Digital Health: Page 5
-
Q&A
ONC’s Micky Tripathi on laying the digital floor for healthcare AI
The agency head discussed ONC’s accomplishments over the past two decades, artificial intelligence opportunities and improving the documentation burden among clinicians.
By Emily Olsen • June 20, 2024 -
FDA official sets out approach to AI in medical devices
Developing quality assurance practices for AI models should be a priority, said Troy Tazbaz, director of the CDRH’s Digital Health Center of Excellence.
By Elise Reuter • June 18, 2024 -
 Explore the Trendline➔
Explore the Trendline➔
 Courtesy of Intuitive Surgical
Courtesy of Intuitive Surgical Trendline
TrendlineTop 5 stories from MedTech Dive
From the top medtech trends to watch in 2026 to haphazard layoffs at the Food and Drug Administration and the evolving use of AI in the medtech sector, here is a collection of top stories from MedTech Dive.
By MedTech Dive staff -
Senators urge CMS to create AI payment pathway
Uniform reimbursement standards are needed to protect patient access to algorithm-based devices and ensure future innovation, the lawmakers said.
By Nick Paul Taylor • June 18, 2024 -
FDA creates transparency principles for AI in medical devices
The agency worked with regulators in the U.K. and Canada to determine how key information about machine learning-enabled devices should be communicated.
By Nick Paul Taylor • June 17, 2024 -
Zimmer, RevelAi partner to sell AI tool for orthopedic care
RevelAi uses generative artificial intelligence to automate clinical workflows and support patients with conditions such as osteoarthritis.
By Nick Paul Taylor • Updated June 14, 2024 -
Q&A
Stronger oversight of AI needed in medical devices, ECRI CEO says
Marcus Schabacker called for more upfront regulations and postmarket monitoring to better understand how AI features affect patient care.
By Elise Reuter • June 5, 2024 -

 Retrieved from Dexcom on June 05, 2024
Retrieved from Dexcom on June 05, 2024
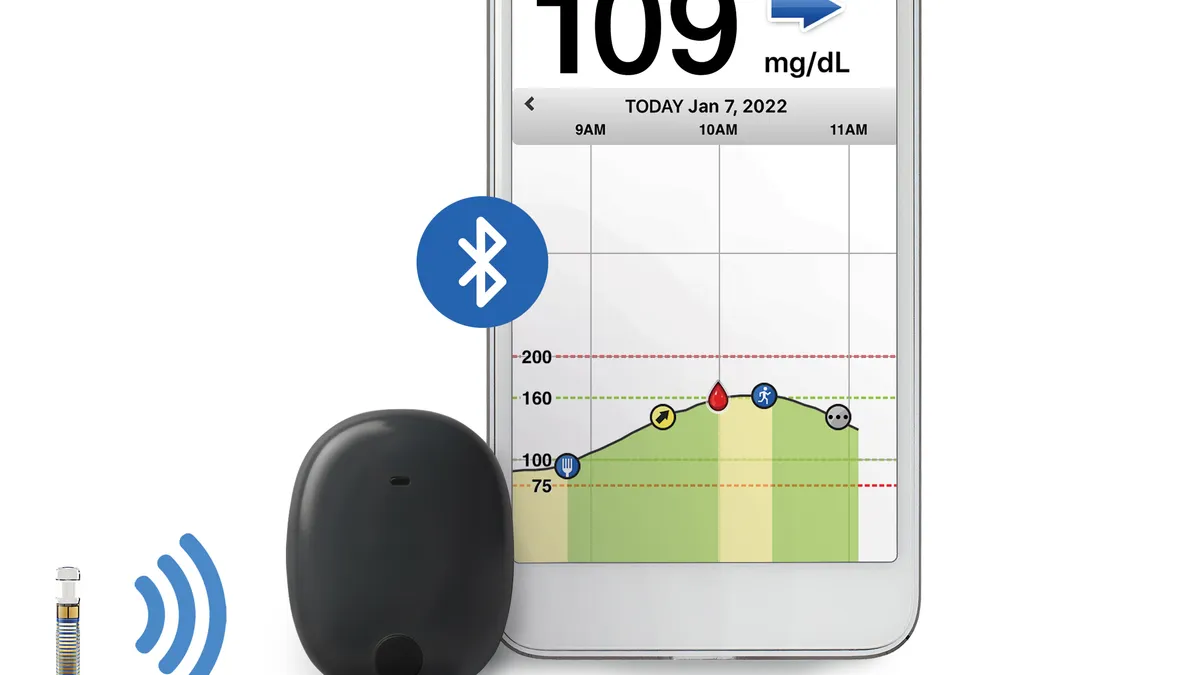
Dexcom connects G7 CGM directly to Apple Watch in US
Apple Watch and G7 users can now view real-time glucose readings on their wrist, regardless of whether they are carrying an iPhone.
By Nick Paul Taylor • Updated June 7, 2024 -
Digital therapeutics firm Akili to sell for $34M
Akili, which makes a video game treatment for ADHD, will merge with Virtual Therapeutics to create a larger digital health entity.
By Elise Reuter • May 30, 2024 -
Otsuka defies digital health downturn with new company
With “no playbook out there” to fall back on, the new company is taking a long-term approach as it develops its portfolio, an Otsuka Precision Health exec told MedTech Dive.
By Elise Reuter • May 29, 2024 -
FDA seeks AI tech to predict, detect Parkinson’s symptoms
By testing artificial intelligence and machine learning models, the agency hopes to come up with best practices for Parkinson’s measures from wearables and smartphones.
By Elise Reuter • May 21, 2024 -
FDA targets health inequality by improving at-home devices
Michelle Tarver, an FDA official, said a new program is meant to advance health equity by considering people’s living conditions as part of medical device design.
By Elise Reuter • May 13, 2024 -
Tandem reports 224 injuries related to faulty insulin pump app
A problem with Tandem’s iPhone app could drain the battery of the connected insulin pump, causing it to power down sooner than expected.
By Nick Paul Taylor • May 9, 2024 -
FDA qualifies Apple Watch AFib feature for use in clinical trials
Officials will accept atrial fibrillation data collected by the wearable as a secondary endpoint in studies of cardiac ablation devices.
By Nick Paul Taylor • May 6, 2024 -
UK sets out position on regulating AI as a medical device
Some AI products that currently can be put on the market without conformity assessment will move to a higher risk class, requiring more scrutiny.
By Nick Paul Taylor • May 3, 2024 -
Senseonics gets FDA clearance to pair CGM implant with insulin pumps
Analysts said the designation and planned one-year sensor could help Senseonics’ device appeal to more patients.
By Nick Paul Taylor • May 1, 2024 -
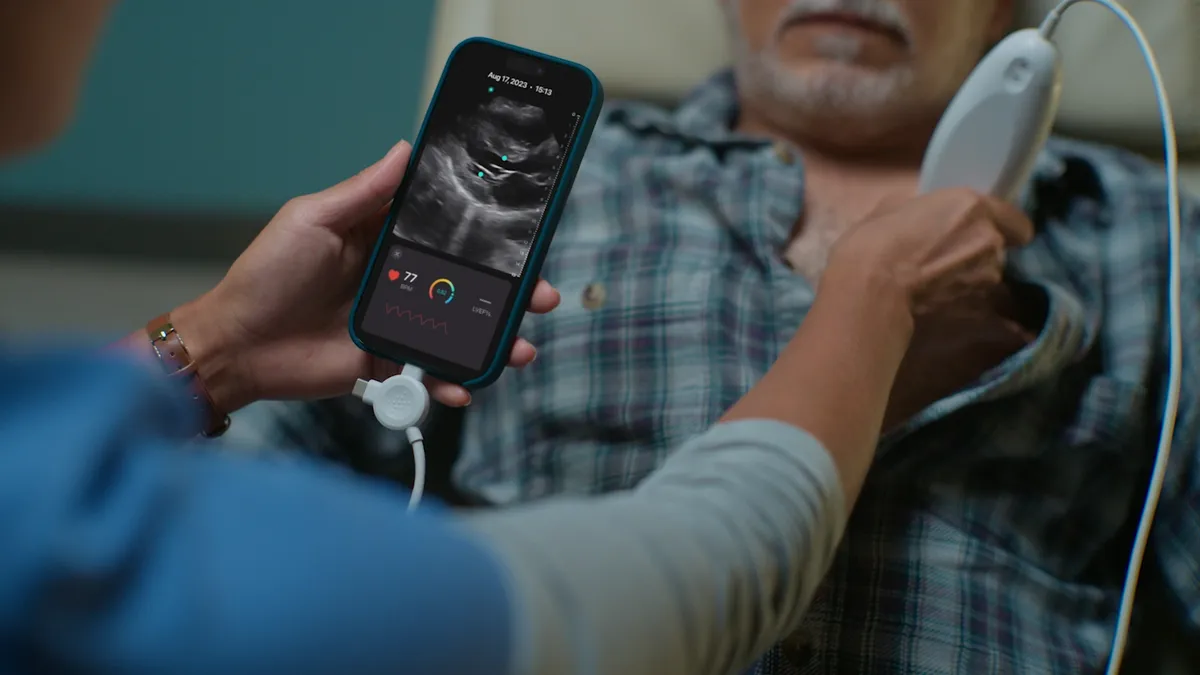
Exo adds FDA-cleared AI tools to handheld ultrasound system
Exo sees the new capabilities as especially helpful for healthcare providers in rural and under-resourced settings.
By Nick Paul Taylor • April 25, 2024 -
Osso VR brings surgery training app to Apple Vision Pro
Users can virtually walk through the steps of two orthopedic procedures: total knee replacement and carpal tunnel release.
By Nick Paul Taylor • April 12, 2024 -
Synchron launches patient registry to prepare for brain implant trial
The startup, which rivals Neuralink, is developing brain-computer interface technology that lets people control electronic devices hands-free.
By Elise Reuter • April 9, 2024 -
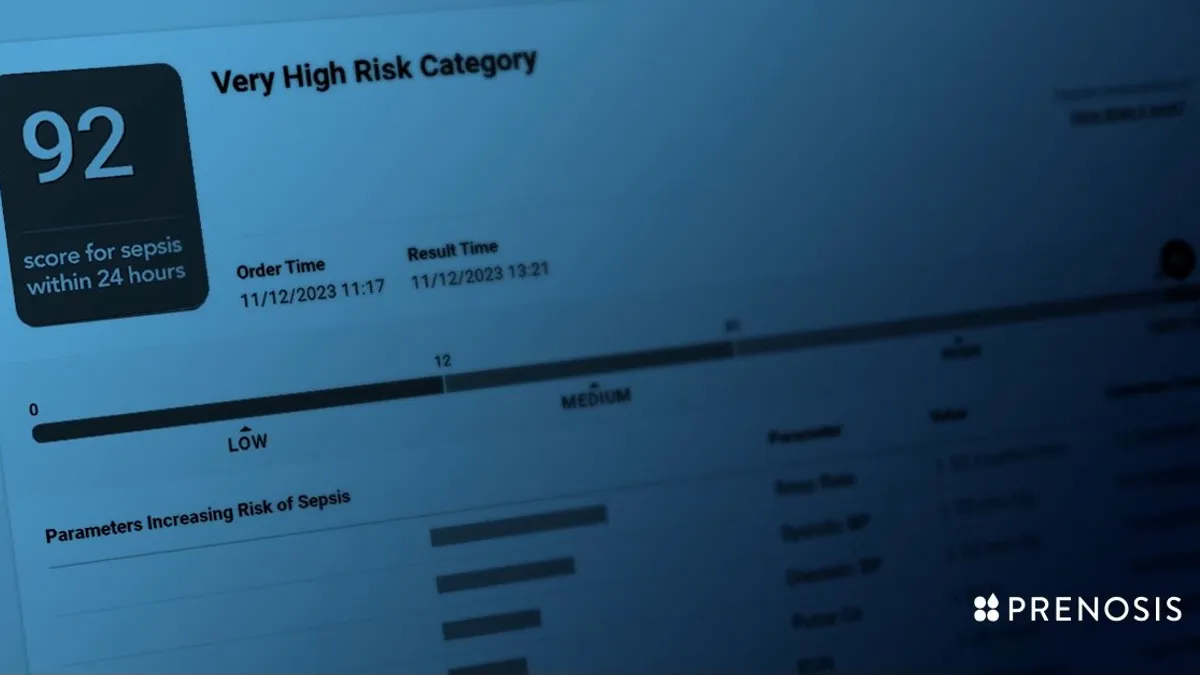
FDA grants de novo nod to AI tool for detecting sepsis
Prenosis CEO Bobby Reddy Jr. told MedTech Dive the company sees third-party validation as important, with the FDA having clarified that certain decision support tools should be regulated as medical devices.
By Elise Reuter • April 4, 2024 -
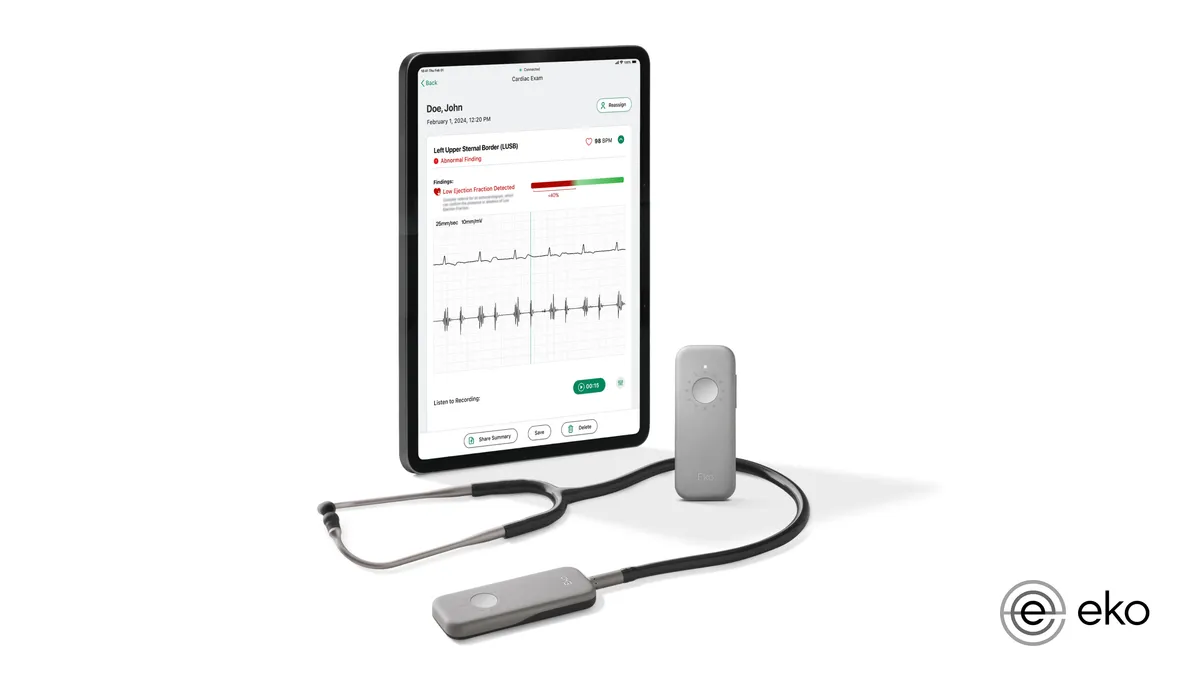
Eko wins FDA nod for AI to detect sign of heart failure using stethoscope
The clearance adds to the list of devices the FDA has authorized this year with AI algorithms to detect health conditions.
By Nick Paul Taylor • April 4, 2024 -
Osso VR to lay off 67 people at corporate HQ
Art and illustration jobs were heavily affected by the layoffs, which will be complete by May 27.
By Nick Paul Taylor • April 1, 2024 -
Deka’s automated insulin delivery system, powered by patient-led app, gets FDA clearance
Sequel Med Tech will sell the new system, which integrates with Tidepool’s Loop insulin dosing algorithm.
By Nick Paul Taylor • March 20, 2024 -
Better Therapeutics lays off staff, considers winding down business
The digital therapeutics developer received authorization for a Type 2 diabetes treatment app last year but has struggled for money.
By Nick Paul Taylor • March 15, 2024 -
AI to expand medtech portfolios, revenue streams: Moody’s
The rating agency predicts AI will start to have a positive impact on medical device companies in the next two years.
By Nick Paul Taylor • March 12, 2024 -
Siemens Healthineers launches anatomy hologram app on Apple Vision Pro
The prototype app has potential uses in patient communication, medical education and surgical planning.
By Nick Paul Taylor • March 8, 2024