Medical Devices: Page 101
-
Edwards posts 97% success rate with tricuspid device in 30-day test as it prepares for Abbott rival
Edwards' device posted a 97% implant success rate, while Abbott's had a 98% success rate. Both are part of a growing market for transcatheter valve repair devices.
By Nick Paul Taylor • May 20, 2022 -
FDA gets over 21,000 medical device reports, including 124 deaths, linked to Philips foam breakdown
The medical device reports were made between April 2021 and April 2022. Both Philips and the FDA contend that the reporting system has limitations, including the underreporting of events and inaccuracies of reports.
By Ricky Zipp • May 20, 2022 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
FDA user-fee bill goes to House; Senate committee draft revamps diagnostic regulations
The House Energy and Commerce Committee unanimously passed a bill to reauthorize the FDA’s user-fee amendments for the next five years. A Senate committee draft of the bill would add substantive changes.
By Elise Reuter • May 19, 2022 -
Study shows that Abbott's Amulet, Boston Scientific's Watchman devices are riskier for women
The analysis builds on previous studies showing that left atrial appendage occlusion devices are riskier for women. However, Abbott's recent study shows comparable outcomes for men and women at 18 months.
By Nick Paul Taylor • May 19, 2022 -
Medtronic links renal denervation to increased time below blood-pressure threshold
Results from two studies come after the medtech company had a setback in renal denervation in October. Medtronic is expected to report results from its pivotal trial this year.
By Nick Paul Taylor • May 18, 2022 -
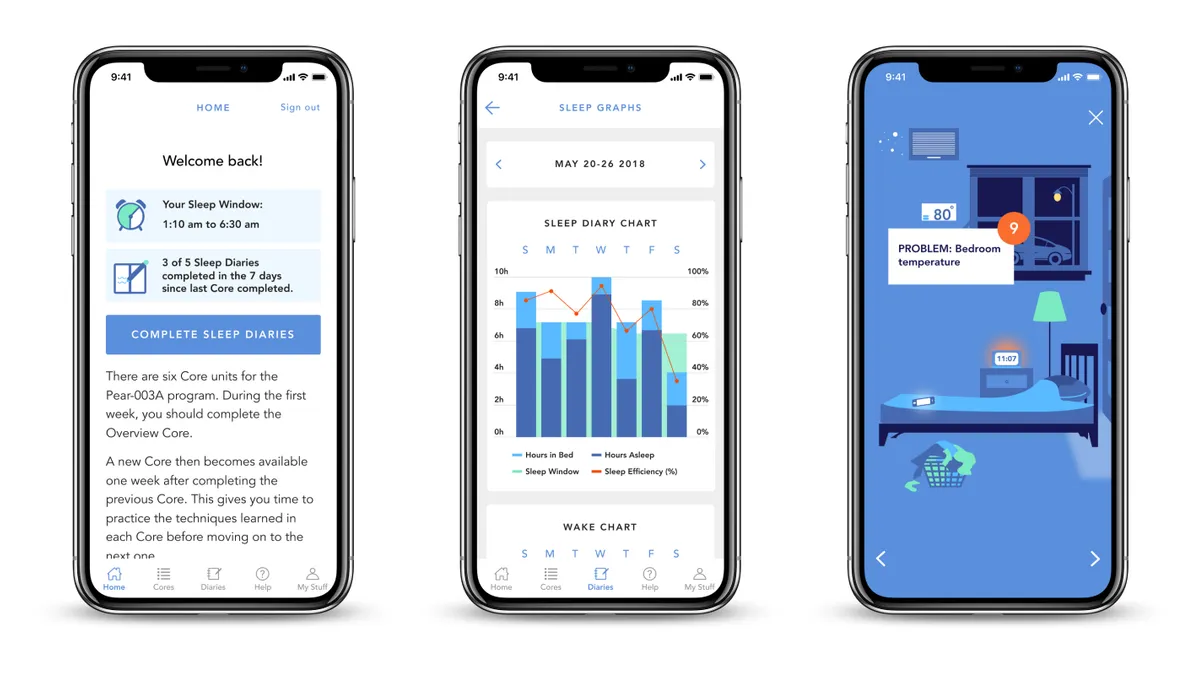
Pear more than doubles revenue from last quarter, shares new economic data
The company is keeping its 2022 revenue forecast of $22 million. Pear also presented economic data, which analysts said would be important in conversations with payers.
By Elise Reuter • May 17, 2022 -
Avanos Medical recall gets Class I label from FDA following patient injury, death reports
The company reported in a recall notice that there have been 60 injuries and 23 patient deaths associated with the Avanos Medical Cortrak 2 Enteral Access System since 2015, according to the FDA.
By Ricky Zipp • May 17, 2022 -
Earnings week 4: Vicarious expects 2024 FDA filing for surgical robot; Steris reports record backlogs
Vicarious Surgical reaffirmed that it would not be making an FDA submission for its surgical robot until 2024, while Globus Medical and Steris discussed rising procedure volumes.
By Elise Reuter • May 16, 2022 -
Dexcom, Insulet, Tandem start 2022 with revenue growth after weathering omicron surge
All three diabetes technology companies reported that omicron pressured business in January before easing as the quarter progressed.
By Ricky Zipp • May 13, 2022 -
House subcommittee debates device remanufacturing definition, sends user-fees bill to next stage
Members of the House Subcommittee on Health passed the user-fees bill by a vote of 30-0 Wednesday, sending the legislation to the full House Committee on Energy and Commerce.
By Elise Reuter • May 12, 2022 -
Roundup: As procedures rebound, supply chain, staffing challenges set to continue for medtechs
Procedure-driven companies saw sales recover at the end of the first quarter, while diagnostics businesses reported increased sales of COVID-19 tests due to the omicron surge in January.
By Elise Reuter • Updated May 19, 2022 -

 Carol Highsmith. (2005). "Apex Bldg." [Photo]. Retrieved from Wikimedia Commons.
Carol Highsmith. (2005). "Apex Bldg." [Photo]. Retrieved from Wikimedia Commons.
Medtronic completes Intersect acquisition following FTC order
The Federal Trade Commission required Medtronic to divest a subsidiary of Intersect ENT to complete the deal. Medtronic announced the $1.1 billion acquisition last August.
By Nick Paul Taylor • Updated May 16, 2022 -
Rising Google searches for procedures suggest recovering demand at medtechs: analysts
Google searches for nonemergent procedures are above pre-pandemic levels, providing another data point to indicate that demand is recovering, according to a Needham report.
By Nick Paul Taylor • May 11, 2022 -
Q&A
Dexcom Chief Tech Officer Leach discusses G7's European launch and FDA review, expanding Dexcom One
The executive also comments on the continuous glucose monitor maker's next steps for the G7 system as well as its plans for reaching new patients and expanding to non-diabetic users.
By Ricky Zipp • May 10, 2022 -
House user-fees bill details clinical trial diversity, cybersecurity requirements
The legislation would let the FDA bring in $1.78 billion in fee revenue from 2023 to 2027 to fund the review of medical devices. That amount could increase to $1.9 billion if the agency meets certain performance goals.
By Elise Reuter • May 10, 2022 -
Earnings week 3: Procedure rebounds, stock price declines, iRhythm's comeback
AtriCure and Axonics reported procedure volume rebounds and both firms increased their 2022 forecasts. Still, the share prices of the companies dropped late last week.
By Ricky Zipp • May 9, 2022 -
FDA finalizes guidance on clinical feasibility studies of diabetic glycemic control devices
The recommendations come amid an increase in requests for feedback from developers of diabetes devices.
By Nick Paul Taylor • May 9, 2022 -
Insulet CEO Petrovic to step down on June 1 due to personal reasons; Hollingshead named sucesssor
Shacey Petrovic, who has served as CEO since 2019, is resigning in the first months of the Omnipod 5 launch, an anticipated product for the diabetes tech space. Jim Hollingshead, an Insulet board member since 2019, will take over.
By Ricky Zipp • May 6, 2022 -
Ortho revenue rebounds in Q1 even amid 'tough' start, labor shortages
Some companies posted sales in the period that matched or surpassed pre-pandemic levels.
By Elise Reuter • May 5, 2022 -

 Retrieved from The White House.
Retrieved from The White House.
Biden should send 'clear' message to semiconductor industry on prioritizing medtech, AdvaMed says
The trade group said it's pushing for the prioritization of medtechs, transparency for future allocations to the industry, and actions to ensure the continuity of patient care in the U.S.
By Nick Paul Taylor • May 4, 2022 -
Boston Scientific's Watchman FLX shows higher success rate than predecessor, boosting Abbott rivalry
Watchman FLX is associated with fewer major adverse events and higher rates of success than its predecessor, according to an analysis of registry data. The study builds on Boston Scientific's new rivalry with Abbott's Amulet device.
By Nick Paul Taylor • May 4, 2022 -
CDRH proposes requirement for Philips to submit recall plan for sleep-apnea, ventilator devices
The order would mean Philips would have to submit a plan to repair or replace the device or refund customers.
By Elise Reuter • May 3, 2022 -
Earnings week 2: Supply chain shortages slowed ResMed, other medtechs from meeting demand
Philips and ResMed were among several medtechs that listed supply chain shortages as a challenge in earnings calls.
By Elise Reuter • May 2, 2022 -
CDRH's Shuren expects center to return to normal this year despite ongoing COVID work
Jeff Shuren, the director of the FDA's Center for Devices and Radiological Health, also stressed the need for increased cybersecurity and supply chain funding and authority during an event on April 29.
By Ricky Zipp • May 2, 2022 -
GE Healthcare, Medtronic turn to outpatient care in new partnership
The companies agreed to a collaboration on ambulatory surgery centers and office-based labs as more procedures are shifting to outpatient care settings.
By Elise Reuter • April 29, 2022