FDA: Page 72
-
Small PAMA victory in aid package, but labs remain in 'untenable situation'
Quest, LabCorp and others scaling up U.S. coronavirus testing will see a delay in Medicare payment reductions as part of the CARES Act signed into law Friday, but industry argues labs have been shortchanged on emergency funds.
By Maria Rachal • Updated March 27, 2020 -
FDA OKs anesthesia gas machines as ventilators amid coronavirus shortage fears
GE Healthcare, which has more than 100,000 of its anesthesia devices worldwide, noted differences but acknowledged the "need to weigh the relative risks and benefits to support patients in these unprecedented times."
By Greg Slabodkin • March 26, 2020 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
UnitedHealth says self-swabbing study spurred FDA coronavirus policy change
The agency now allows onsite, self-collection of samples, after a nearly 500-patient study found self-administered tests about as accurate as those carried out by clinicians.
By Nick Paul Taylor • March 26, 2020 -
Medtech industry asks for FEMA's help allocating ventilators to frontlines
AdvaMed said a lead federal agency should be designated to manage distribution of the breathing machines as manufacturers rapidly scale up production in response to the COVID-19 crisis.
By Susan Kelly • March 25, 2020 -
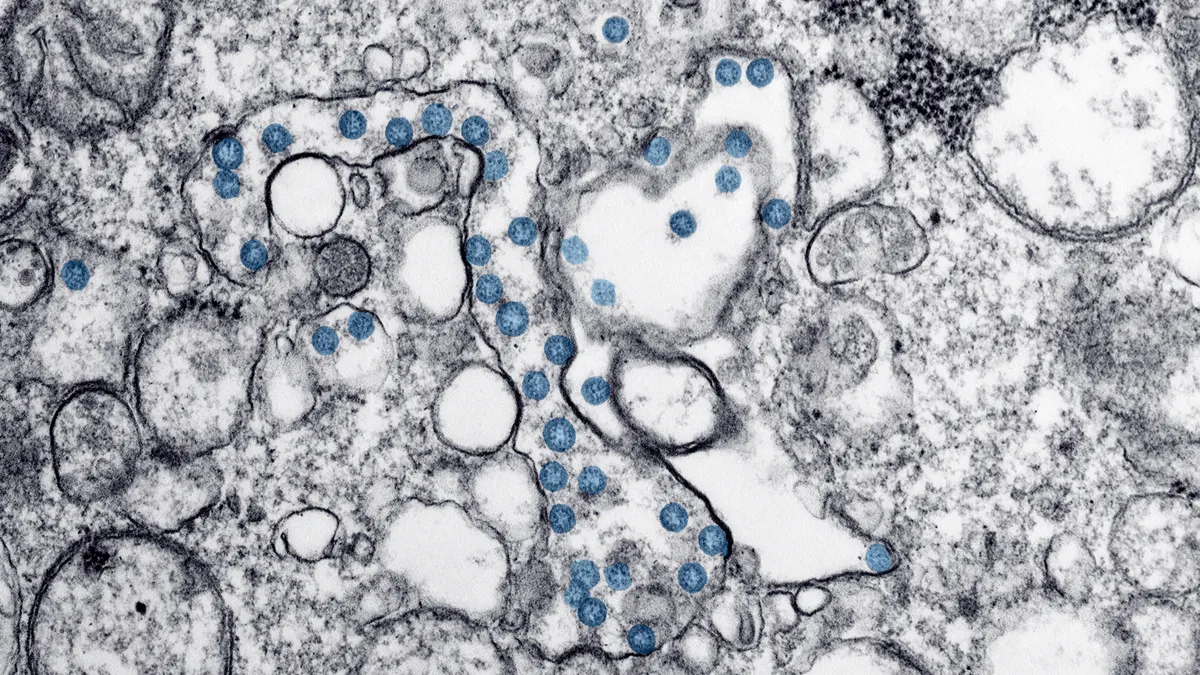
 U.S. Centers for Disease Control. "CDC 2019-Novel Coronavirus (2019-nCoV) test kit". Retrieved from https://www.cdc.gov/coronavirus/2019-ncov/about/testing.html.
U.S. Centers for Disease Control. "CDC 2019-Novel Coronavirus (2019-nCoV) test kit". Retrieved from https://www.cdc.gov/coronavirus/2019-ncov/about/testing.html.
How health systems are responding as COVID-19 squeezes the medical supply chain
Procurement of supplies will become more difficult if cases surge and federal stockpiles don't trickle down to the state level quickly enough.
By Deborah Abrams Kaplan • March 25, 2020 -
EU regulators propose 1-year MDR delay
The European Commission formally proposed Friday the Medical Device Regulation start date be moved to May 26, 2021. It needs the quick backing of the European Parliament and Council.
By Maria Rachal • Updated April 3, 2020 -
FDA still trying to fine-tune Pre-Cert as pilot enters 2020
The Pre-Cert pilot, which includes Apple, J&J and Fitbit, is moving forward with the agency "working through the nine companies, trying to figure out how they actually do business," FDA's Bakul Patel told MedTech Dive.
By Greg Slabodkin • March 25, 2020 -
Clinical labs, White House tell different stories on testing backlog
The American Clinical Laboratory Association said without sufficient funds from the federal government, commercial labs might be forced to "retrench rather than keep building the capacity that is needed."
By Greg Slabodkin • March 24, 2020 -
MedTech Europe calls for delay of MDR until coronavirus crisis passes
The trade group argued industry is “working relentlessly” on getting devices to healthcare professionals fighting COVID-19, while simultaneously trying to maintain supply of other products, leaving little bandwidth to focus on new regs.
By Nick Paul Taylor • March 24, 2020 -
FDA eases rules on ventilator makers as Medtronic and Tesla talk
Hardware and software modifications can occur without submission of a 510(k) as traditional medtechs team with other manufacturers to meet coronavirus-driven demand for the devices.
By Nick Paul Taylor • March 23, 2020 -
FDA greenlights expanded remote patient monitoring to keep patients out of coronavirus-slammed hospitals
The agency said it wants to help healthcare providers diagnose and treat patients who remain at home in the midst of the national emergency.
By Susan Kelly • March 20, 2020 -
Lab industry 'alarmed,' says Senate stimulus bill fails to back testing ramp-up
Without $5 billion in assistance, the American Clinical Laboratory Association contends commercial labs are "being set up to perform COVID-19 testing at a loss." A Senate vote could come Monday.
By Greg Slabodkin • Updated March 23, 2020 -
US won't suspend China tariffs during coronavirus outbreak
The tariffs present a risk and additional cost to medical supply chains as the need for critical products grows urgent.
By Shefali Kapadia • March 19, 2020 -
CMS urges hospitals to put off elective procedures
The guidelines suggest three tiers, with procedures like hip and knee replacements falling into the "consider postponing" category.
By Shannon Muchmore • March 19, 2020 -

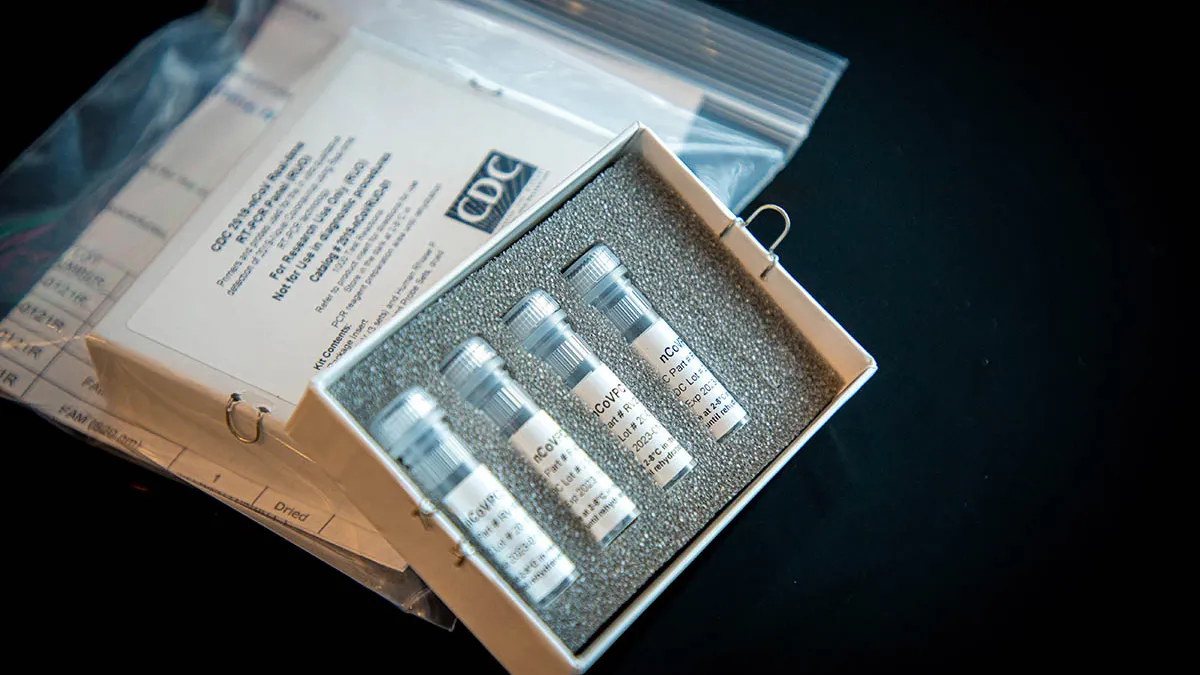
 Retrieved from Abbott, PRNewswire on March 19, 2020
Retrieved from Abbott, PRNewswire on March 19, 2020
Abbott latest to get FDA emergency use nod for coronavirus test
It's the seventh medtech to win the agency's emergency use authorization within the last week as the Trump administration attempts to leverage the private sector, including commercial labs, to boost the nation's testing capacity.
By Greg Slabodkin • March 19, 2020 -
New notified bodies trickle in ahead of looming MDR start date
The 12th notified body authorized for MDR work, and the first from Hungary, was listed in the NANDO database Friday: CE Certiso.
By Nick Paul Taylor • Updated March 20, 2020 -
How an 'overwhelmed' Buffalo lab facing coronavirus supply shortage signals nationwide struggles
The Western New York regional public health lab is a microcosm of the insufficient swab and reagent stocks labs face as they attempt to confirm COVID-19 cases and gauge the scope of their region's outbreak.
By Greg Slabodkin • March 18, 2020 -
MedTech Europe sounds alarm over 'inconsistent' enforcement amid flurry of MDR updates
A lack of harmonized standards worries the trade association. Meanwhile, the Medical Device Coordination Group issued new documents on software requirements, implant cards and unique device identifiers.
By Nick Paul Taylor • March 18, 2020 -
As coronavirus roils region, EU device body adamant about steps to hit MDR deadline
The Medical Device Coordination Group acknowledged implementation “has proven to be a very challenging task” but for now appears to be pushing ahead with the planned May start date.
By Nick Paul Taylor • March 17, 2020 -
Hologic, LabCorp win emergency use nods as FDA eases rules to boost US coronavirus test volume
In some cases, manufacturers can distribute, and labs can use, new commercially developed tests prior to an emergency use authorization. FDA's updated policy also boosts states' authority to greenlight tests.
By Greg Slabodkin • March 17, 2020 -

 Official White House Photo by Andrea Hanks. (2020). "President Trump meets with the Coronavirus Task Force" [Photograph]. Retrieved from https://www.flickr.com/photos/whitehouse/49613832638/in/photostream/.
Official White House Photo by Andrea Hanks. (2020). "President Trump meets with the Coronavirus Task Force" [Photograph]. Retrieved from https://www.flickr.com/photos/whitehouse/49613832638/in/photostream/.
To fight coronavirus spread, high-throughput tests bring potential benefits, challenges
Deborah Birx, White House coronavirus response coordinator, said labs will just as importantly need supplies to ramp up testing. The American Clinical Laboratory Association is concerned about potential supply shortages.
By Greg Slabodkin • March 15, 2020 -
Thermo Fisher coronavirus test gets FDA nod
Trump administration officials promised on Sunday that 2,000 labs would be running 1.9 million tests this week. Separately, the American Clinical Laboratory Association expects daily testing capacity to exceed 20,000 next week, with the potential to grow to 280,000 by April 1.
By Maria Rachal • March 15, 2020 -
Trump declares national emergency, opening up billions for coronavirus
The president also teased an imminent FDA emergency use authorization for a test from Thermo Fisher, with LabCorp and Quest saying they're collaborating with Roche.
By Samantha Liss • Updated March 13, 2020 -
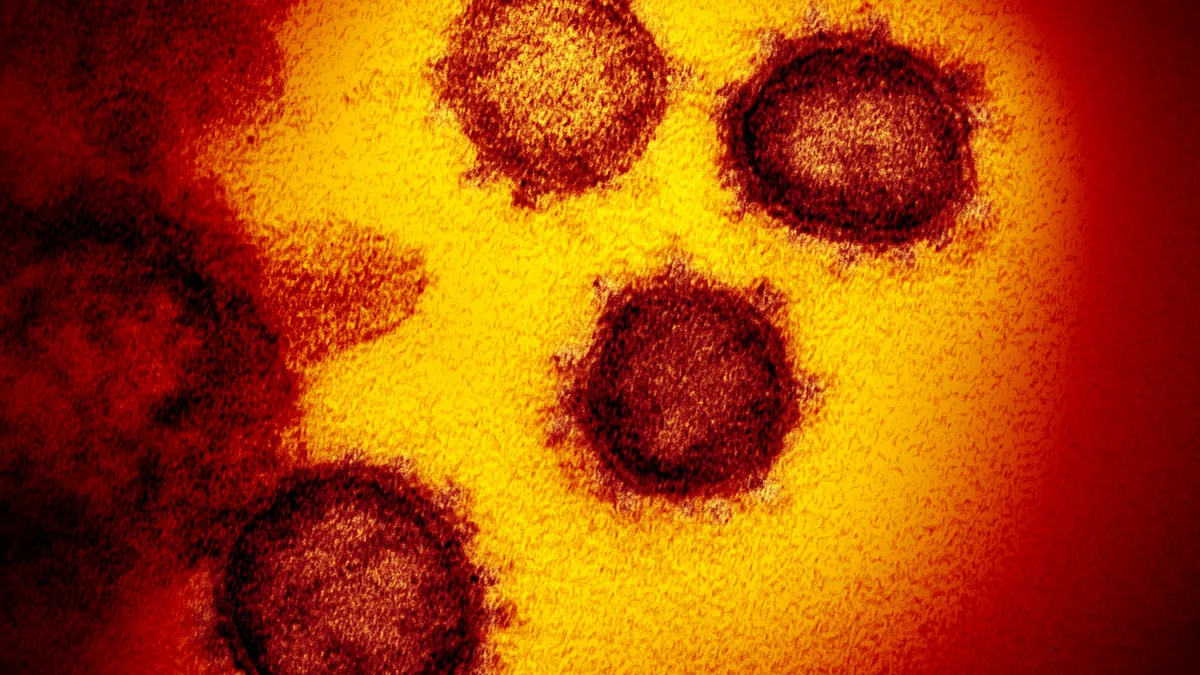
 National Institute of Allergy and Infectious Diseases. (2020). "Novel coronavirus SARS-CoV-2" [Microscope image]. Retrieved from https://www.flickr.com/photos/nihgov/49535193876/in/album-72157713108522106/.
National Institute of Allergy and Infectious Diseases. (2020). "Novel coronavirus SARS-CoV-2" [Microscope image]. Retrieved from https://www.flickr.com/photos/nihgov/49535193876/in/album-72157713108522106/.
CDC chief: Reagents critical to coronavirus tests 'now are in short supply'
Separately, the government Friday announced funding for development of COVID-19 diagnostic tests from DiaSorin Molecular and Qiagen meant to detect the pathogen in about an hour.
By Greg Slabodkin • March 13, 2020 -
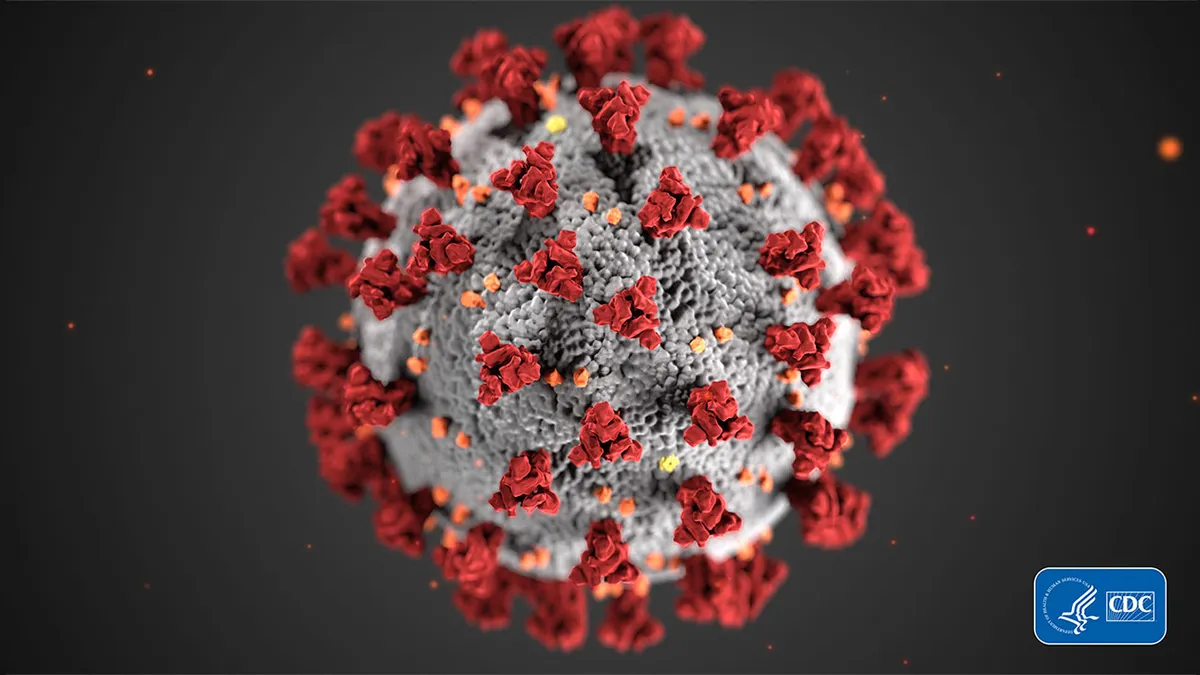
 CDC/Alissa Eckert, MS. "covid-19 coronavirus on black background". Retrieved from https://www.cdc.gov/media/subtopic/images.htm.
CDC/Alissa Eckert, MS. "covid-19 coronavirus on black background". Retrieved from https://www.cdc.gov/media/subtopic/images.htm.
As US officials shift tone on coronavirus, Trump stops short of 1 provider ask
The president did not declare a disaster or national emergency, which groups like the American Hospital Association had sought.
By Samantha Liss , Shannon Muchmore • March 12, 2020