Medical Devices: Page 91
-
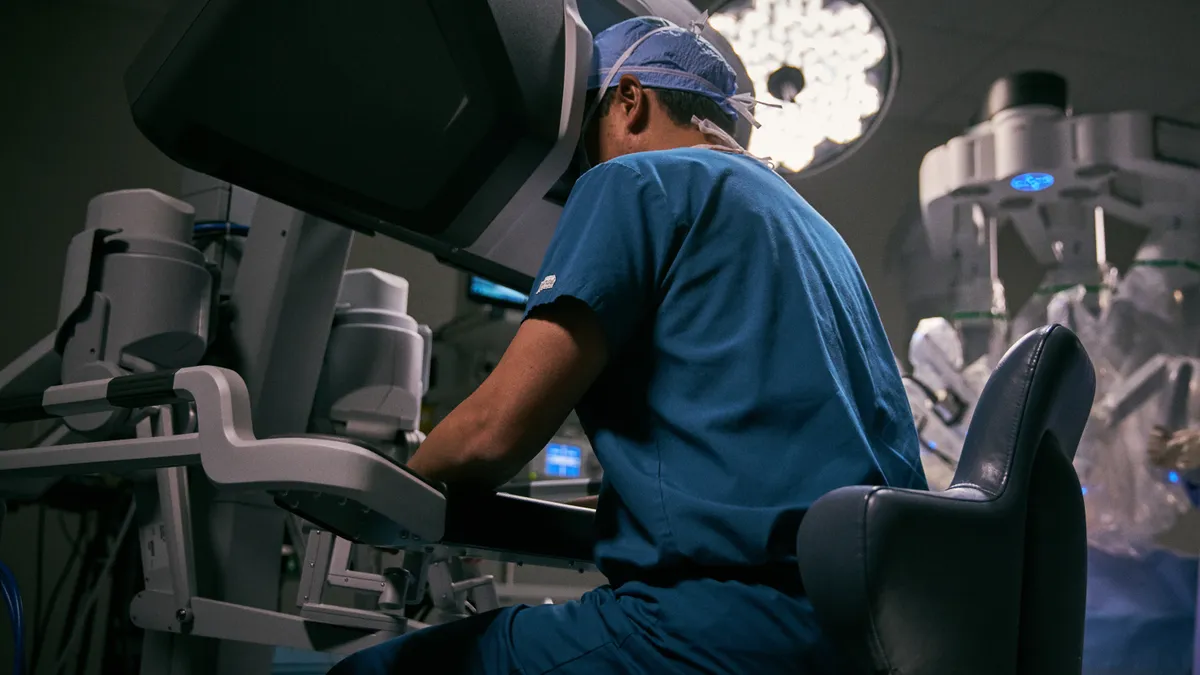
Intuitive’s robotic surgery volumes recovered in Q3; shares climb
As hospital staffing increased and COVID-19 wariness eased, the number of procedures performed with the company’s hallmark da Vinci system rose 20% in the third quarter from a year earlier.
By Peter Green • Oct. 18, 2022 -
J&J touts rising procedure volumes, lowers full-year forecast on foreign-exchange concerns
Analysts said the results were an “encouraging start” to medtech earnings for the third quarter.
By Elise Reuter • Oct. 18, 2022 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
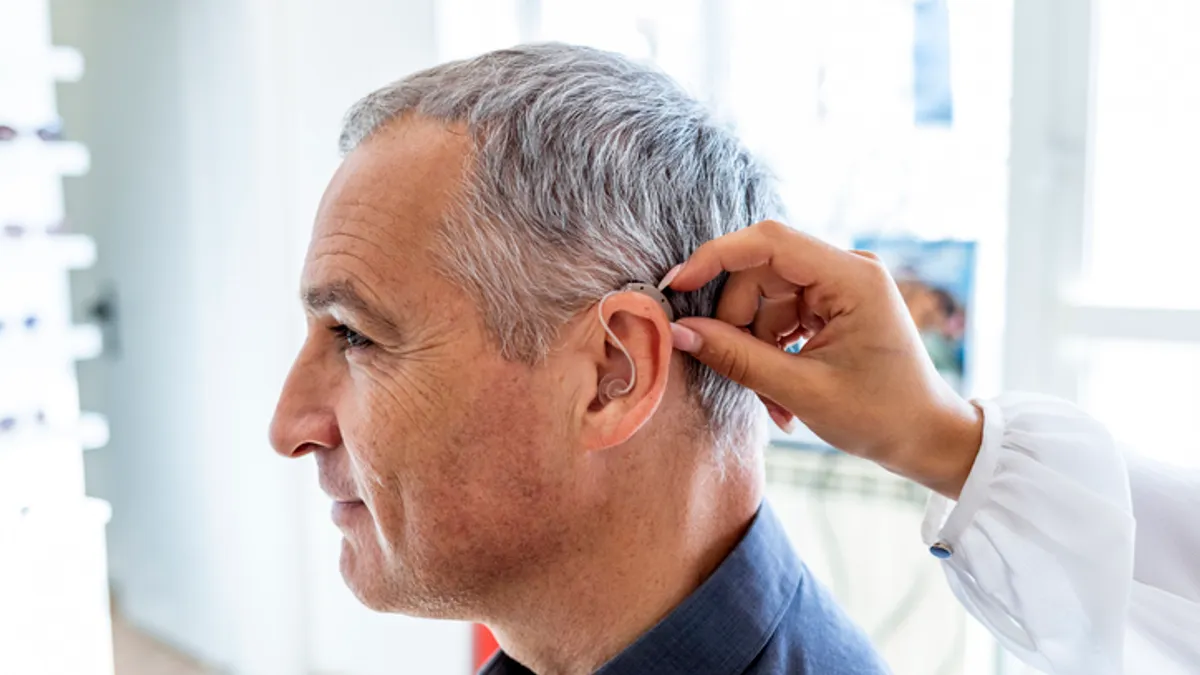
Lucid enters OTC hearing aid market, undercutting rivals with devices starting at $200
Lucid joins Sony and the Bose-partnered Lexie Hearing with aids priced thousands of dollars below prescription devices, putting auditory assistance within reach of tens of millions of Americans.
By Nick Paul Taylor • Oct. 18, 2022 -
Coloplast told by FDA to keep transvaginal mesh off market after 36-month surveillance study
The mesh provides similar effectiveness and safety outcomes to native tissue repair but also comes with additional risks, regulators concluded.
By Nick Paul Taylor • Oct. 18, 2022 -
Medtronic, in a first, secures expanded label for cardiac pacing lead
The approval is part of Medtronic’s push to expand its share of the market for cardiac conduction system pacing.
By Nick Paul Taylor • Oct. 18, 2022 -
Insulet flags battery problem with Omnipod DASH system
The company is expected to spend $35 million to $45 million replacing all of its personal diabetes manager devices after reports that batteries in the handheld controller may be swelling, leaking fluid or overheating.
By Elise Reuter • Oct. 17, 2022 -

 Courtesy of https://news.medtronic.com/Left-Ventricular-Assist-Device-For-Advanced-Heart-Failure#assets_34137_10-122:19299
Courtesy of https://news.medtronic.com/Left-Ventricular-Assist-Device-For-Advanced-Heart-Failure#assets_34137_10-122:19299
Medtronic’s faulty HVAD controllers get an emergency-only software fix
The new controllers will be available to all healthcare providers with a patient on HVAD support, but the company cautioned that the software should only be used after previous attempts to restart the pump have failed.
By Elise Reuter • Oct. 17, 2022 -
Johnson & Johnson, Alcon agree on $75M settlement to resolve contact-lens antitrust claims
A class-action suit accused the manufacturers of violating federal antitrust law by setting the same minimum price for their lenses.
By Nick Paul Taylor • Oct. 17, 2022 -
Sponsored by Esper
Why Android is the future of connected HealthTech devices
How Android can make your connected HealthTech device strategy a success.
Oct. 17, 2022 -
 Retrieved from Abbott/PRNewswire on June 15, 2020
Retrieved from Abbott/PRNewswire on June 15, 2020
Abbott, J&J and Intuitive to lead off medtech earnings season; procedures, supply chain in spotlight
Results from the three companies are expected to shed light on how the economic slowdown is affecting procedure trends in the industry.
By Nick Paul Taylor • Oct. 17, 2022 -
Hospital staff shortages spurred drop in TAVR procedures in Q3, surgeon survey shows
Staffing at hospitals is expected to remain disrupted until at least 2024, reducing the number of surgeries, more than 40% of respondents said.
By Nick Paul Taylor • Oct. 14, 2022 -
Bose-partnered Lexie to launch $999 OTC hearing aid, challenging Sony, Eargo in nascent market
Lexie has matched the price of the first Sony device and is seeking to differentiate itself through the use of a rechargeable battery.
By Nick Paul Taylor • Oct. 14, 2022 -
Sony begins sale of OTC hearing aids with entry-level price of $999
Lower-cost, over-the-counter hearing aids are becoming available to the 70% of older Americans who need the devices but don’t have them.
By Peter Green • Oct. 13, 2022 -
GE advances healthcare spinoff plan, providing close look at workings of the unit
Localization requirements, third-party servicers, and more sophisticated data security threats are cited as risks to the healthcare unit.
By Nick Paul Taylor • Oct. 13, 2022 -
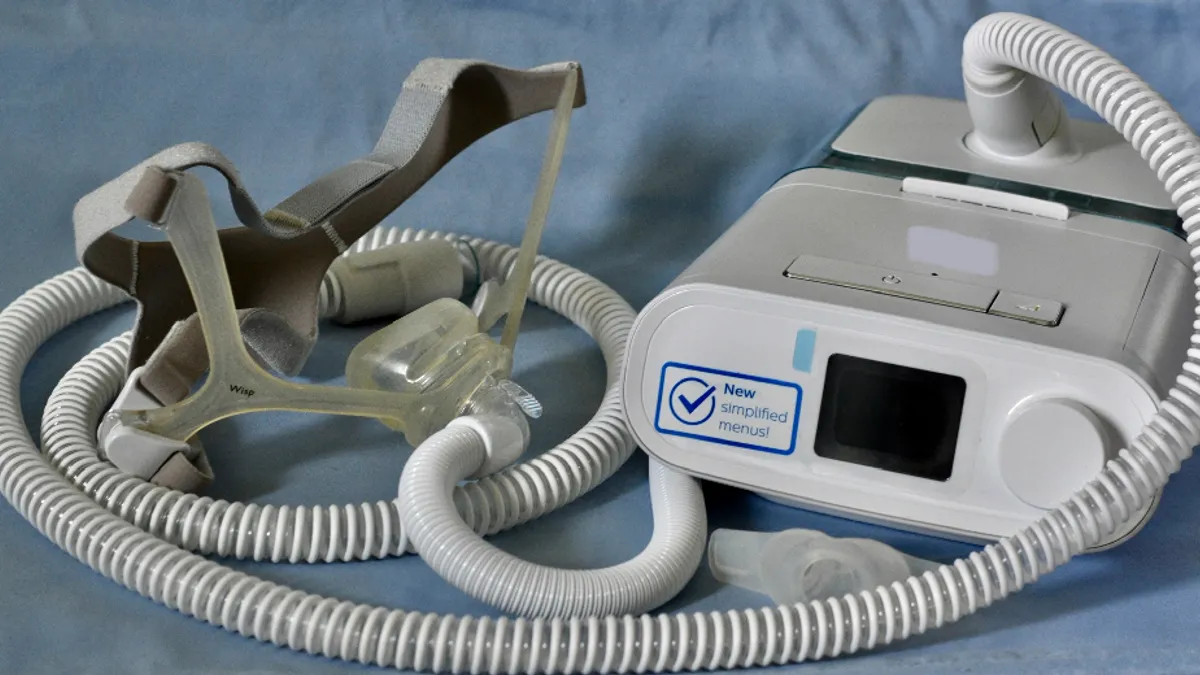
Philips says Q3 revenue fell, records $1.26B charge for Respironics; shares plunge
The Dutch-based medtech company remains dogged by sleep apnea machine recalls and supply chain issues.
By Peter Green • Oct. 12, 2022 -
FDA starts advisory program pilot to reduce ‘valley of death’ risk for medical devices
A plan to ensure more medical devices pass from testing to clinical application aims to help dozens of devices through the approval process each year.
By Nick Paul Taylor • Oct. 12, 2022 -
‘Do-it-yourself’ artificial pancreas system beats production-line pump in controlled trial
Type 1 diabetes patients who used a customizable, open-source artificial pancreas, an insulin pump and a Dexcom G6 CGM found it more effective at controlling blood glucose than conventional sensor-augmented insulin pumps.
By Nick Paul Taylor • Oct. 12, 2022 -
FDA finalizes postapproval study guidance in light of AdvaMed, Foundation Medicine feedback
The agency resisted calls to give sponsors more time to prepare protocols for post-approval studies.
By Nick Paul Taylor • Oct. 11, 2022 -

 Retrieved from Abbott/PRNewswire on June 15, 2020
Retrieved from Abbott/PRNewswire on June 15, 2020
Abbott’s FreeStyle Libre 2 CGM beats fingerstick testing in independent clinical trial
Going into the study, the researchers were unsure if CGMs with optional alarms for high and low blood glucose levels benefit patients with Type 1 diabetes.
By Nick Paul Taylor • Oct. 11, 2022 -
NovaSight’s digital treatment for lazy eye gets FDA nod, providing alternative to patching
A randomized controlled trial found the digital device to be as effective as wearing an eye patch, the current gold standard for treatment, opening the way for more children to complete necessary therapy for amblyopia.
By Nick Paul Taylor • Oct. 10, 2022 -
Owlet seeks clearance of blood-oxygen-measuring baby sock after FDA warning letter
Owlet now is a step closer to selling a prescription medical device designed to alert parents when their baby’s heart rate or oxygen saturation levels move outside of prescribed ranges.
By Nick Paul Taylor • Oct. 10, 2022 -
‘Inadequate’ progress of Bayer’s study on birth-control implant called out by FDA as patients drop out
The FDA saw a rise in patients dropping out of a study on the Essure birth-control implant, prompting it to tell Bayer to develop and implement strategies to ensure the work continues.
By Nick Paul Taylor • Oct. 7, 2022 -

 Retrieved from Abbott/PRNewswire on June 15, 2020
Retrieved from Abbott/PRNewswire on June 15, 2020
Dexcom, Abbott have ‘massive opportunity’ with new CGM coverage proposal: analysts
Analysts at J.P. Morgan said the proposal reads “very favorably” for the two leading makers of continuous glucose monitors and unlocks “a major near-term driver for growth.”
By Nick Paul Taylor • Oct. 7, 2022 -
BD discloses cybersecurity vulnerability in cervical cytology processing machine
The company is advising users to address the threat by restricting access to the instrument while it works on a software patch.
By Nick Paul Taylor • Oct. 6, 2022 -
Dexcom starts global rollout of G7 CGM system, launching device in U.K. and Germany
Software updates have delayed the introduction of the product in the U.S. at a time when the company seeks to take market share from rival Abbott.
By Nick Paul Taylor • Oct. 6, 2022