FDA: Page 68
-

 "State Public Health Laboratory in Exton Tests for COVID-19" by Governor Tom Wolf is licensed under CC BY 2.0
"State Public Health Laboratory in Exton Tests for COVID-19" by Governor Tom Wolf is licensed under CC BY 2.0
UK study to validate, compare COVID-19 tests in real-world settings
The hospital arm will assess 10 to 20 new diagnostics the National Health Service has identified as high priority, including those designed to deliver results at the point of care in minutes, which could include Abbott’s ID NOW.
By Nick Paul Taylor • June 25, 2020 -
Fauci: Trump never asked to slow down testing, says US needs more, not less
During a House panel Tuesday, top U.S. infectious disease doctor Anthony Fauci and other public health officials denied they'd been directed to impede testing efforts as the White House targets up to 50 million tests a month by fall.
By Rebecca Pifer Parduhn • June 24, 2020 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
Medtronic Evolut TAVR system gets expanded label in Europe
New indications allow a less-invasive alternative to traditional open heart surgery for a group of severe aortic stenosis patients who tend to be younger and more active, and in higher-risk patients with bicuspid aortic valves.
By Susan Kelly • June 23, 2020 -
FDA affirms potential to miss MDUFA deadlines, weighs virtual advisory panels
Separately, the agency announced virtual meetings in September on using patient preference information in medical device regulatory decisions and considering patient-reported outcomes in evaluating devices.
By Nick Paul Taylor • June 23, 2020 -
Mainstay wins FDA approval for chronic back pain device following mixed clinical data
The premarket approval comes 19 months after the ReActiv8 neurostimulation system failed a clinical trial aimed at generating data to break into the U.S. market.
By Nick Paul Taylor • June 23, 2020 -

 Dufour, Tia. (2020). "White House Press Briefing" [Photograph]. Retrieved from Flickr.
Dufour, Tia. (2020). "White House Press Briefing" [Photograph]. Retrieved from Flickr.
Pandemic pushes FDA to 'accelerate' real-world evidence efforts, Hahn says
The agency has grappled with how to leverage real-world data in regulating medical devices, and is now leaning on it to update emergency use authorizations, FDA chief Stephen Hahn said at a forum on Thursday.
By Maria Rachal • June 19, 2020 -
Lack of data encryption for Baxter devices flagged in flurry of DHS alerts
The Department of Homeland Security's cyber agency published four advisories, ranging from 7.5 to 8.6 on its 10-point severity scale, about vulnerabilities in infusion pumps, hemodialysis delivery systems and other tech.
By Nick Paul Taylor • June 19, 2020 -
Senators back sustaining telehealth momentum post pandemic
Top priorities include axing geographic restrictions, expanding Medicare and Medicaid reimbursement for virtual care services, and upping use of remote patient monitoring and digital health tools for patients with chronic conditions.
By Rebecca Pifer Parduhn • June 18, 2020 -
FDA cracks down on sellers of at-home COVID-19 antibody tests
A series of warning letters is the latest example of U.S. regulators weeding out serology tests that don't meet the agency's standards.
By Nick Paul Taylor • June 18, 2020 -
TÜV SÜD becomes 4th notified body designated under IVDR
It's the first notified body OK'd by the European Commission in 2020 to increase capacity for the forthcoming In Vitro Diagnostic Regulation, set to come into force in less than two years.
By Nick Paul Taylor • June 18, 2020 -
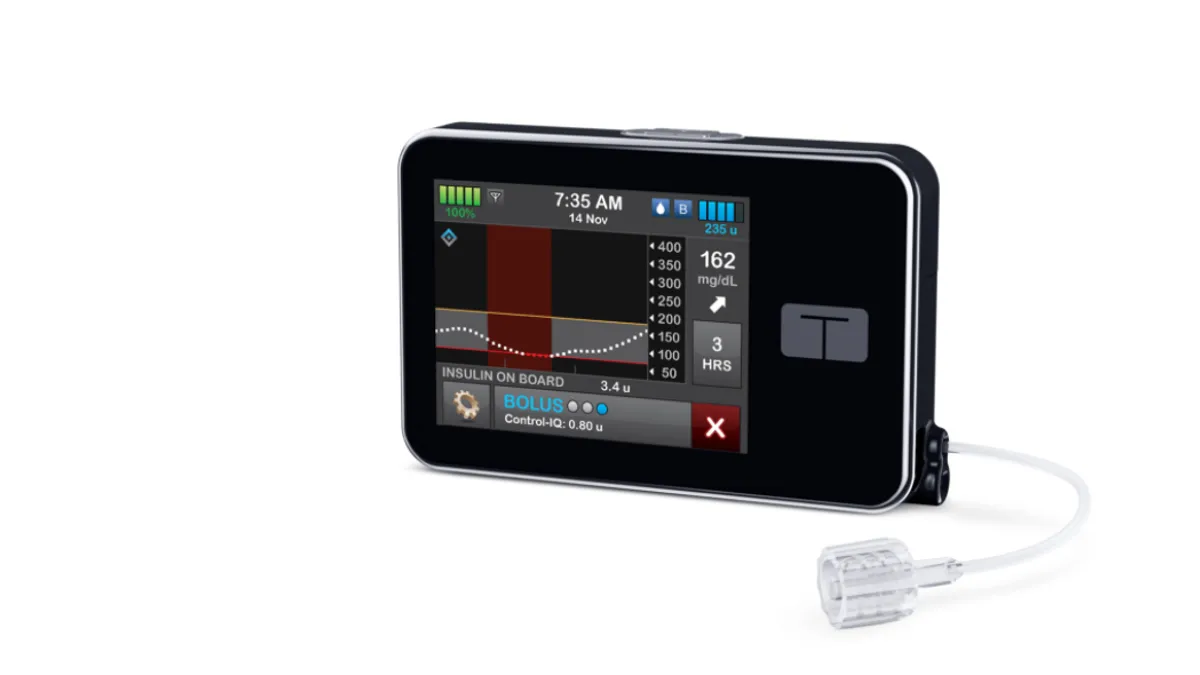
Tandem's insulin dosing algorithm OK'd for use in younger kids
FDA's decision to expand the pediatric indication was supported by an NIH-funded trial that found the automated insulin dosing system helped improve time in range for children ages 6 to 13.
By Maria Rachal • June 17, 2020 -
Thermo Fisher, MedRhythms gain breakthrough device designations
A companion diagnostic for certain brain tumor patients and a digital therapeutic for stroke patients with walking impairments are among the latest technologies tapped by FDA for the priority review program.
By Susan Kelly • June 17, 2020 -
FDA revokes emergency authorization for Chembio's COVID-19 antibody test
It's the first time the agency has exercised such authority for a coronavirus test, citing more false results than expected. Chembio Diagnostics' stock plunged 60% Wednesday.
By Greg Slabodkin • June 17, 2020 -

 Retrieved from Akili Interactive/Businesswire on June 15, 2020
Retrieved from Akili Interactive/Businesswire on June 15, 2020
In FDA 1st, game-based therapeutic gets marketing OK
Digital therapeutics developer Akili Interactive Labs won De Novo authorization for a treatment meant to improve attention function in children with ADHD, building on a rollout it began under a special COVID-19 policy.
By Maria Rachal • June 16, 2020 -
Heart rhythm bodies see 'clear concerns' with using wearables to detect arrhythmias
A paper published on Monday by the Heart Rhythm Society, along with counterpart groups in Asia, Europe and Latin America, found wearable-triggered false positives can cause unwarranted concerns and screening.
By Nick Paul Taylor • June 16, 2020 -
Labs urge CDC to change COVID-19 antibody test guidelines
The trade group for Quest Diagnostics and LabCorp criticized the federal agency's advice against use of serology test results to make decisions about who can return to work.
By Nick Paul Taylor • June 16, 2020 -
Paclitaxel-coated balloon, stent labels in Europe to have mortality warning update
A notice posted by British medical device regulators outlined 12 affected products including Boston Scientific's ELUVIA stent, Cook Medical's Zilver PTX stent, and Medtronic's IN.PACT Admiral balloon.
By Greg Slabodkin • June 15, 2020 -
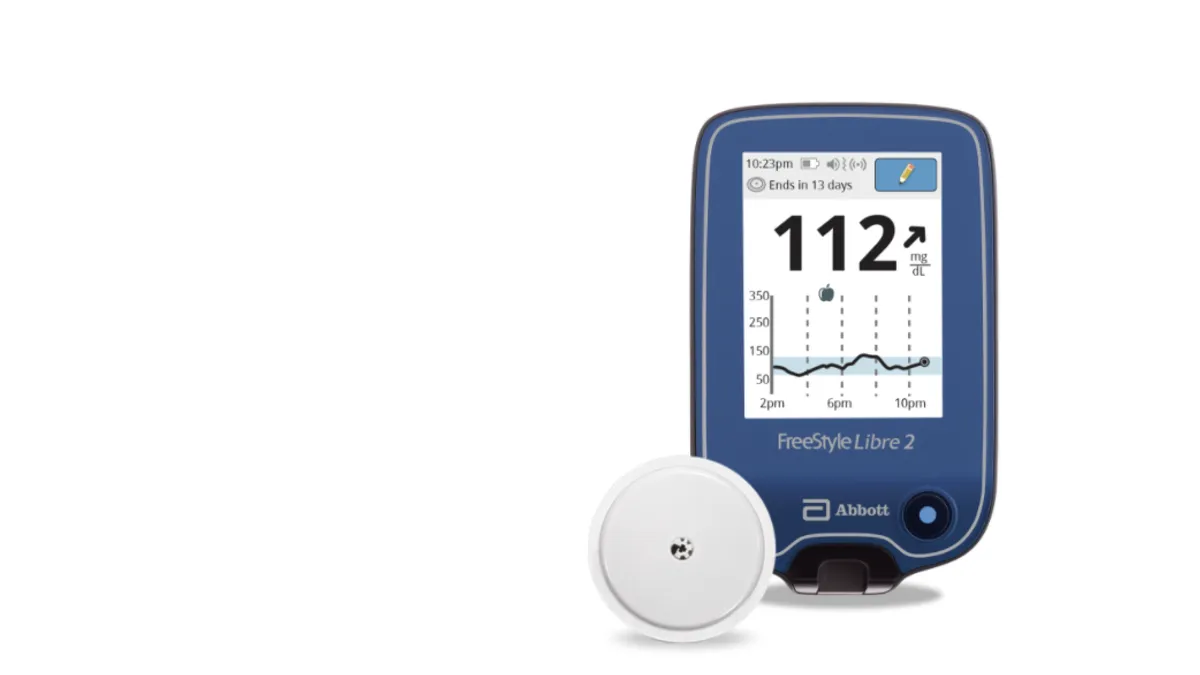
 Retrieved from Abbott/PRNewswire on June 15, 2020
Retrieved from Abbott/PRNewswire on June 15, 2020
Abbott's FreeStyle Libre 2 gets long-awaited iCGM nod from FDA
The designation may stiffen competition for Dexcom, whose G6 device was the sole integrated continuous glucose monitor on the U.S. market. But Abbott's device isn't yet OK'd for use with automated insulin dosing systems.
By Nick Paul Taylor , Maria Rachal • June 15, 2020 -
CMS to review at-home ventilation of COPD patients amid variability in device use
Findings from a July Medicare Evidence Development & Coverage Advisory Committee meeting and resulting policy changes could potentially impact ResMed and Philips Respironics.
By Nick Paul Taylor • June 12, 2020 -
MedTech Europe pitches virtual audits to clear MDR coronavirus logjam
The trade group contends technology such as smart glasses and webcams can enable notified bodies to view facilities without having to visit in person.
By Nick Paul Taylor • June 11, 2020 -
Ex-FDA chief scientist slams 'chaos' caused by agency approach to antibody tests
Jesse Goodman said the agency’s decision to allow developers of COVID-19 serology tests to self-validate their products led unqualified entities to flood the market.
By Nick Paul Taylor • June 10, 2020 -
Edwards wins approval to introduce Sapien 3 valve in China
The transcatheter heart device specialist has been eyeing the wealthy, aging nation's untapped market potential for new cardiovascular disease treatments.
By Susan Kelly • June 9, 2020 -
Lawmakers push HHS to funnel funds to clinical labs amid routine testing decline
Members of Congress on both sides of the aisle argue the investments laboratories are making to scale up COVID-19 testing mean they need more targeted financial support, which should come out of a $25 billion relief fund.
By Nick Paul Taylor • June 9, 2020 -
Surmodics gets CE mark for Abbott-backed paclitaxel-coated balloon
The company will receive a $10.8 million milestone payment from Abbott, which licensed exclusive commercialization rights to the device for peripheral artery disease patients in 2018 in a bid to take on Medtronic's drug-coated balloons.
By Maria Rachal • June 8, 2020 -
COVID-19 false negatives fuel call for urgent focus on asymptomatic patients
Harvard and Yale physicians writing in the New England Journal of Medicine called for development of a reference standard for measuring sensitivity of coronavirus tests in those without symptoms who may infect others.
By Nick Paul Taylor • June 8, 2020