FDA
-
Medtronic seeks clearance for Hugo surgical robot in more indications
Securing 510(k) authorization would position Medtronic to compete with Intuitive in major robotic surgery markets.
By Nick Paul Taylor • June 4, 2026 -
J&J recalls Impella heart pumps after patient dies
Johnson & Johnson asked customers to quarantine and return affected products. A spokesperson said J&J conducted quality system audits as part of its integration of Abiomed that resulted in some field actions.
By Nick Paul Taylor • May 29, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
J&J corrects Impella controller over issue linked to 2 injuries, 1 death
The controller can stop for about 35 seconds, during which time the patient is unsupported by the heart pump.
By Nick Paul Taylor • May 26, 2026 -
Guardant wins FDA approval for updated liquid biopsy
The company could secure a higher price for the new test than for its older, narrower product, Wall Street analysts said.
By Nick Paul Taylor • May 21, 2026 -
Natera wins FDA approval for companion diagnostic
The test will enable physicians to assess if post-surgery cancer patients need to receive treatment to minimize the risk of recurrence.
By Nick Paul Taylor • May 18, 2026 -
Artera gets FDA clearance for AI breast cancer risk prediction tool
The company launched in 2023 with $90 million in funding from organizations including Johnson & Johnson’s venture capital arm.
By Nick Paul Taylor • May 12, 2026 -
FDA chief Marty Makary resigns from agency, ending tumultuous tenure
Makary’s exit ends a turbulent run marred by leadership upheaval, mass layoffs, political pressure and public spats with drugmakers.
By Ben Fidler • May 12, 2026 -
Bayesian Health gets FDA nod for AI sepsis detection tool
Created by Johns Hopkins University researchers, the early warning system for sepsis aims to help physicians detect the serious condition earlier.
By Elise Reuter • May 12, 2026 -
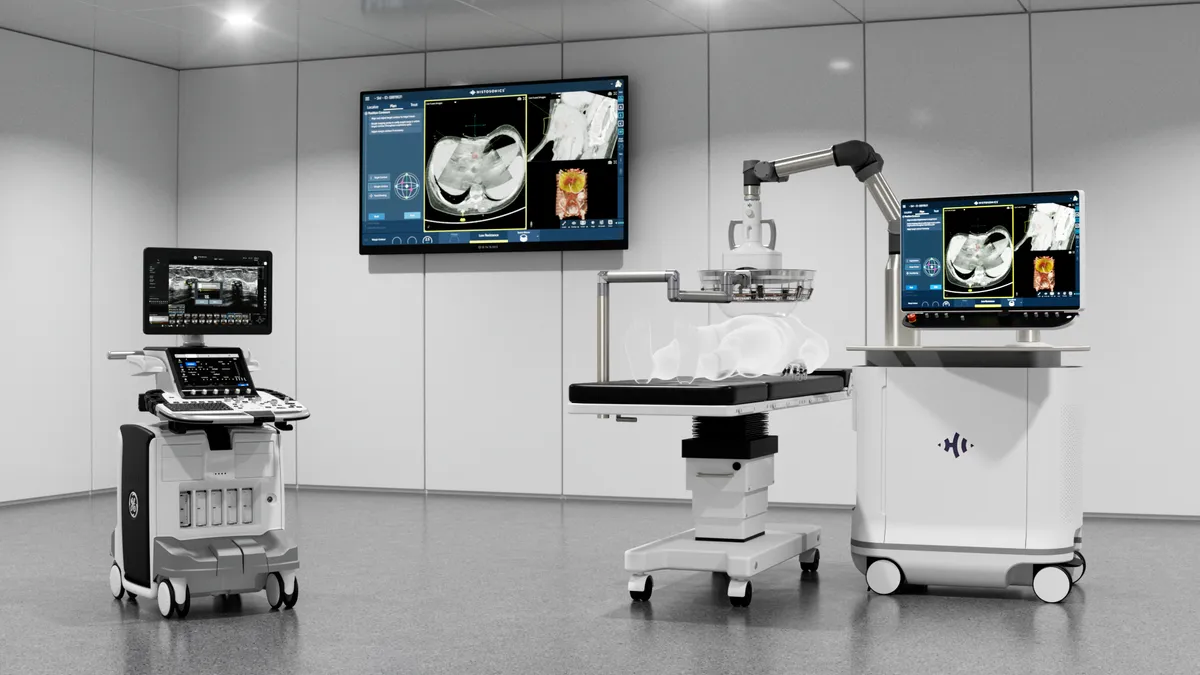
HistoSonics seeks FDA de novo authorization in kidney cancer
Investors including Bezos Expeditions and Thiel Bio supported HistoSonics’ expansion plans last year by participating in a $250 million financing round.
By Nick Paul Taylor • May 12, 2026 -
FDA warns of neurosurgical supply disruptions
Interruptions in the supply of neurosurgical patties, sponges and strips are expected to continue through 2026.
By Elise Reuter • May 6, 2026 -
Abbott gets FDA nod, CE mark for AI-enabled imaging device
The device joins a portfolio of vascular technologies that generated 9.5% sales growth on a comparable basis in the first quarter.
By Nick Paul Taylor • May 4, 2026 -
FDA advises users of Trividia glucometer to switch to alternatives
The potential for error code confusion to cause delayed or improper treatment led the FDA to recommend users transition to other blood glucose meters.
By Nick Paul Taylor • April 30, 2026 -
4 studies to know from HRS 2026
Boston Scientific, Abbott and Medtronic shared new cardiac device data at the annual Heart Rhythm Society meeting, where market share shifts in pulsed field ablation were on analysts’ minds.
By Susan Kelly • April 27, 2026 -
CMS, FDA unveil speedier Medicare coverage pathway for breakthrough devices
The pathway is designed to reduce the delay between FDA authorization and Medicare coverage for certain Class II and Class III breakthrough medical devices.
By Ricky Zipp • April 23, 2026 -
CDRH Director Tarver previews AI guidance at AAMI event
Michelle Tarver said the device center plans to finalize guidance on AI lifecycle management and provide some initial thoughts on generative AI following an advisory committee meeting last year.
By Elise Reuter • April 22, 2026 -
RFK Jr. defends HHS tenure, 12% proposed budget cut
Republicans largely praised the HHS secretary’s track record during a marathon of House hearings last week, but some GOP lawmakers raised concerns about proposed budget cuts to the National Institutes of Health.
By Emily Olsen • April 20, 2026 -
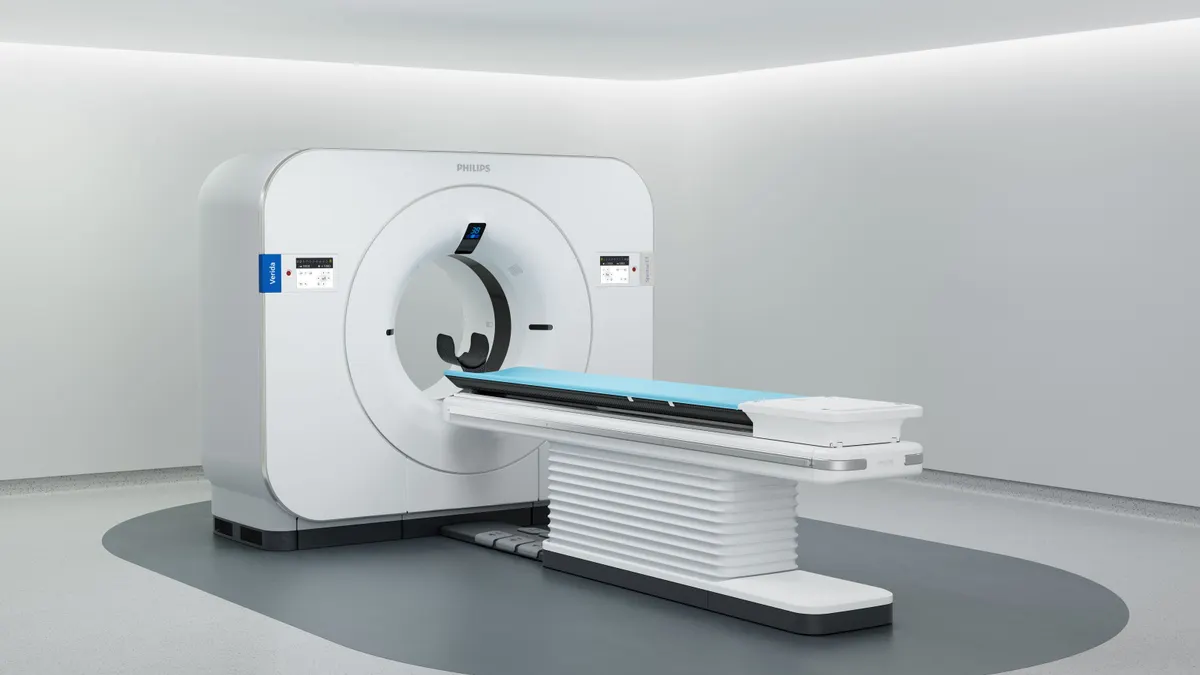
Philips secures FDA clearance for AI-enabled CT system
The company aims to increase its share of the premium CT device market through 2028.
By Nick Paul Taylor • April 17, 2026 -
FDA, medtech industry near MDUFA VI agreement
The FDA said it intends to hire “substantial numbers” of medical device review staff as part of the agreement for the next medical device user fee amendments.
By Elise Reuter • April 9, 2026 -
CDRH targets hospital readmissions with home device innovation challenge
The center plans to pick nine devices to help support patients and caregivers after a hospital stay.
By Elise Reuter • April 9, 2026 -
UK steps up push to align medtech regulations with the US
Health regulators on both sides of the Atlantic are assessing alignment opportunities and exploring mutual recognition mechanisms.
By Nick Paul Taylor • April 7, 2026 -
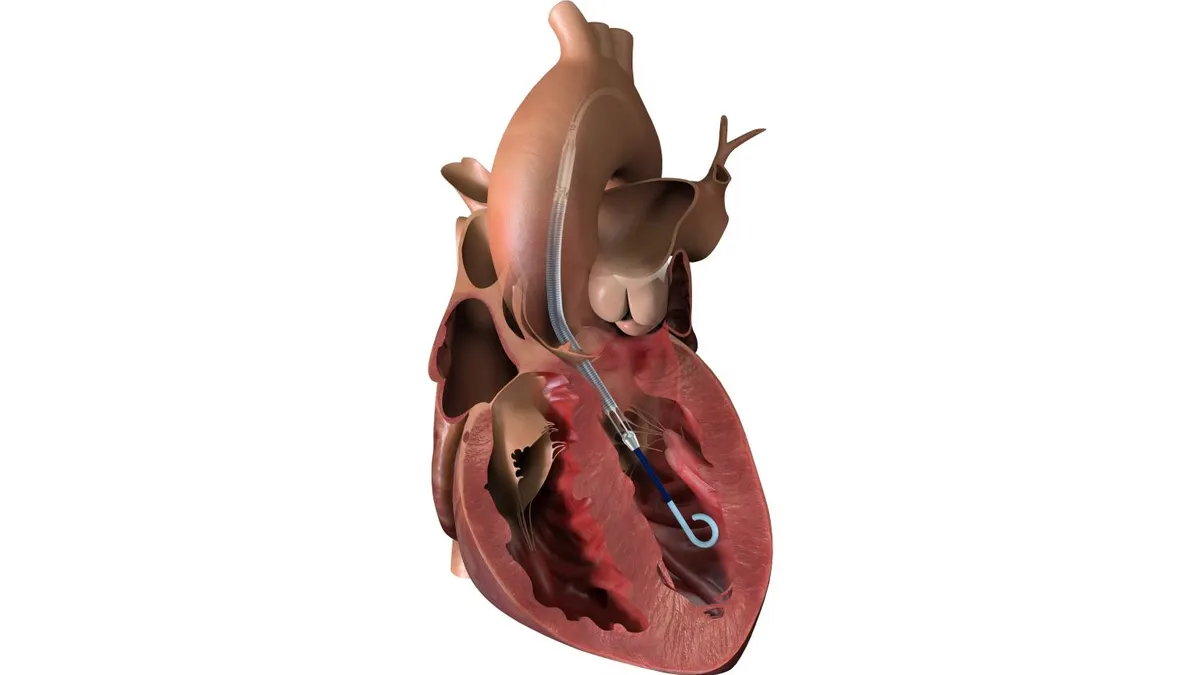
VDyne secures FDA nod to start pivotal trial for tricuspid valve
With approval to study the transcatheter replacement device in patients with severe tricuspid regurgitation, VDyne is advancing a treatment to compete with Edwards Lifesciences’ Evoque system.
By Susan Kelly • April 6, 2026 -
Deep Dive
‘Cracks show’ as CDRH staff contend with heavy workloads
Former device leaders said a culture of “fear and anxiety” has driven experienced staff from the Food and Drug Administration. An overburdened workforce has affected communications from the agency.
By Elise Reuter , Shaun Lucas • April 1, 2026 -
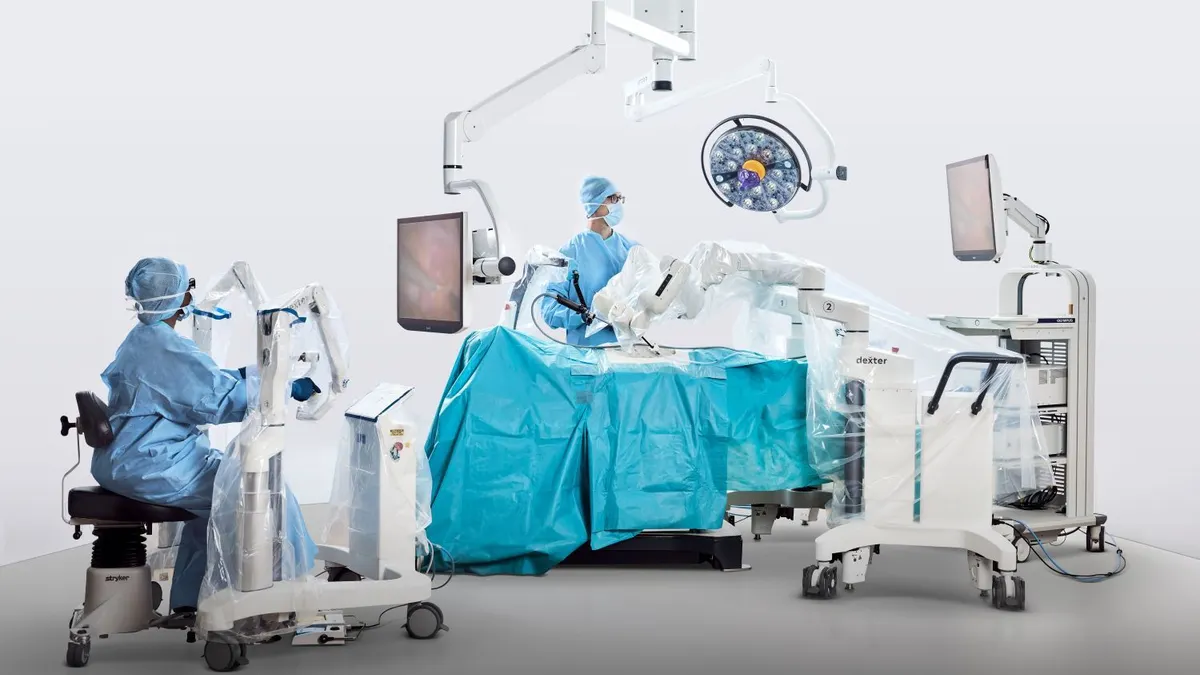
Distalmotion targets ASC robotic gynecology programs with FDA filing
The company is seeking clearance in additional gynecological indications to support outpatient use of its surgical robot.
By Nick Paul Taylor • April 1, 2026 -
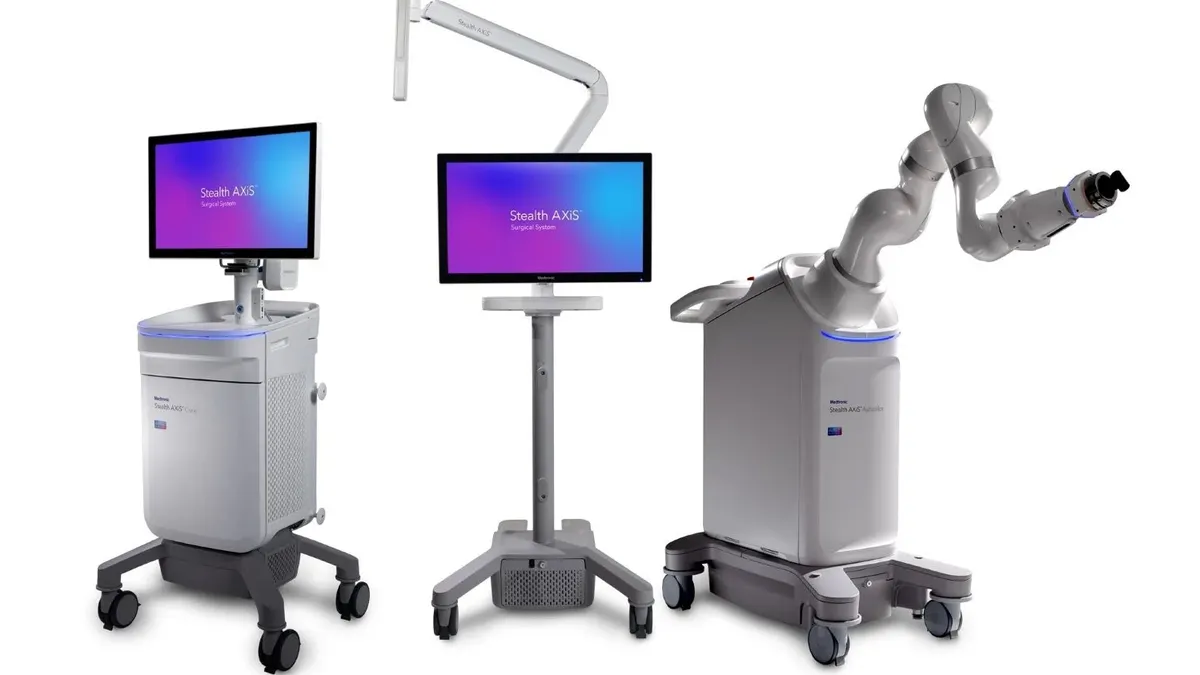
Medtronic wins FDA clearance for robot in cranial, ENT surgeries
The surgical navigation and robotics system is part of the company’s push to expand its share of the $15 billion cranial and spinal technologies market.
By Nick Paul Taylor • March 30, 2026 -
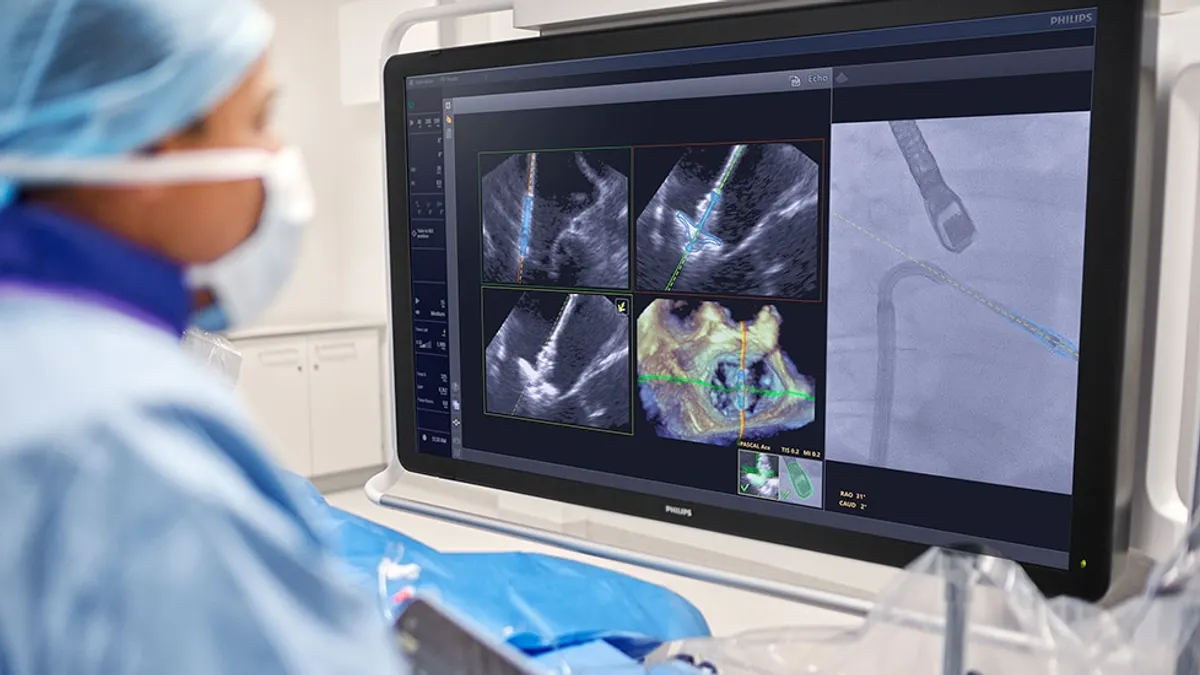
Philips wins FDA clearance for imaging device developed with Edwards
The system uses AI to optimize the placement of Edwards’ mitral valve repair device.
By Nick Paul Taylor • March 27, 2026