Medical Devices: Page 5
-
JenaValve snags FDA approval in aortic regurgitation
The approval comes two months after Edwards Lifesciences scuttled plans to buy JenaValve for $945 million amid opposition from the Federal Trade Commission.
By Susan Kelly • March 18, 2026 -
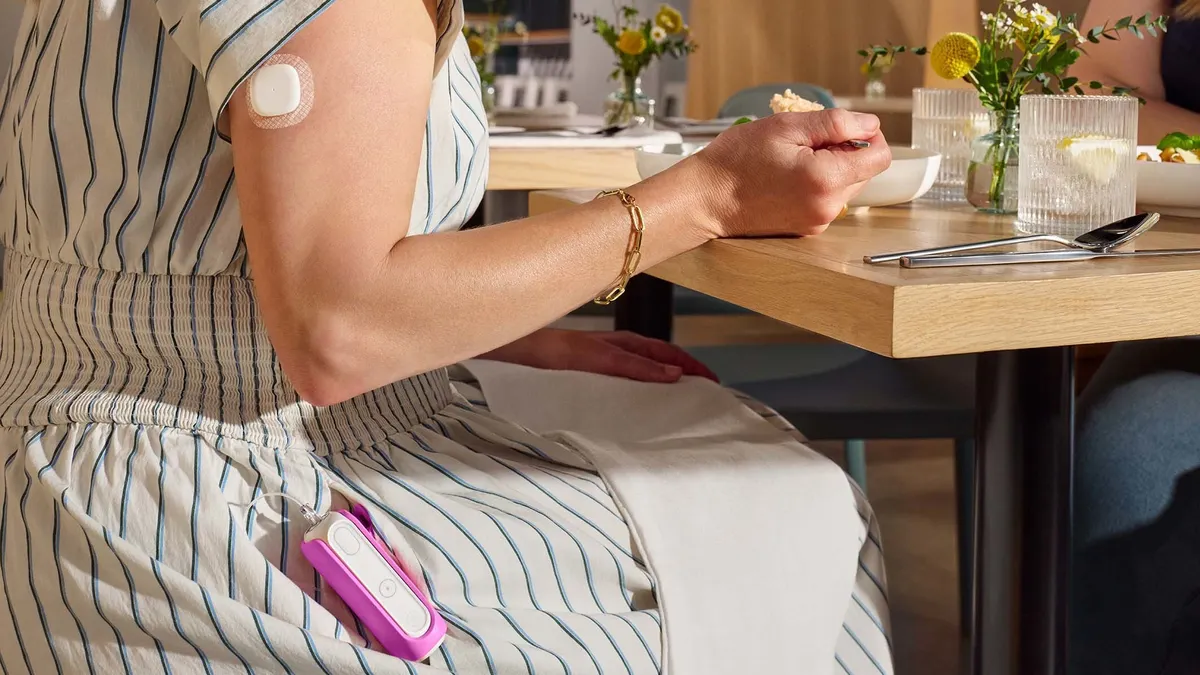
MiniMed gets FDA nod for smaller insulin pump
MiniMed Flex is a smartphone-controlled insulin pump about half the size of MiniMed’s previous device. It’s the company’s first launch since separating from Medtronic in early March.
By Elise Reuter • March 18, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
PFA startup Field Medical appoints Mark Turco as CEO
Turco, an interventional cardiologist, has held roles including CEO of JC Medical and chief medical officer of Medtronic’s aortic and peripheral vascular business.
By Nick Paul Taylor • March 18, 2026 -
Baxter CFO Joel Grade steps down
Grade is leaving after a two-year stint defined by changes to Baxter’s business, including the sale of its kidney care unit.
By Nick Paul Taylor • March 18, 2026 -
Vicarious Surgical applies for Nasdaq listing
The robotics company reduced its 2026 cash burn forecast and said its development timeline remains on track, with a system design freeze expected by year-end.
By Susan Kelly • March 16, 2026 -
EPA proposes weakening ethylene oxide emission regulations
The proposal would relax standards for facilities that use ethylene oxide, a cancer-causing gas, to sterilize medical devices.
By Elise Reuter • March 16, 2026 -
Insulet reports 18 serious adverse events with recall of Omnipod 5 insulin pumps
The company is recalling certain lots of its insulin patch pumps after finding a small tear in the internal tubing may cause insulin to leak inside the pod.
By Elise Reuter • March 13, 2026 -
Advita Ortho receives warning letter related to shoulder system
Advita Ortho was formed after orthopedic implant maker Exactech sold its assets in bankruptcy.
By Elise Reuter • March 12, 2026 -
Insulet posts clinical data on fully closed-loop insulin delivery system
The company aims to start a pivotal trial this year, file for 510(k) clearance in 2027 and launch the device in 2028.
By Nick Paul Taylor • March 12, 2026 -
Medtronic elevates Sean Haag to president of ENT
Haag, who has worked at Medtronic for 20 years, will also serve on the leadership team for the neuroscience group.
By Susan Kelly • March 11, 2026 -
Coloplast names former J&J executive Gavin Wood as CEO
Wood spent 20 years at J&J, culminating in a spell as company group chairman for the medtech business in Europe, the Middle East and Africa.
By Nick Paul Taylor • March 11, 2026 -
Vitestro raises $70M to ready blood collection robot for US launch
The company is scaling up manufacturing and commercial activities as it prepares to enter the U.S. market via the Food and Drug Administration’s de novo pathway.
By Nick Paul Taylor • March 11, 2026 -
ARPA-H launches program to develop biosensors that can track multiple signals
The research initiative is part of a broader focus on wearables at the Department of Health and Human Services.
By Elise Reuter • March 10, 2026 -
Medtronic to acquire Scientia Vascular for $550M
The Scientia deal follows Medtronic’s announcement in February that it’ll acquire CathWorks for up to $585 million.
By Susan Kelly • March 10, 2026 -
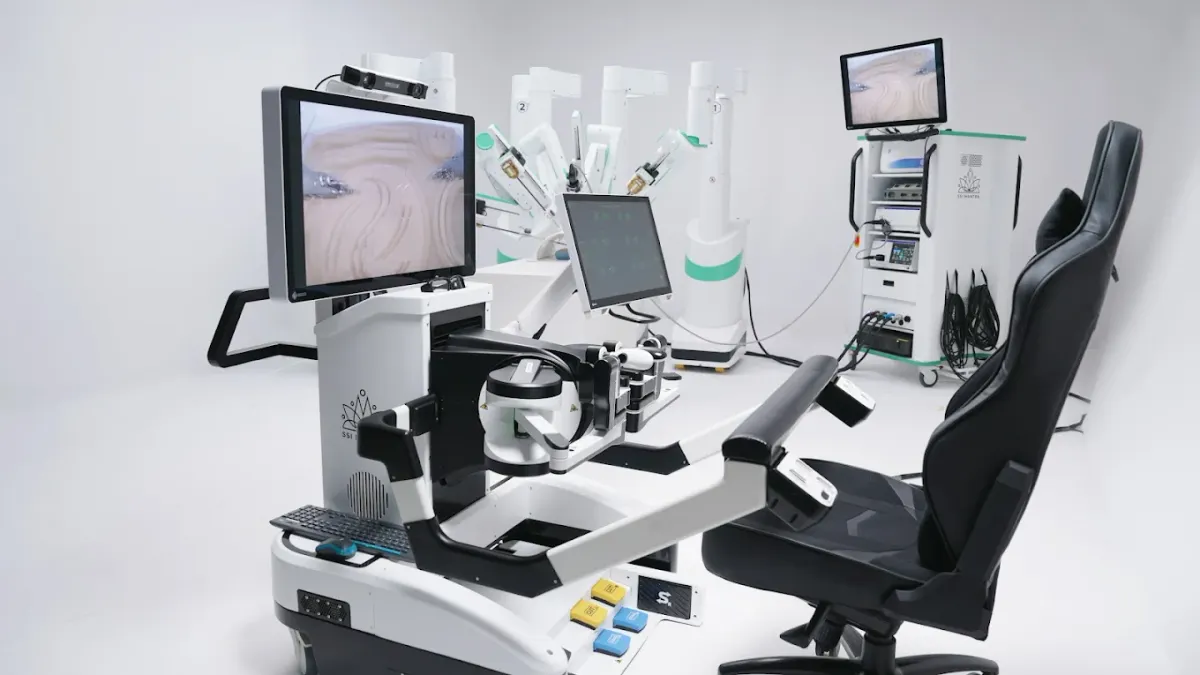
SS Innovations raises $18.6M as surgical robot nears US market
Intuitive co-founder Fred Moll invested $2 million, supporting SS Innovations’ efforts to launch its robot in the U.S.
By Nick Paul Taylor • March 10, 2026 -
Science Corporation raises $230M for Prima vision restoration device
The startup is seeking regulatory approval in Europe and the U.S. for the device, which restored vision to patients with late-stage macular degeneration in a small clinical trial.
By Susan Kelly • March 6, 2026 -
Medtronic’s MiniMed goes public for $560M
The diabetes tech business is now operating as a standalone, public company. Shares began trading on Friday.
By Elise Reuter • March 6, 2026 -
Zimmer Biomet shares smart knee data at AAOS
The orthopedic firm published data showing patients with its Persona IQ implant had lower revision and physical therapy rates than those who received traditional knee implants.
By Elise Reuter • March 6, 2026 -
Cognito raises $105M to bring Alzheimer’s treatment device to market
Expecting pivotal data this year, the company has raised a Series C round to support the launch of a noninvasive device designed to treat brain diseases using light and sound.
By Nick Paul Taylor • March 6, 2026 -
Trump tariffs
Tariff refunds: Court provides first step with liquidation order
The Court of International Trade on Wednesday directed Customs and Border Protection to remove defunct tariffs when finalizing non-liquidated entries.
By Phil Neuffer , Antone Gonsalves • March 5, 2026 -
Trump tariffs
US to hike global tariff to 15% ‘sometime this week,’ Bessent says
The Treasury Secretary also said he expects tariff rates struck down by the Supreme Court to return within the next five months, following ongoing trade investigations.
By Phil Neuffer • March 5, 2026 -
Medtech industry calls for FDA staffing transparency in MDUFA talks
Stakeholders also discussed changing international user fees and the FDA’s spending trigger for the program.
By Elise Reuter • March 5, 2026 -
Vicarious Surgical receives NYSE delisting notice
The robotics company, which has taken steps to preserve cash amid the latest change of course in the development of its surgical system, got approval to move its shares to the over-the-counter market.
By Susan Kelly • March 4, 2026 -
Stryker execs discuss Mako RPS launch at AAOS
Keith Evans, general manager of Stryker’s Mako and enabling technologies unit, said the handheld robot will be in a limited release for the first half of 2026.
By Elise Reuter • March 4, 2026 -
Profile
Inside CMR Surgical’s big pivot before US robot launch
CEO Massimiliano Colella delayed introducing the company’s soft tissue surgery system in the U.S. by a year but says the improved robot that is hitting the market now was worth the wait.
By Susan Kelly • March 4, 2026