Dive Brief:
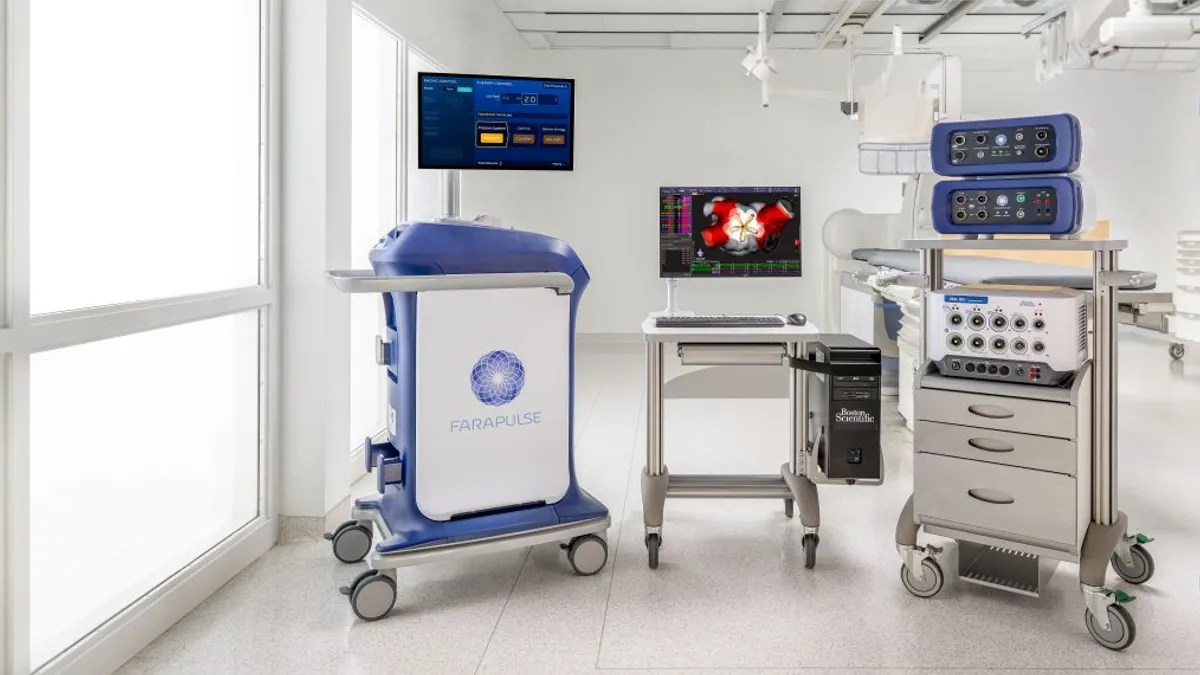
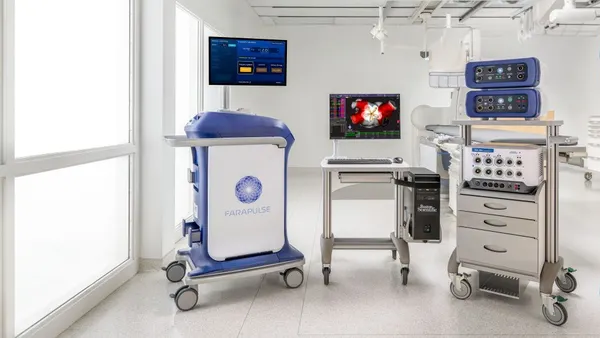
- Boston Scientific said its Farapulse pulsed field ablation platform was granted CE mark approval for an expanded label to include treatment of patients who have persistent atrial fibrillation, an abnormal heart rhythm lasting at least seven days.
- The expanded indication is supported by data from the ADVANTAGE AF clinical trial that showed the safety and effectiveness of Farapulse in patients with the persistent form of AFib, the company said this week in a LinkedIn post.
- Boston Scientific, in the post, called the approval an important step forward in addressing the needs of the millions of people affected by persistent AFib across Europe.
Dive Insight:
The persistent form of AFib covered in the European label expansion can cause dizziness, fatigue and shortness of breath, and increases stroke risk.
Expanded labeling for Farapulse, Boston Scientific’s signature PFA platform, to treat patients in Europe with persistent AFib follows a label expansion received in July from the Food and Drug Administration in the same patient population.
Boston Scientific, through its 2021 acquisition of privately held Farapulse, was the first of the big cardiac device makers to offer a PFA system in Europe.
The company won the CE mark for Farapulse in 2021 to treat paroxysmal, or intermittent, AFib. Boston Scientific then gained the FDA’s nod in January 2024 for treating those patients, accelerating a market share battle in the fast-growing PFA space with Medtronic and later Johnson & Johnson and Abbott.
PFA is considered a safer alternative for treating AFib than older approaches to cardiac tissue ablation because it can reduce the risk of damage to surrounding tissue and shorten procedure times.
On an earnings call last month, Boston Scientific CEO Mike Mahoney said the company expects to outpace growth in the electrophysiology market in 2026, led by its PFA portfolio amid continued adoption of the technology across the globe.
An estimated 59 million people worldwide have AFib, and many have the persistent form, according to Boston Scientific.