Cardiac
-
Boston Scientific posts positive data for coronary IVL catheter
With the study results, Boston Scientific plans a regulatory submission for the intravascular lithotripsy treatment for patients with severely calcified coronary artery disease.
By Susan Kelly • May 19, 2026 -
Artivion closes Endospan buyout to acquire recently approved heart device
Artivion is targeting a $150 million annual U.S. market for the Nexus Aortic Arch System.
By Nick Paul Taylor • May 19, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
Boston Scientific buys $1.5B stake in TAVR developer MiRus
The deal gives Boston Scientific a 34% equity stake in MiRus and an option to acquire its TAVR platform by making additional payments totaling $3 billion.
By Susan Kelly • May 18, 2026 -
J&J launches improved Shockwave catheter
The latest generation of the coronary intravascular lithotripsy device is designed to be more flexible to help physicians navigate tight arteries to break up calcified deposits and restore blood flow.
By Susan Kelly • May 13, 2026 -

 Retrieved from Medtronic on May 11, 2026
Retrieved from Medtronic on May 11, 2026
Medtronic to close California site amid restructuring
The decision to wind down the operation in Santa Rosa in about two years comes as the medtech giant combines several businesses within its cardiovascular portfolio.
By Susan Kelly • May 11, 2026 -
Abbott gets FDA nod, CE mark for AI-enabled imaging device
The device joins a portfolio of vascular technologies that generated 9.5% sales growth on a comparable basis in the first quarter.
By Nick Paul Taylor • May 4, 2026 -
Medtronic wins FDA approval for updated mitral replacement valve
Surgeons have performed initial implants of the Mosaic Neo valve at centers across the U.S., as well as the first robotic implantation.
By Susan Kelly • April 29, 2026 -
J&J starts head-to-head PFA trial against Boston Scientific device
The study will compare outcomes on J&J’s Varipulse Pro platform and Boston Scientific’s rival Farapulse PFA system.
By Nick Paul Taylor • April 28, 2026 -
4 studies to know from HRS 2026
Boston Scientific, Abbott and Medtronic shared new cardiac device data at the annual Heart Rhythm Society meeting, where market share shifts in pulsed field ablation were on analysts’ minds.
By Susan Kelly • April 27, 2026 -
J&J to acquire Atraverse Medical
The FDA-cleared radiofrequency guidewire will slot into J&J’s fast-growing cardiovascular portfolio.
By Nick Paul Taylor • April 27, 2026 -
Edwards raises 2026 forecast as TAVR sales surge
The stronger outlook follows mixed results from other major medical device makers in the latest quarter, after both Abbott and Boston Scientific cut their full-year guidance.
By Susan Kelly • April 24, 2026 -
Medtronic leads $100M investment in Pulnovo Medical
Medtronic formed a commercial agreement with Pulnovo alongside its investment.
By Nick Paul Taylor • April 21, 2026 -
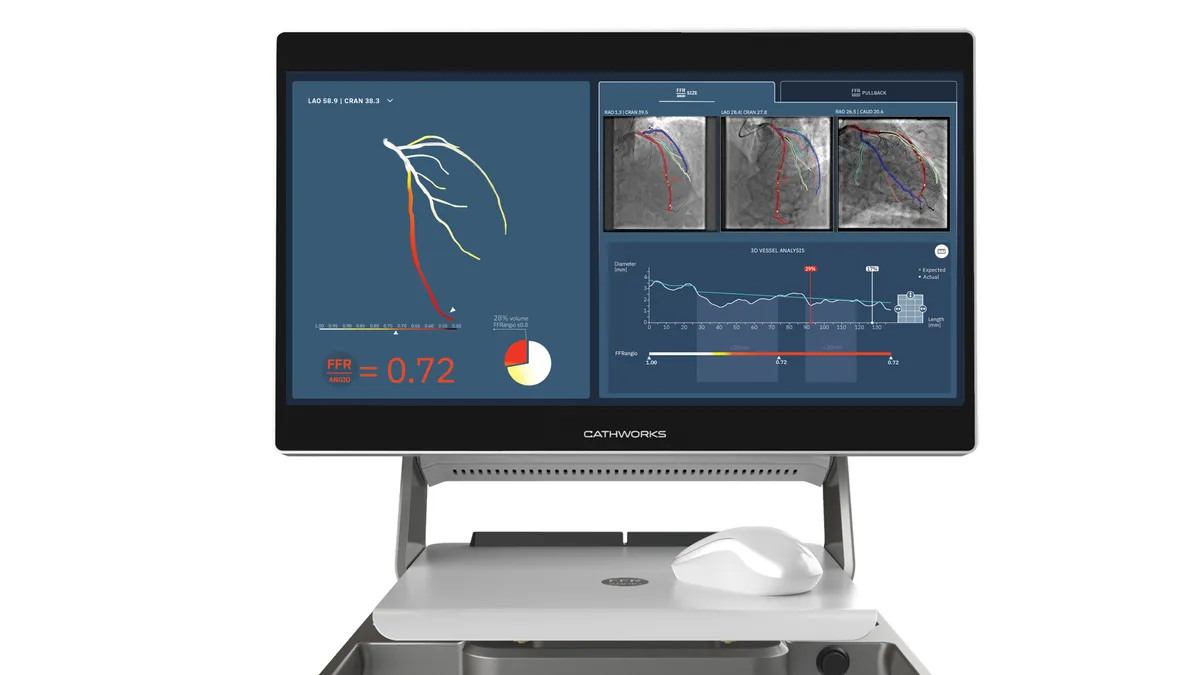
Medtronic closes $585M purchase of CathWorks
The deal is the second tuck-in acquisition this year for Medtronic, which has signaled its intent to pursue more M&A in its core business lines.
By Susan Kelly • April 20, 2026 -
Stryker to buy Amplitude Vascular Systems
Amplitude’s intravascular lithotripsy technology could emerge as a challenger to Shockwave IVL, Leerink Partners analyst Mike Kratky wrote.
By Elise Reuter • April 13, 2026 -
Pulse Biosciences hires Liane Teplitsky as COO
The appointment gives a former Abbott leader oversight of efforts to bring a pulsed field ablation device to market.
By Nick Paul Taylor • April 10, 2026 -
J&J launches enhanced PFA device in Europe
The Varipulse Pro pulsed field ablation platform, which has received a CE mark, will be a focus at the upcoming European Heart Rhythm Association annual meeting in Paris.
By Susan Kelly • April 7, 2026 -
J&J’s Impella heart pump shows no patient benefit in 2 trials
Study investigators cautioned against routine use of the device in complex percutaneous coronary interventions and to treat heart attack patients who don’t have cardiogenic shock.
By Susan Kelly • April 1, 2026 -
Medtronic wins CE mark for OmniaSecure defibrillation lead
The device maker designed the lead to have the safety benefits of small-diameter devices without sacrificing durability.
By Nick Paul Taylor • March 31, 2026 -
Medtronic to study renal denervation combined with PCI
Medtronic detailed plans to further study the blood pressure treatment while releasing additional data from the SPYRAL HTN trials that showed the procedure reduced hypertensive emergencies.
By Susan Kelly • March 31, 2026 -
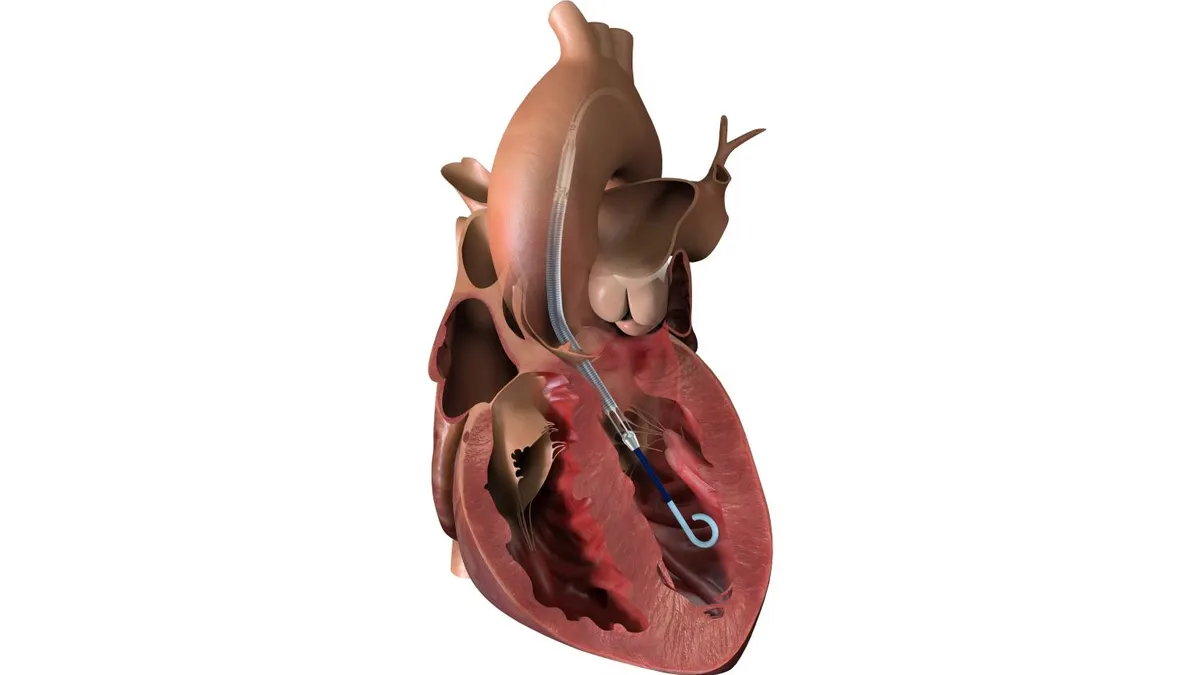
Boston Scientific posts positive stroke prevention data for Watchman
The company said it will seek an expanded indication for the left atrial appendage closure device after it met all safety and efficacy goals in the closely watched CHAMPION-AF study.
By Susan Kelly • March 30, 2026 -
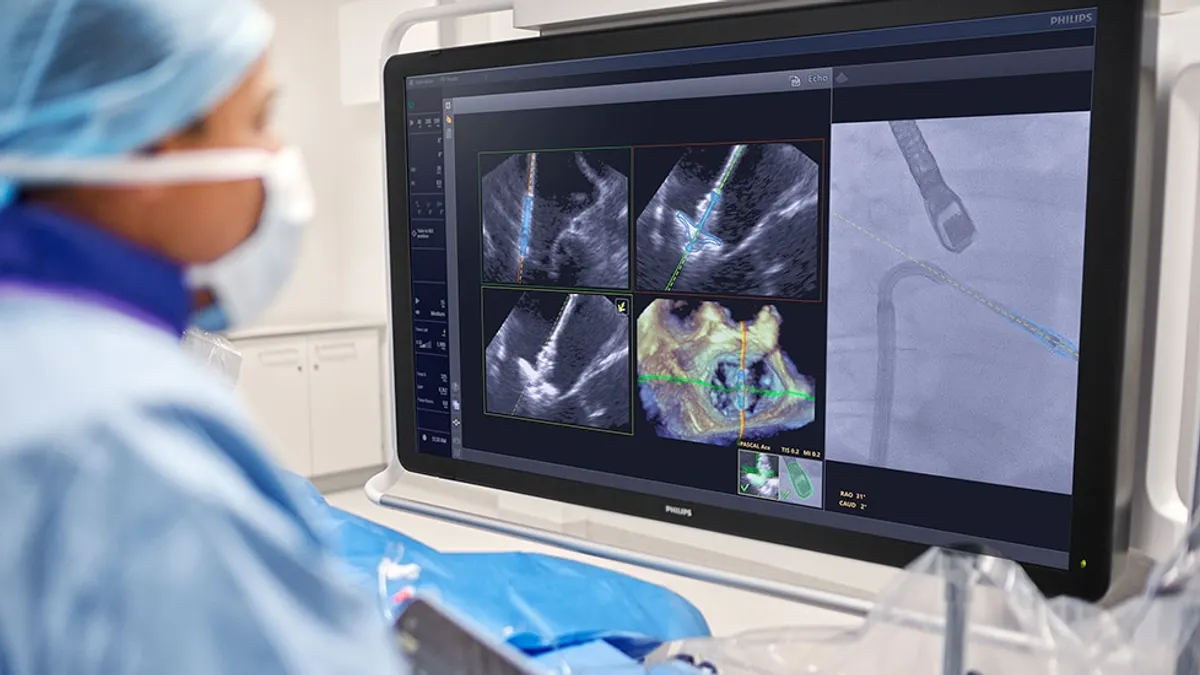
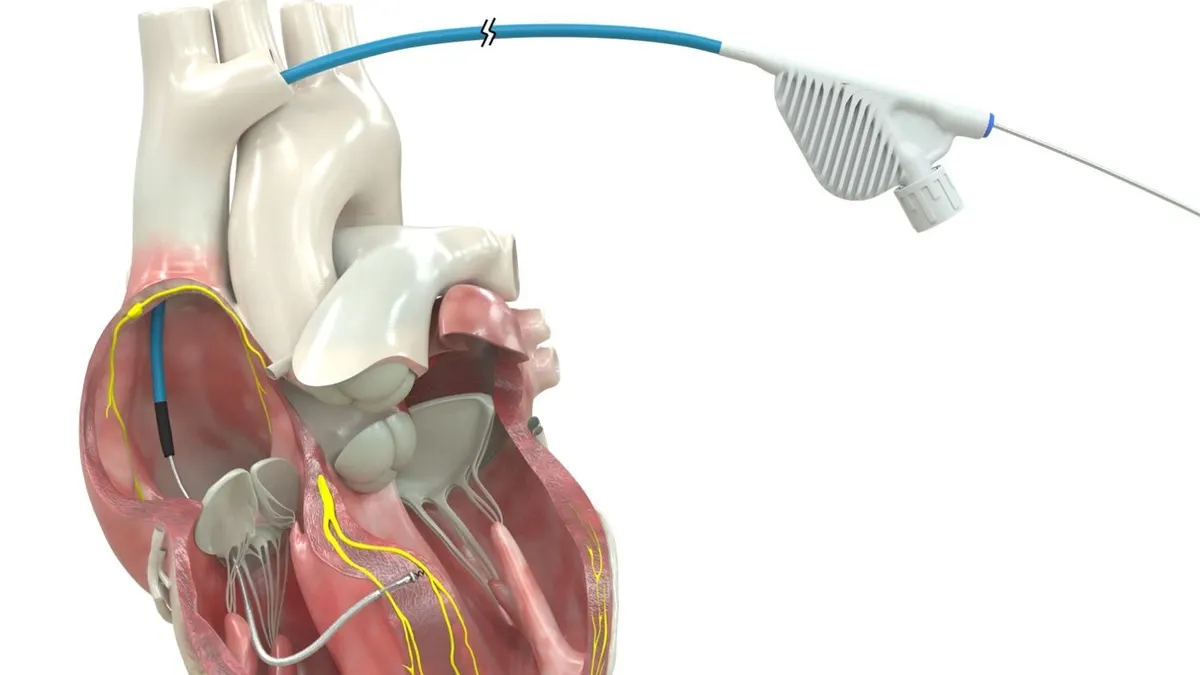
Philips wins FDA clearance for imaging device developed with Edwards
The system uses AI to optimize the placement of Edwards’ mitral valve repair device.
By Nick Paul Taylor • March 27, 2026 -
Endologix to close production facility, lay off 31 people
The company acquired the facility in the PQ Bypass takeover it struck in the aftermath of its bankruptcy filing.
By Nick Paul Taylor • March 26, 2026 -
Medtronic’s OmniaSecure defibrillation lead earns expanded FDA indication
The OmniaSecure lead gained the first U.S. approval for placement in the heart’s left bundle branch area, to help activate the natural electrical system.
By Susan Kelly • March 25, 2026 -
Thermo Fisher completes $8.9B buyout of Clario
Thermo Fisher plans to create a fast-growing business by integrating Clario’s data capture and management system with its existing capabilities.
By Nick Paul Taylor • March 25, 2026 -
JenaValve snags FDA approval in aortic regurgitation
The approval comes two months after Edwards Lifesciences scuttled plans to buy JenaValve for $945 million amid opposition from the Federal Trade Commission.
By Susan Kelly • March 18, 2026