Medical Devices: Page 127
-
Healthcare funding shatters records in 2020, helped by COVID-19
Medical device startups raised roughly $6 billion in the fourth quarter, a high point over the last three years, according to a CB Insights report.
By Rebecca Pifer Parduhn • Jan. 22, 2021 -
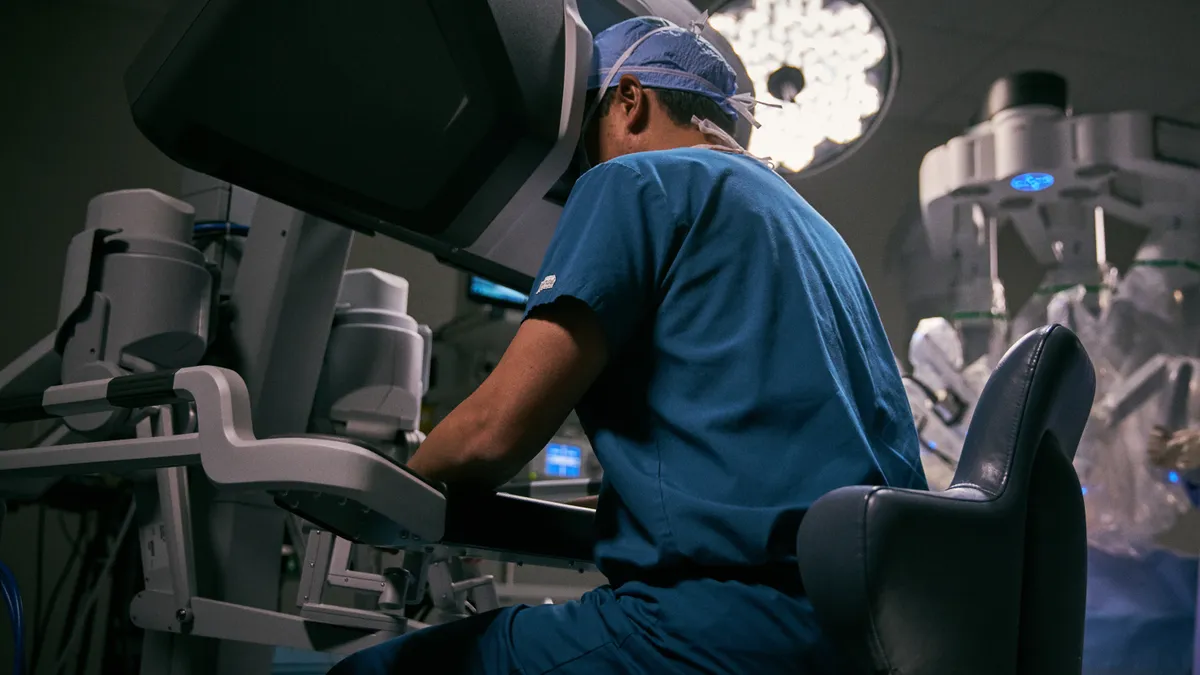
Intuitive braces for bumpy 2021 as COVID-19 resurgence hits robotics
The company, which gave details on its fourth quarter, is not giving guidance for the year amid uncertainty. Shares fell nearly 5% Friday morning.
By Nick Paul Taylor • Jan. 22, 2021 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
4 key trends for payers and providers in 2021
The COVID-19 crisis may spur speedier adoption of value-based care and partnerships between health systems and payers in the year ahead.
By Samantha Liss • Jan. 22, 2021 -
Boston Scientific bets on cardiac wearables with $925M Preventice buy
The deal is the latest in the fast-growing space, coming on the heels of Hillrom's $375 million BardyDx buy on Tuesday and Philips' $2.8 billion purchase of BioTelemetry last month.
By Ricky Zipp • Jan. 21, 2021 -
Haemonetics inks $510M Cardiva takeover to expand hospital business
The blood products medtech contends the deal will strengthen its portfolio in interventional cardiology and electrophysiology markets.
By Nick Paul Taylor • Jan. 21, 2021 -
FDA breakthrough nods go to Alzheimer's devices, cardiovascular products
Cognito and Boston Scientific-backed Functional Neuromodulation picked up agency designations, positioning them for speedy reviews as they work to get their Alzheimer offerings to market.
By Nick Paul Taylor • Jan. 20, 2021 -
Hillrom makes cardiac monitoring play with $375M BardyDx buy
The deal fits Hillrom's existing heart portfolio and efforts to connect devices to the internet, analysts said, but one raised concerns about Medicare reimbursement.
By Susan Kelly • Jan. 20, 2021 -
CMS expands transcatheter mitral coverage, boosting Abbott's MitraClip device
Wall Street analysts said the national coverage determination could triple the patient base eligible for Abbott's MitraClip device, adding fuel to a growing and under-penetrated market.
By Ricky Zipp • Jan. 20, 2021 -
Forging trust in AI, sustaining the virtual care boom and other CES takeaways
"You can't just set a piece of software in front of somebody and say 'trust me,'" Christina Silcox, digital health fellow at the Duke-Margolis Center for Health Policy, said at the consumer tech conference.
By Rebecca Pifer Parduhn • Jan. 19, 2021 -
Philips to acquire Capsule Technologies in $635M deal, growing patient data capabilities
The move follows the medtech giant's $2.8 billion acquisition of cardiac monitoring company BioTelemetry in December, one of 2020's largest deals.
By Ricky Zipp • Jan. 19, 2021 -
Neuromod portfolios advance at Medtronic, Boston Scientific, Abbott
The flurry of activity is further evidence of the medtech industry’s latest attempts to make progress in a sector criticized as stagnant just one year ago.
By Nick Paul Taylor • Jan. 15, 2021 -
Google calls Fitbit acquisition complete despite ongoing DOJ review
The search giant closed the deal because the department's 14-month investigation period expired without any objection. However, DOJ could still sue to unwind the buy.
By Rebecca Pifer Parduhn • Jan. 14, 2021 -
Intuitive, J&J, Medtronic and Zimmer talk robotic surgery ambitions: JPM21
Medtech majors used this week's virtual J.P. Morgan healthcare conference to talk up their plans in the hot, increasingly competitive sector.
By Nick Paul Taylor • Jan. 14, 2021 -
Remote monitoring, wearable companies look to capitalize on virtual care boom: JPM21
Diabetes players Dexcom and Insulet both projected business growth and market expansion for this year during presentations.
By Ricky Zipp • Jan. 14, 2021 -
Boston Scientific Q4 misses Wall Street, a laggard in early medtech previews: JPM21
The fourth quarter miss points to "further softness" in the first quarter as COVID-19 cases surge, J.P. Morgan analysts said.
By Greg Slabodkin • Jan. 13, 2021 -
FDA issues action plan for regulating AI in medical devices
The proposal brings the agency a step closer to draft guidance for regulating the fast-evolving field of artificial intelligence and machine learning-based software in medical devices.
By Susan Kelly • Jan. 13, 2021 -
Steris inks $4.6B buy of Cantel Medical amid flurry of M&A
It's the biggest deal so far this year. Wall Street analysts said COVID-19 infection concerns should boost Cantel in the long term.
By Ricky Zipp • Jan. 13, 2021 -
CMS breakthrough rule called tailwind for Abbott, J&J and Medtronic
The policy gives developers of FDA-designated breakthrough products coverage for Medicare's 60 million beneficiaries on the day of approval.
By Nick Paul Taylor • Jan. 13, 2021 -
3 takeaways from JP Morgan: Electives, M&A and looking past COVID-19
Execs from Medtronic, Baxter and BD are among those previewing the year ahead at the bank's annual healthcare conference.
By Greg Slabodkin , Ricky Zipp • Jan. 12, 2021 -
In medtech win, MDR-IVDR remote audits OK'd amid pandemic pressures
An official for MedTech Europe, which pushed for flexibility ahead of new medical device rules set to kick in later this year, called the move "significant and positive."
By Nick Paul Taylor • Jan. 12, 2021 -

 "State Capitol Building, USA, California, Sacramento" [Photograph]. Retrieved from Pixy.
"State Capitol Building, USA, California, Sacramento" [Photograph]. Retrieved from Pixy.
California proposes expanding Medicaid coverage of continuous glucose monitors
The budget proposal seeks to increase access to CGMs among adults with Type 1 diabetes covered by the state's Medicaid program Medi-Cal. Abbott, Dexcom, Medtronic and Senseonics compete for the U.S. CGM market.
By Nick Paul Taylor • Jan. 11, 2021 -
Medtech M&A to take off after pandemic slowed deals in 2020: EY
Dealmaking in 2021 will be fueled by financial firepower across the industry at an "all-time high" of nearly $500 billion, according to the consultants.
By Ricky Zipp • Jan. 10, 2021 -
New Mexico sues device sterilizer Sterigenics over ethylene oxide emissions
The suit alleges ethylene oxide levels were often elevated due to "lax oversight" practices such as leaving sterilization chamber doors ajar.
By Nick Paul Taylor • Updated Jan. 7, 2021 -
Wall Street paints rosy 2021 for medtech as electives snap back
After economic challenges throughout 2020, sell-side analysts predict procedure-dependent companies like Medtronic, Abbott and Zimmer Biomet to outperform this year as procedure volumes return.
By Ricky Zipp • Jan. 7, 2021 -
French medtechs Carmat, CorWave advance heart failure device plans
Carmat is gearing up to start selling its artificial heart, while CorWave disclosed a $40 million financing round for its twist on the LVADs sold by companies including Abbott and Medtronic.
By Nick Paul Taylor • Jan. 7, 2021