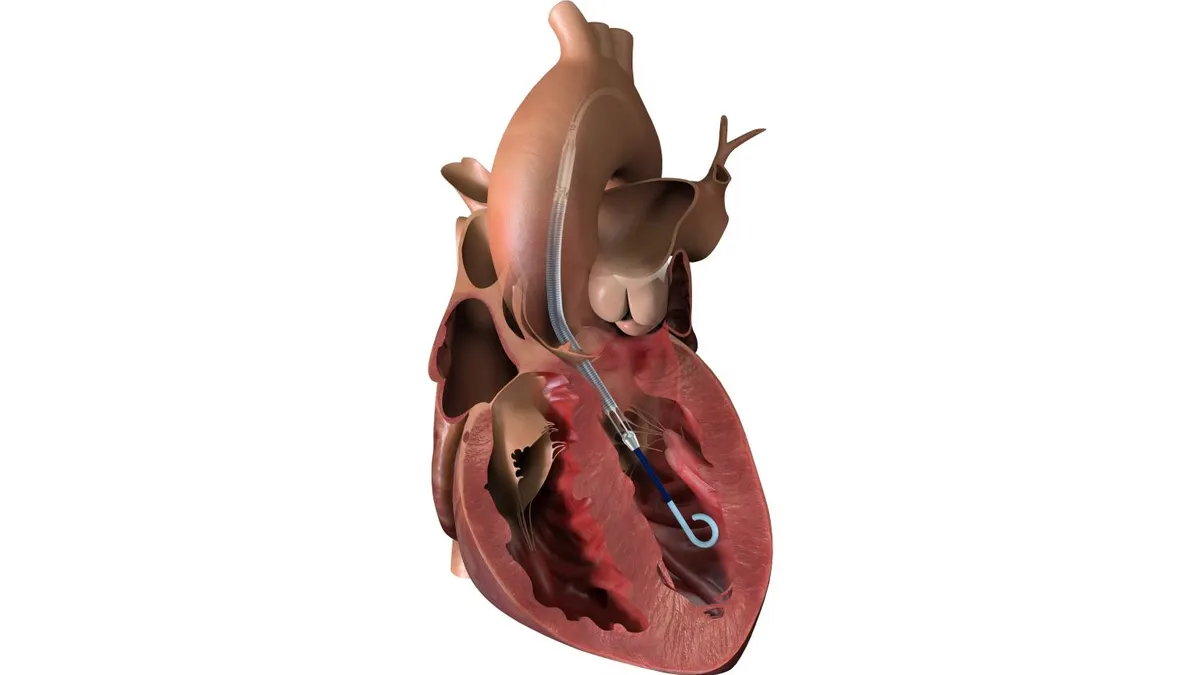
FDA advisory panel votes unanimously against J&J heart shunt
Panelists focused on study data that showed the implant did not improve outcomes in heart failure patients.
By Susan Kelly • Dec. 5, 2025 -
MMI tackles Alzheimer’s; SS Innovations advances telesurgery
MMI won FDA approval to study robotic surgery for Alzheimer’s disease. Elsewhere, SS Innovations’ CEO performed remote bypass surgery with a new console, and Procept BioRobotics’ Q3 revenue rose 43%.
By Susan Kelly • Nov. 10, 2025 -
Sponsored by IQVIA MedTech
Designing smarter trials for new devices: 3 best practices for developers
Discover three strategies for MedTech companies to design streamlined, effective trials and how quality data powers smarter study design.
Nov. 3, 2025 -
3 takeaways from TCT 2025
Trials from Edwards, Philips and Penumbra were among the most-watched studies at the annual cardiovascular meeting.
By Susan Kelly • Oct. 31, 2025 -
Thermo Fisher to acquire clinical trial data firm Clario for $8.9B
The purchase is expected to complement Thermo’s clinical research services as companies are conducting more trials.
By Elise Reuter • Oct. 29, 2025 -
Medtronic links renal denervation to 3-year blood pressure reduction
Patients treated with Symplicity Spyral had significantly lower blood pressure than people who received a sham treatment after three years.
By Nick Paul Taylor • Oct. 28, 2025 -
Ōura to pursue FDA clearance of blood pressure feature for smart ring
The company has received approval to validate and define the feature in a study that is scheduled to start this year.
By Nick Paul Taylor • Oct. 20, 2025 -
Medtronic begins study of Hugo robot in gynecology
With positive trial results in urology and hernia repair already under its belt, the company is getting closer to taking on Intuitive Surgical in the U.S. robotic surgery market.
By Susan Kelly • Oct. 9, 2025 -
Boston Scientific unveils plans for new Watchman device
The company hopes to launch its next-generation Watchman system in the second half of 2027 or early 2028.
By Elise Reuter • Updated Oct. 14, 2025 -
J&J launches IVL device in Europe; Medtronic looks at pacing for more patients
J&J’s Shockwave Javelin intravascular lithotripsy catheter treats people with peripheral artery disease. Elsewhere, Medtronic began a pivotal trial to evaluate its pacemakers in a new patient group.
By Susan Kelly • Sept. 18, 2025 -
Pulse wins IDE approval; Galvanize pulls in $100M
Pulse Biosciences will study PFA in cardiac surgery. Meanwhile, Galvanize Therapeutics also named a new CEO, and PFA pioneer Steven Mickelsen has launched another company.
By Susan Kelly • Sept. 9, 2025 -
Sponsored by Veeva
Smith+Nephew improves evidence generation by aligning clinical and medical affairs
Clinical teams focus on generating evidence for a product's safety and efficacy, while medical affairs teams act as a bridge to the healthcare community.
Sept. 8, 2025