Medical Devices: Page 98
-
Hologic's stock rating cut by BTIG amid worsening supply chain challenges, drop in COVID testing
The company is facing challenges including increasing operating expenses and shortages of semiconductor chips, according to the analyst report.
By Ricky Zipp • July 18, 2022 -
Notified bodies have yet to issue MDR certificates for 85% of legacy devices: survey
The EU's Medical Device Regulation certification process is taking twice as long as the old directive pathway and smaller companies are struggling to get started, the poll shows.
By Nick Paul Taylor • July 18, 2022 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
FDA faces furloughs if Senate misses August deadline for user fee bill
If legislation isn’t passed before the August recess, the FDA would have to furlough workers, slowing its review of medical device and pharmaceutical products.
By Elise Reuter • July 15, 2022 -
Medtronic warns European healthcare providers of safety problem in defibrillators
The warning adds to Medtronic’s growing list of product safety issues over the last 18 months.
By Ricky Zipp • July 15, 2022 -
Abbott, Intuitive, J&J to kick off quarterly earnings season next week as recession threat looms
Earnings results are poised to set expectations for the impact of a potential slowdown and shed light on trends with implications for companies including Boston Scientific, Medtronic and Stryker.
By Nick Paul Taylor • July 15, 2022 -
With medtech shares down 30%, investors worry about inflation, staffing shortages: William Blair
Data suggests earlier pessimism about the sector could drive stocks to outperform major indices later this year.
By Elise Reuter • July 14, 2022 -
Rising procedure volumes boost hopes for Abbott, J&J as orthopedics lag, survey finds
Stifel analysts see the findings as positive for Abbott, J&J and Intuitive and supportive of Edwards and Medtronic’s TAVR targets. The survey comes just before the medtech industry reports its latest earnings.
By Nick Paul Taylor • July 14, 2022 -
Abbott gets FDA breakthrough tag for deep brain stimulation system in depression
The breakthrough device designation builds on a series of clinical trials of other systems that have yielded mixed results.
By Nick Paul Taylor • July 13, 2022 -
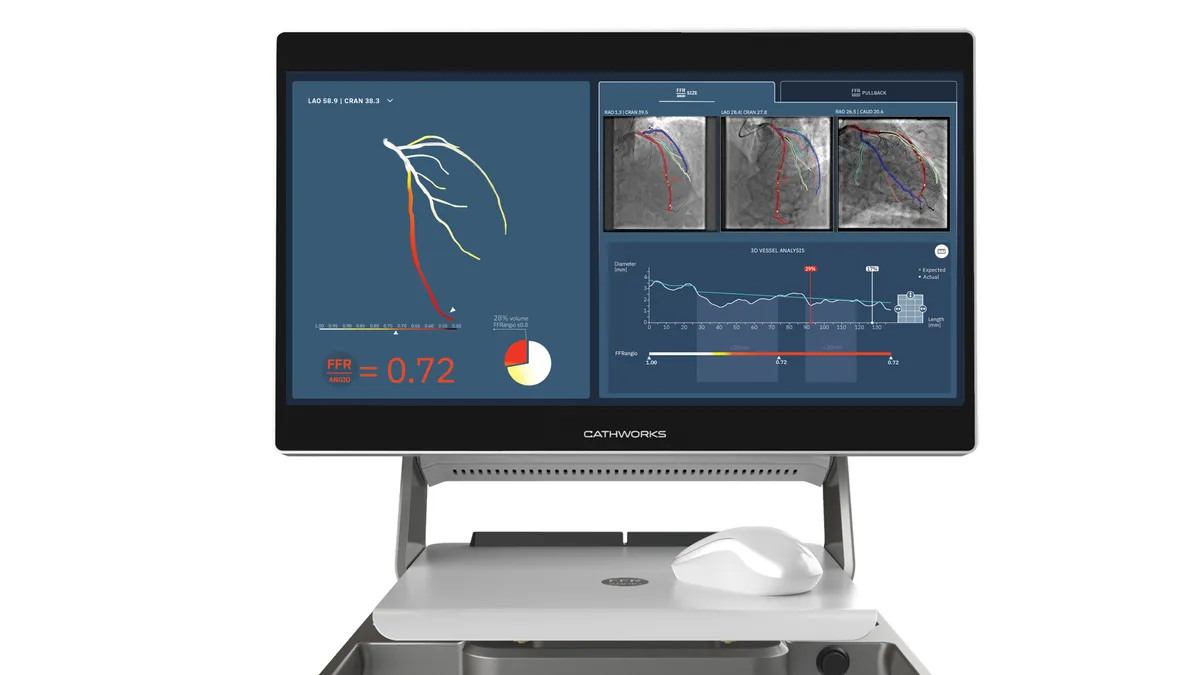
Medtronic lands option to buy artery disease player CathWorks for up to $585M
CathWorks has the right to compel Medtronic to complete the acquisition if it chooses not to exercise its option.
By Nick Paul Taylor • July 13, 2022 -
Faulty oximeter readings led to racial, ethnic disparities in treatment: study
Researchers previously noted that the devices may be less accurate when used by people with darker skin, and a recent study found the difference led to patients failing to receive needed treatment.
By Elise Reuter • July 12, 2022 -
CMS makes 'market-moving proposals' to some medtech Medicare rates, say analysts
BTIG analysts warned in a recent report that the changes “might curtail usage of some higher-dollar” medical devices.
By Nick Paul Taylor • July 12, 2022 -
European Commission targets spring of 2024 for fully functional Eudamed database
The Commission is planning a two-phase transition to mandatory use of the database after it goes live.
By Nick Paul Taylor • July 11, 2022 -
Medtronic recalls over 1M dialysis catheters due to malfunction
The recall is the latest in a growing list of product safety issues for Medtronic, including Class I recalls and an FDA warning letter for its diabetes unit.
By Ricky Zipp • July 11, 2022 -
Ex-Mazor executive charged by SEC with insider trading over Medtronic's $1.6B buyout
Doron Tavlin, who was Mazor’s vice president of business development, is accused by the agency of profiting from his inside knowledge of the pending takeover.
By Nick Paul Taylor • July 11, 2022 -
Q&A
Friday Q&A: Roy Jakobs, Philips' head of connected care, talks sleep apnea recall, changing safety protocols
In the midst of a recall of 5.5 million sleep apnea devices and ventilators, Jakobs spoke about the company's recall process, communication efforts, and how the repair and replacement program is progressing.
By Ricky Zipp • July 8, 2022 -
CMS proposes national rates for cardiac monitoring; iRhythm's stock jumps
The proposed rule could end a years-long saga for the cardiac market, although pricing specifics may change in the final version.
By Ricky Zipp • July 8, 2022 -
Semiconductor shortage leaves medtech industry 'more pessimistic' as customers leave, says Deloitte
Hospitals and health systems are looking into alternative products as a result of the disruption, according to a report from Deloitte and AdvaMed.
By Nick Paul Taylor • July 7, 2022 -
Getinge recalls anesthesia machines due to cracked, broken suction power switches
The FDA labeled the recall a Class I event, marking Getinge’s fifth Class I recall since September.
By Ricky Zipp • July 7, 2022 -
Intuitive's ecosystem is making it harder for J&J, Medtronic to crack robotic surgery market, say analysts
Intuitive’s competitors will need an “ecosystem approach” that goes beyond hardware to make it easier for customers to switch, say analysts from BTIG.
By Nick Paul Taylor • July 7, 2022 -
Wearables are a growing part of doctor-patient talks for cardiac patients: study
As mentions of wearables in primary care grew, Apple increasingly replaced Fitbit as the most-discussed wearable brand in talks between physicians and patients in the study, which covered the years 2005 to 2019.
By Nick Paul Taylor • July 6, 2022 -
Senators press FDA to finalize OTC hearing aids, accuse industry of 'astroturf' campaigns
The two cross-aisle politicians said they want to “expand access, reduce costs, and ensure a robust new market for safe and effective OTC hearing aids” and accused the device manufacturing lobby of “harming American consumers.”
By Elise Reuter • July 6, 2022 -
'Little sign' that digital tools are being deployed effectively in fight against COVID outbreak, study finds
To help reduce the spread of the next pandemic, there is “a substantial practical advantage” to integrating the features of multiple smartphone apps, a Scripps Research Translational Institute team found.
By Nick Paul Taylor • July 6, 2022 -
American Contract Systems' COVID-19 test recall gets Class I label from FDA
Off-site assembly by workers who may not have been properly trained prompted the company to recall COVID-19 tests amid concerns they may yield false results.
By Nick Paul Taylor • Updated July 6, 2022 -
AliveCor ECG patent ruling sets stage for block on Apple Watch imports
Apple Watch imports to the U.S. could be barred if ruling by International Trade Court judge is finalized; Judge says Apple Watch infringes two cardiogram patents.
By Nick Paul Taylor • July 5, 2022 -
Q&A
Friday Q&A: Canary Medical CEO Bill Hunter discusses knee-implant sensors, device reimbursement
Canary’s founder discusses how the firm plans to create predictive tools from data collected by the devices, and long-term plans to embed its sensors in other implants.
By Elise Reuter • July 1, 2022