Digital Health
-
Q&A
Nvidia’s David Niewolny on the future of AI in medical devices
Nvidia, which is already working with J&J and Medtronic, plans to partner with medtech companies to build platforms for AI-enabled robotics, imaging and other features.
By Elise Reuter • June 2, 2025 -
Sponsored by COMSOL, Inc.
Simulation app introduces personalized oncology care
See one company's idea for bringing personalization and precision to oncology care
By Dixita Patel • May 19, 2025 -
FDA, aiming to speed scientific reviews, names chief AI officer
Commissioner Martin Makary also said the agency would roll out a generative AI system across its centers that will help FDA scientists spend less time on repetitive tasks.
By Susan Kelly • May 9, 2025 -
Abbott integrates diabetes data into Epic’s electronic health records
Linking the systems will allow clinicians to view glucose data captured by Libre glucose sensors within Epic’s systems.
By Nick Paul Taylor • May 1, 2025 -
Senseonics, Sequel partner to use 1-year CGM in automated insulin dosing system
The partnership extends the compatibility of Sequel’s Twiist system to allow people with Type 1 diabetes to pair the device with Senseonics’ Eversense 365 CGM.
By Nick Paul Taylor • April 30, 2025 -
Kandu Health merges with BCI company Neurolutions
The merger brings together Neurolutions’ brain computer interface technology and Kandu Health’s telehealth services to try to improve stroke care.
By Nick Paul Taylor • April 9, 2025 -
Eargo, Hearx merge into one OTC hearing aid firm, receive $100M boost
Hearx, the maker of Lexie hearing aids, and Eargo competed for the OTC hearing aid market created by the FDA in 2022.
By Nick Paul Taylor • April 3, 2025 -
Sponsored by Veeva MedTech
Cardinal Health turns complaints into quality improvements
Transform complaint management with a single source of truth.
March 31, 2025 -
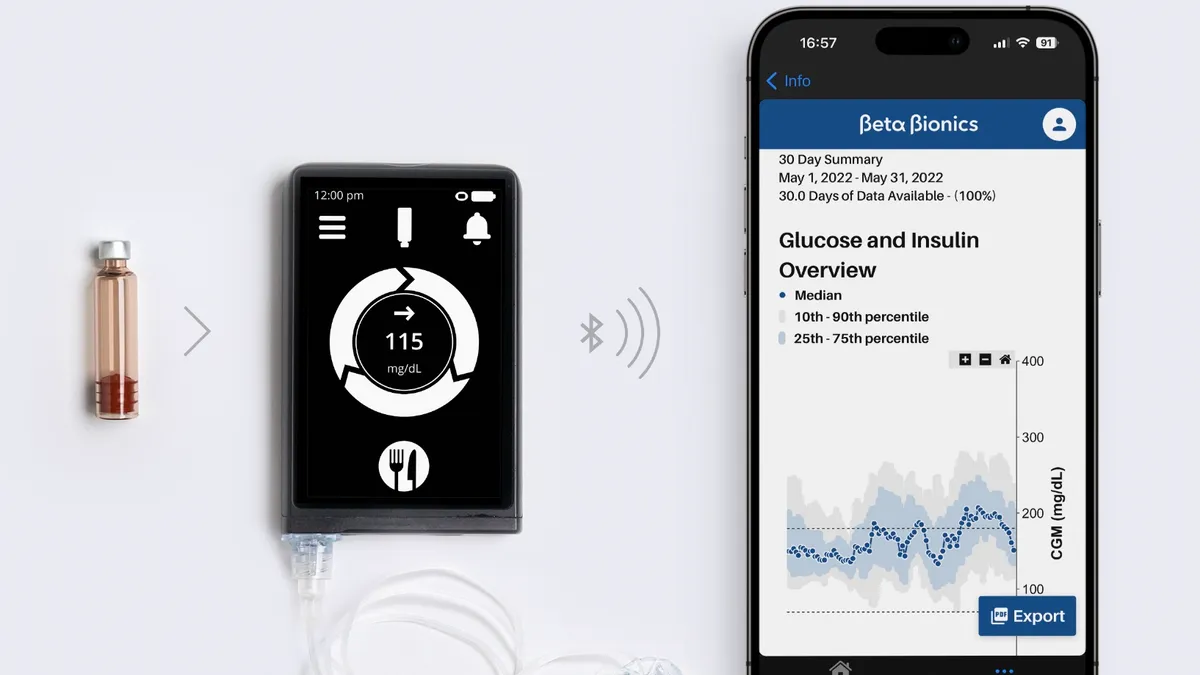
Newly public Beta Bionics reports sales surge; Dexcom hires CCO
Sales of Beta Bionics insulin pumps spiked 145% in the fourth quarter behind solid new patient starts. Elsewhere, former Masimo exec Jon Coleman joined Dexcom, and Sibionics’ CGM earned the CE mark.
By Nick Paul Taylor • March 28, 2025 -
Deep Dive
5 tips for building a predetermined change control plan
The new framework is intended to make postmarket changes easier for products. Experts recommend having a clear roadmap and contacting regulators early.
By Elise Reuter • March 25, 2025 -
Hospital-at-home companies DispatchHealth, Medically Home to merge
DispatchHealth CEO Jennifer Webster will lead the combined company once the deal closes, which is expected in mid-2025.
By Emily Olsen • March 21, 2025 -
News roundup
Quest, Google team on gen AI; GE Healthcare plans autonomous imaging
GE Healthcare, Synchron and robotics companies were among the medtech firms that used Nvidia’s GTC 2025 conference to share updates on their work with the AI computing leader.
By Nick Paul Taylor • Updated March 20, 2025 -
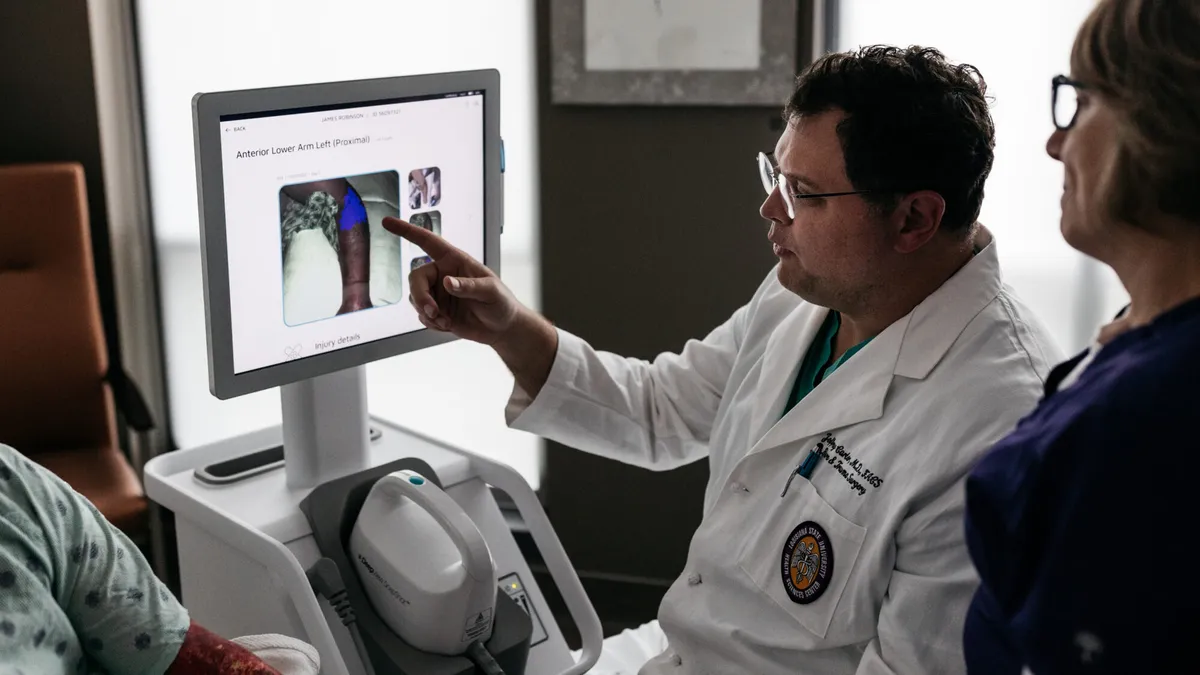
 Retrieved from Spectral AI on March 18, 2025
Retrieved from Spectral AI on March 18, 2025
Spectral AI algorithms beat physicians in burn tissue assessments
The company plans to file the results with the FDA by mid-2025 and launch the product in 2026.
By Nick Paul Taylor • March 18, 2025 -
Hinge Health, a virtual physical therapy firm, files for IPO
The digital health IPO market has slowed to a crawl in recent years, but experts say it could be poised for a turnaround in 2025.
By Emily Olsen • March 13, 2025 -
ECRI warns insufficient AI governance threatens patient safety
ECRI included other medtech-related threats on its list of patient safety concerns, noting that cybersecurity breaches can delay care and diagnostic errors can lead to improper treatment.
By Nick Paul Taylor • March 11, 2025 -
Q&A
Medtronic’s Orleigh Bogle on changing the culture of surgical training
Bogle, Medtronic’s head of medical affairs for digital surgery, also discussed the company’s plans to train surgeons for its Hugo surgical robot.
By Elise Reuter • Feb. 6, 2025 -
Troy Tazbaz, CDRH digital health leader, resigns
Tazbaz was director of the FDA’s Digital Health Center of Excellence for two years, leading key policies on artificial intelligence and software as a medical device.
By Elise Reuter • Jan. 31, 2025 -
Sponsored by Altium365
What’s next for medical device hardware development?
Discover how cloud-based tools and AI are transforming collaboration, innovation and speed to market.
Jan. 27, 2025 -
Deep Dive
Top healthcare technology trends in 2025
How the incoming Trump administration will regulate AI this year remains unclear. Meanwhile, experts say healthcare companies will continue bolstering cyber defenses to withstand increasing attacks.
By Emily Olsen • Jan. 17, 2025 -
Eko Health names Justin Spencer as CFO
Spencer’s arrival at Eko comes months after the digital health company appointed his former colleague Brent Lang to its board of directors.
By Nick Paul Taylor • Jan. 10, 2025 -
4 takeaways from the FDA’s first digital health advisory committee
Industry and patient representatives debated how the FDA should regulate generative AI in medical devices and address new challenges with the technology, such as the use of complex models that can change quickly.
By Elise Reuter • Dec. 12, 2024 -
AI-enabled apps top ECRI’s list of health tech hazards in 2025
Other health tech hazards on ECRI’s annual list include the unmet technology support needs of home care patients, cybersecurity threats and substandard or fraudulent medical devices.
By Nick Paul Taylor • Dec. 9, 2024 -
FDA issues final guidance on postmarket updates to AI-enabled devices
By using pre-determined change control plans, manufacturers can update AI-enabled devices without filing an additional FDA submission.
By Elise Reuter • Dec. 4, 2024 -
Movano receives FDA nod for smart ring’s pulse oximeter feature
Movano Health plans to market the Evie Ring to organizations that run clinical trials and healthcare companies that are helping patients manage chronic diseases.
By Nick Paul Taylor • Dec. 3, 2024 -
Sponsored by IFS
The power of composability: Overcoming field service management challenges in MedTech
Composable software seamlessly integrates with core systems, facilitating communication and real-time data sharing between diverse systems.
Dec. 2, 2024