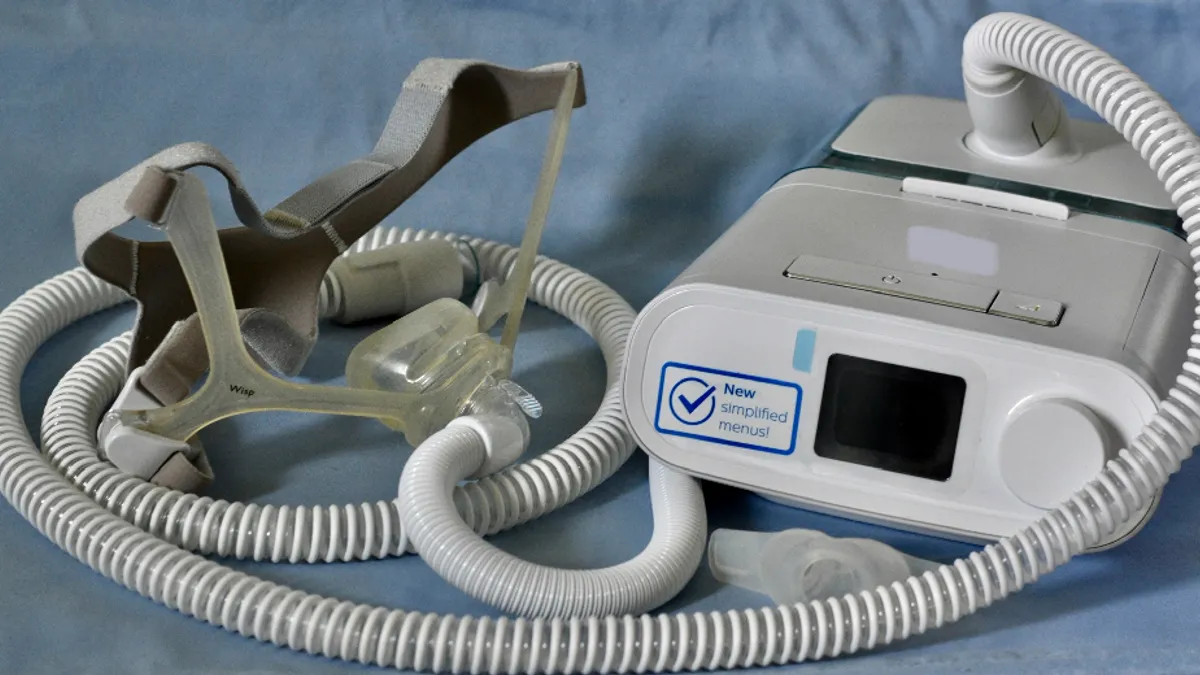
More reports of deaths and complaints related to Philips’ ongoing recall of its sleep apnea devices and ventilators have been received by the Food and Drug Administration, the regulator said.
Since April 2021, the agency has received 260 reports of deaths amid more than 90,000 medical device reports “reportedly associated” with the breakdown of soundproofing foam in the recalled devices. The most recent update, from August to October 2022, includes 21,000 reports and 91 deaths.
The FDA cautioned in a statement that these reports alone can’t determine the prevalence of an event and may be affected by under-reporting or lack of verification that the device caused the problem.
Philips began the recall in June 2021 due to potential health risks with PE-PUR foam used to soundproof the devices breaking down and being inhaled by patients. Some of the injuries reported to the FDA include cancer, pneumonia, asthma and other respiratory problems.
The company currently is repairing or replacing more than 5.5 million affected devices — a process that is expected to extend into 2023.
That could be compounded by recent problems with Philips’ Trilogy ventilators that had been reworked as part of the recall. In a Nov. 17 report, the FDA reported that the new silicone sound abatement foam installed in the ventilators can separate from the plastic backing and block the device’s air inlet. The company also detected trace amounts of particulate matter in some of the reworked devices, including PE-PUR foam particles.
Philips is in consent decree talks with the U.S. Department of Justice related to its handling of the recall, but neither party has revealed the terms of the agreement. In the meantime, patients continue to face long waits for CPAP machines.
In October, the company said 90% of the recall is expected to be completed by the end of the year.
In an emailed statement, Philips spokesperson Steve Klink wrote, "we are very sorry for the concern that recall has caused, and we are working very hard to repair or replace the affected devices."
In an update on Tuesday, the company said it had "relied on an initial, limited data set and toxicological risk assessment, and assumed a reasonable worst-case scenario for the possible health risks" at the time of the recall. After the company's public statements about the possible risks to users and the recall announcement in June, Philips said it received a steep increase in complaints related to possible foam degradation.
This article has been updated with a statement from Philips.