Cardiac: Page 8
-

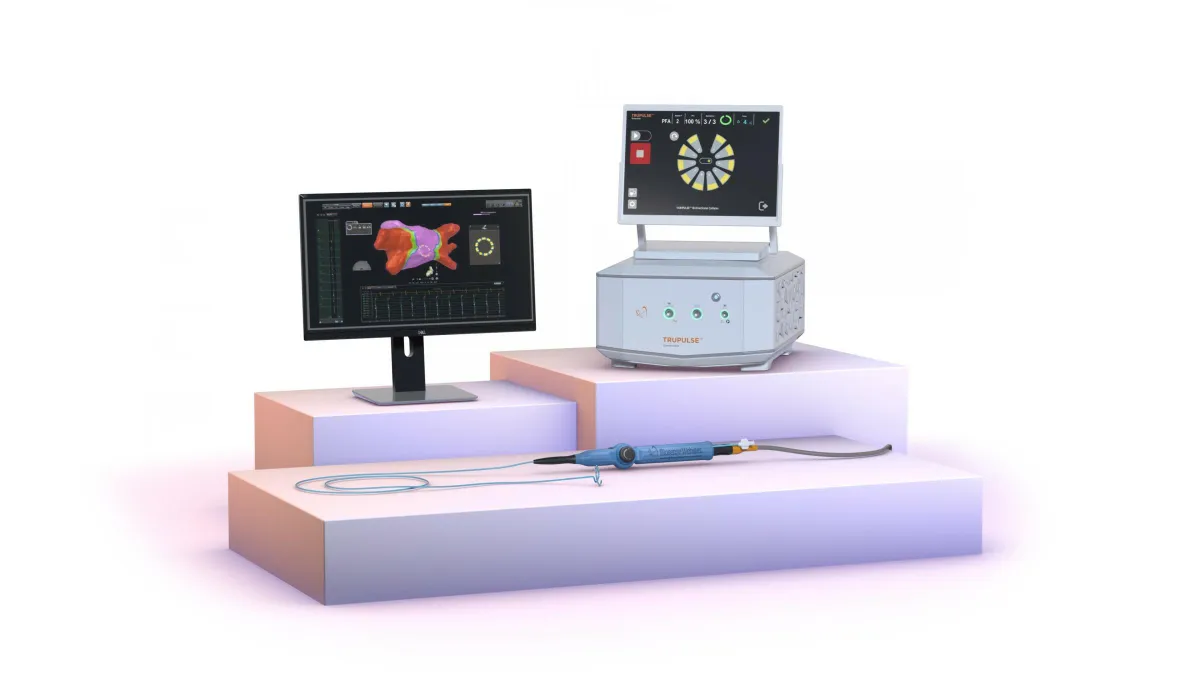
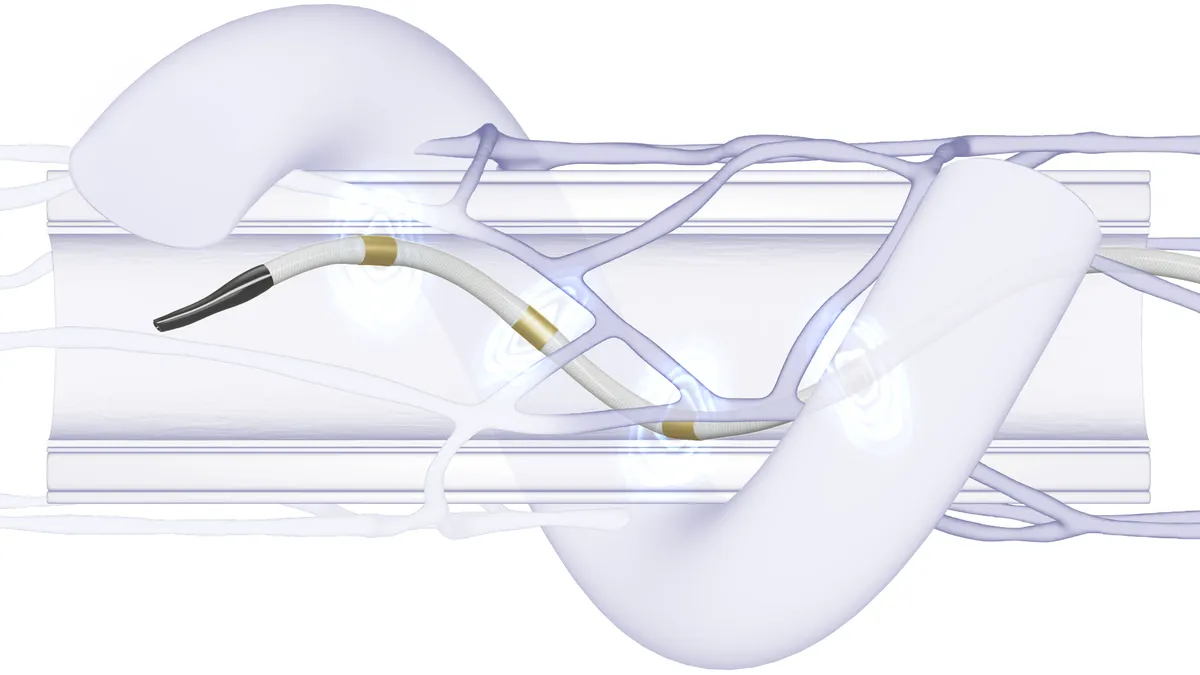
 Retrieved from Merit Medical on December 17, 2024
Retrieved from Merit Medical on December 17, 2024
Merit Medical president to resign after unspecified conduct allegations
Joseph Wright’s resignation is effective in January, and CEO and founder Fred Lampropoulos has been reappointed president.
By Susan Kelly • Dec. 18, 2024 -
PFA to surpass radiofrequency ablation in 2025: Citi survey
Pulsed field ablation could soon be used in the majority of electrophysiology procedures to treat atrial fibrillation as Medtronic, Boston Scientific and Johnson & Johnson battle for market share.
By Susan Kelly • Dec. 16, 2024 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
Capstan Medical raises $110M for heart valve with robotic system
The startup’s third round of financing includes support from Intuitive Surgical’s venture capital arm.
By Susan Kelly • Dec. 12, 2024 -
Artivion says cybersecurity incident has disrupted order, shipping processes
The heart device manufacturer has taken certain systems offline in response to the incident, which involved the acquisition and encryption of files.
By Nick Paul Taylor • Dec. 11, 2024 -
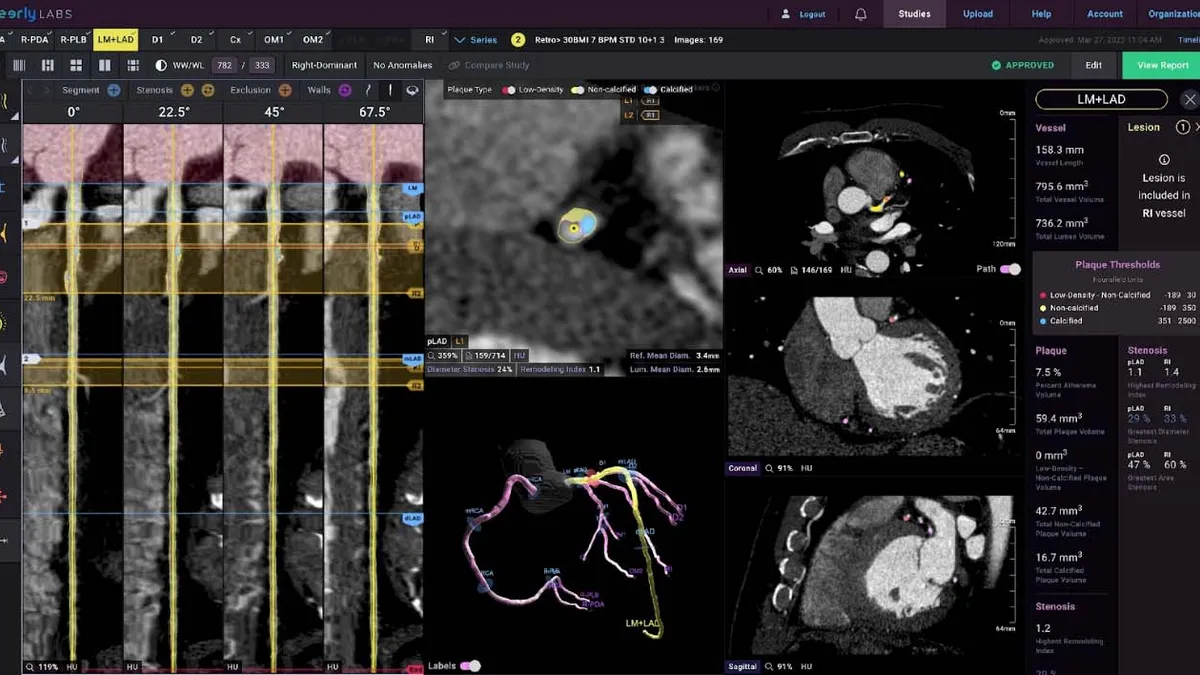
 Retrieved from Cleerly on December 06, 2024
Retrieved from Cleerly on December 06, 2024
Cleerly raises $106M to fund AI-driven heart disease tool
The company has generated evidence that shows its system is better than experts at assessing vessel narrowing, plaque volume and composition.
By Nick Paul Taylor • Dec. 6, 2024 -
Edwards expects new TAVR approval in mid-2025 to boost sales
U.S. approval of an expanded indication for transcatheter aortic valve replacement in patients without symptoms is among the growth catalysts Edwards outlined in an investor day presentation.
By Susan Kelly • Dec. 5, 2024 -
Haemonetics to offload whole-blood business for up to $67M
Selling the assets to Italy’s GVS should help Haemonetics improve its revenue growth and profit margins, analysts said.
By Susan Kelly • Dec. 4, 2024 -
Biden administration proposes Medicare, Medicaid coverage of pricey weight loss drugs
About 7.4 million Americans with obesity could have insurance coverage for drugs like Wegovy if the Trump administration allows the rule to go into effect — at the cost of $40 billion to federal and state governments.
By Rebecca Pifer Parduhn • Nov. 27, 2024 -
Medtronic’s ablation sales slowed by PFA supply issues
CEO Geoff Martha said the business did not meet internal growth expectations last quarter due to third-party supply issues that have now been resolved.
By Ricky Zipp • Nov. 19, 2024 -
Boston Scientific’s Watchman could be new option for patients post ablation: study
The trial compared the stroke prevention device for left atrial appendage closure to blood thinners in people who underwent cardiac ablation for AFib.
By Susan Kelly • Nov. 18, 2024 -
The PFA race is heating up. Here’s where the market stands after J&J’s approval.
Boston Scientific, Medtronic and J&J are moving quickly to secure their place in the fast-growing pulsed field ablation space as new devices and heart imaging technology hit the market.
By Susan Kelly • Nov. 18, 2024 -
Haemonetics to close California facility, lay off 75 people
Over the past year, Haemonetics has reshaped its product portfolio through restructuring and M&A, adding devices used in cardiology procedures.
By Susan Kelly • Nov. 14, 2024 -
Boston Scientific resumes PFA trial in new patient group after pausing enrollment
Boston Scientific expects to complete enrollment in the coming months in the study comparing pulsed field ablation to anti-arrhythmic drugs in patients with persistent AFib, a spokesperson said.
By Susan Kelly • Nov. 12, 2024 -
J&J wins FDA approval for Varipulse PFA system
J&J is the third medtech company to gain U.S. approval for pulsed field ablation, a new atrial fibrillation treatment seeing rapid adoption.
By Susan Kelly • Nov. 7, 2024 -
Medtronic, Recor win CMS transitional payment coverage for renal denervation devices
Patient and physician education is the next challenge to change the treatment paradigm for drug-resistant hypertension, Citi Research analyst Joanne Wuensch said.
By Susan Kelly • Nov. 5, 2024 -
Edwards, Medtronic and Inari share trial data at TCT conference
The three medtech firms unveiled new data this week during TCT 2024 in Washington, D.C. Here’s what the results mean for the companies and their device markets.
By Nick Paul Taylor • Oct. 31, 2024 -
Boston Scientific’s Acurate Neo2 inferior to rival TAVR valves in study
Analysts said the findings complicate the outlook for a U.S. launch of Boston Scientific's aortic valve replacement device.
By Susan Kelly • Oct. 31, 2024 -
Edwards data show benefit of early TAVR over ‘watchful waiting’
Study author Philippe Genereux said the trial data “shatter 60 years of ingrained belief on the treatment for severe aortic stenosis.”
By Susan Kelly • Oct. 29, 2024 -
J&J to seek FDA approval after small-bore Impella heart pump hits trial goal
The company has predicted the narrower Impella ECP will be easier to insert and implant, as well as enable the use of small bore access and closure techniques.
By Nick Paul Taylor • Oct. 29, 2024 -
Boston Scientific to close Silk Road Medical headquarters, lay off 138 people
Following its acquisition of Silk Road in September, Boston Scientific said it plans to consolidate the company’s work in Minnesota.
By Elise Reuter • Oct. 28, 2024 -
Edwards’ TAVR sales faced ongoing hospital constraints in Q3
Hurt by a slowdown in procedures for its heart valve replacement devices, Edwards said it is working with hospitals to manage workflow challenges.
By Susan Kelly • Oct. 25, 2024 -
Medtronic wins FDA approval for Affera mapping and ablation system
Affera combines mapping technology with a catheter capable of performing radiofrequency and pulsed field ablation.
By Nick Paul Taylor • Oct. 25, 2024 -
Boston Scientific boosts PFA expectations, citing rapid Farapulse adoption
Demand for the pulsed field ablation device drove the company’s electrophysiology sales up 177% in the third quarter compared to a year ago.
By Susan Kelly • Oct. 23, 2024 -

 Retrieved from iRhythm on October 22, 2024
Retrieved from iRhythm on October 22, 2024
iRhythm’s Zio AT design changes win FDA clearance
The agency cleared one of two 510(k) submissions iRhythm filed for the heart monitor after receiving a warning letter from the agency last year.
By Susan Kelly • Oct. 23, 2024 -
Boston Scientific, Edwards and Dexcom usher in second week of earnings
Johnson & Johnson, Abbott and Intuitive Surgical made a mixed start to earnings season last week.
By Nick Paul Taylor • Oct. 22, 2024