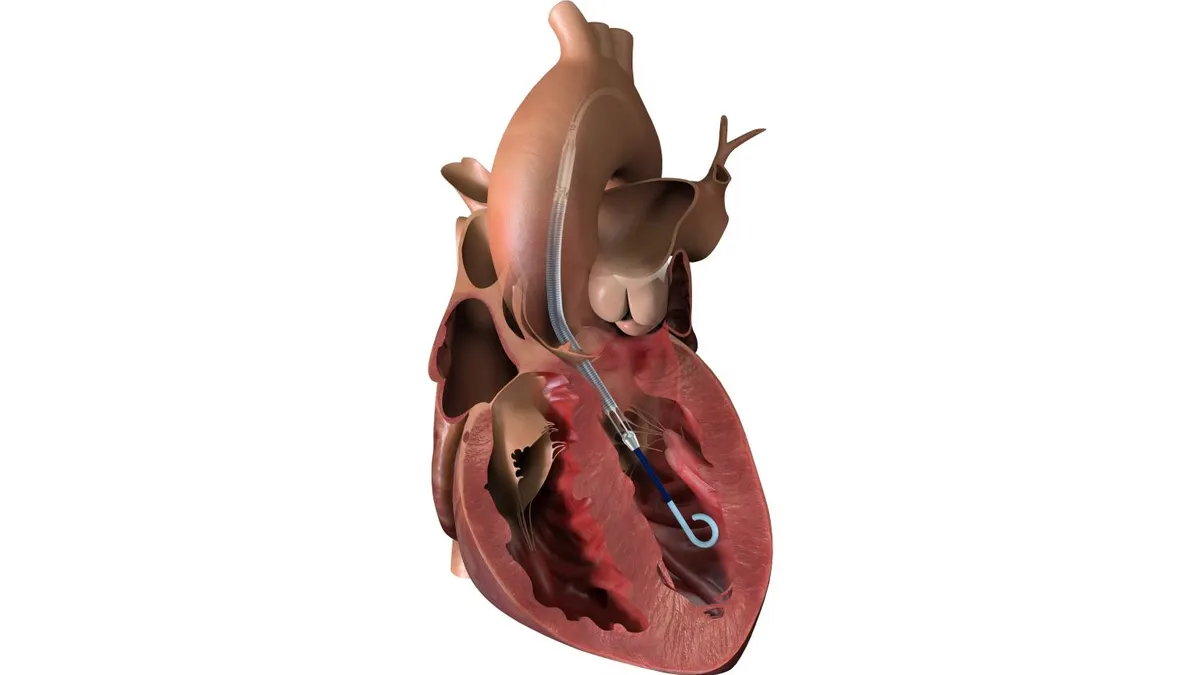
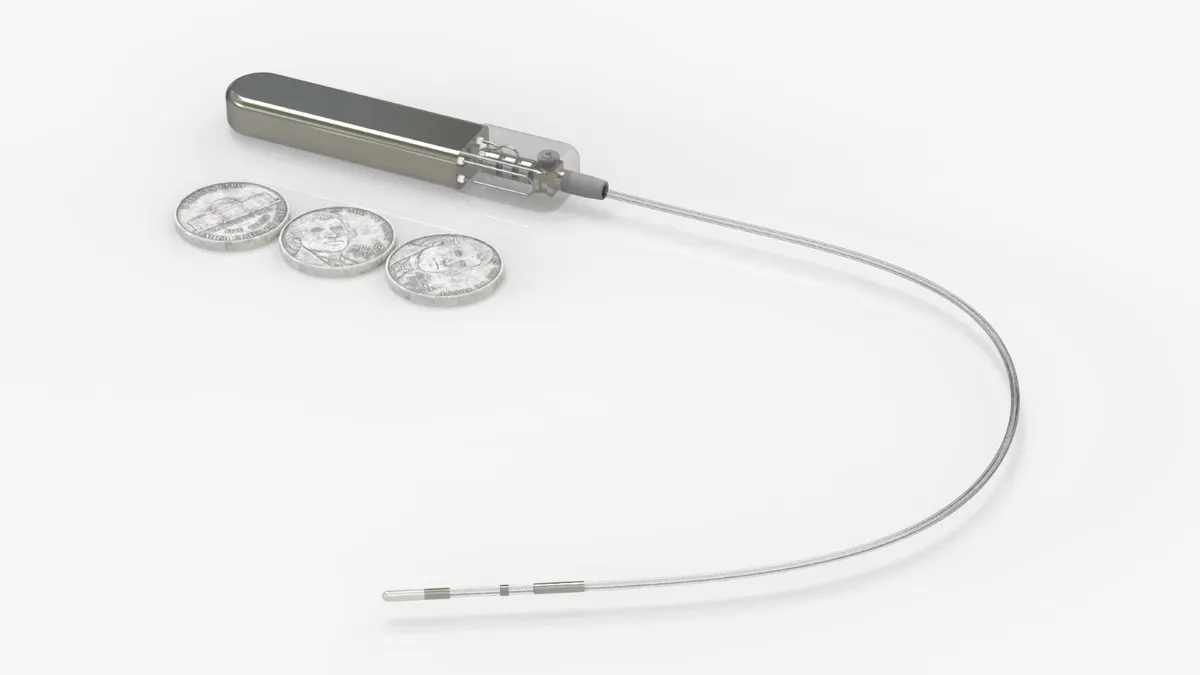
The $16 billion global electrophysiology market is growing fast due to the adoption of new pulsed field ablation treatments for irregular heart rhythm atrial fibrillation, but share shifts are underway.
Analysts who attended the Heart Rhythm Society’s annual meeting this weekend said feedback from companies suggests the electrophysiology market continues to grow at a mid- to high-teens rate, making it one of medtech’s hottest fields. Doctors said PFA has made procedures safer and faster, expanding the number of patients eligible for ablation treatment, Stifel analysts wrote in an investor note.
In PFA, Medtronic appears to be gaining momentum at the expense of rival Boston Scientific, which cut its 2026 sales and earnings forecasts last week. Slower first-quarter growth in PFA was partly to blame.
Competition is intensifying, with Johnson & Johnson and Abbott also in the PFA race.
“With J&J, Abbott, and Boston now all reporting 1Q results, it’s clear Medtronic should be the big winner this quarter,” J.P. Morgan analyst Robbie Marcus told clients Sunday.
Stifel analysts said the physicians they spoke with are increasingly using Medtronic’s technology.
“For the near-term, the physician commentary we heard at the meeting argues most favorably for Medtronic,” they wrote, adding that for the longer term, doctors expect each major PFA player to gain a respectable market position.
Here are four clinical trial updates to know from the HRS meeting:
1. Boston Scientific’s AVANT GUARD trial
Boston Scientific’s Farapulse PFA system worked better than drugs as an initial strategy for treating patients with persistent AFib, researchers found in the AVANT GUARD study.
For patients with intermittent AFib, catheter ablation is established as a safe and superior alternative to antiarrhythmic drugs for first-line therapy, the researchers said. However, patients with persistent AFib often have electrical and structural remodeling, which leads to worse procedure outcomes with thermal-based ablation.
PFA, by delivering electrical pulses to areas of the heart causing abnormal rhythms, offers a potentially safer approach than thermal energy to ablate the tissue.
The Boston Scientific-funded study compared PFA using the Farawave catheter to drug therapy in 440 patients with persistent AFib. The results, published in the New England Journal of Medicine, showed a significantly lower risk of arrhythmia recurrence in patients who received PFA as the first-line treatment. At 12 months, the primary effectiveness rate was about 56% for PFA and 30% for drug treatment.
The study met its primary safety endpoint, with a major adverse event rate of 5.1%. Six deaths that occurred during the 12-month follow-up period were independently adjudicated by a clinical events committee as being unrelated to the procedure or device.
The positive findings should help Boston Scientific secure an expanded indication for Farapulse as a first-line therapy, beyond its current approval for drug-resistant persistent AFib, Truist Securities analysts wrote to clients. They noted Boston Scientific has estimated persistent AFib could be a $1 billion market by 2028.
2. Boston Scientific’s CHAMPION-AF sub-analysis
A sub-analysis of the CHAMPION-AF trial compared left atrial appendage closure with the Watchman FLX implant to non-vitamin K antagonist oral anticoagulants in patients with and without prior ablation for non-valvular atrial fibrillation.
The analysis included 1,434 patients who had a previous cardiac ablation for AFib and 1,565 patients who did not have a prior ablation.
In both arms, the device showed similar efficacy to the blood thinners for occurrence of stroke, death or systemic embolism regardless of whether they had a prior ablation, Boston Scientific said. The rates were 3.9% for both the device and drugs in patients with prior ablation. For patients with no previous ablation, the rates were 7.5% for the device group, compared with 5.7% for the drugs group.
3. Abbott TactiFlex Duo 6-month results
Abbott presented six-month outcomes from its FlexPulse trial assessing use of the TactiFlex Duo dual-energy pulsed field and radiofrequency ablation catheter to treat patients with intermittent AFib.
In the 188-patient investigational device exemption study, 87% of patients reported being free from documented arrhythmias. Abbott said the data showed a high safety profile (98.3%), with no major safety events. The majority of patients (93.3%) were treated exclusively with PFA, the company noted.
The study confirmed the strong safety and efficacy profile seen in the CE mark study for treating complex AFib cases, Abbott said. TactiFlex Duo received Europe’s CE mark in January.
J.P. Morgan analysts said TactiFlex Duo is positioned as a complementary PFA option aimed at higher-complexity cases, with Abbott’s Volt device aimed at new cases.
4. Medtronic’s Sphere-9 in ventricular tachycardia
Medtronic presented six-month data for its Affera mapping and ablation system and Sphere-9 catheter to treat ventricular tachycardia, a potentially life-threatening fast heart rate.
Sphere-9 is a dual-energy pulsed field and radiofrequency ablation catheter. Results from the ongoing early feasibility study showed 65.5% of patients remained free from VT recurrence at six months after treatment with the Medtronic system following a heart attack.
The Food and Drug Administration granted a breakthrough device designation for the Sphere-9 catheter to treat VT and approved Medtronic’s U.S. pivotal trial, the company announced Saturday.