Medical Devices: Page 66
-
Orthofix fires CEO, CFO after investigation finds misconduct
The executives engaged in “repeated inappropriate and offensive conduct” inconsistent with the company’s values, Orthofix said.
By Susan Kelly • Sept. 13, 2023 -
FDA finalizes combination product guidance 7 years after sharing draft
AdvaMed called overlapping human factor requirements of the draft guidance “overly burdensome.”
By Nick Paul Taylor • Sept. 13, 2023 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
TB deaths prompt FDA warning on reducing transmission risk
The agency outlined risk mitigation strategies for cell and tissue products after two recipients of bone matrix products died.
By Nick Paul Taylor • Sept. 13, 2023 -
CMS digs deeper into impact of proposed breakthrough device pathway in JAMA paper
The article discusses what the TCET pathway will mean for products at different stages of the life cycle.
By Nick Paul Taylor • Sept. 12, 2023 -
Bausch + Lomb seeks funding for $1.75B eye drop acquisition from Novartis
Fitch Ratings called the deal to buy the Xiidra treatment “strategically sound” even as the company’s plan to fund it would increase its leverage.
By Nick Paul Taylor • Sept. 12, 2023 -
Centinel Spine to sell fusion business, focus on disc replacement
Switzerland’s Silony Medical, which is buying the technology, sees itself as a challenger to “big medtech” in spinal fusion.
By Susan Kelly • Sept. 11, 2023 -
PFA procedures expected to rise after Boston Scientific study results
Analysts estimate the technology could be used in up to half of all cardiac ablation cases in a few years.
By Elise Reuter • Sept. 8, 2023 -
Q&A
Intuitive’s Tony Jarc on how AI is improving robotic surgery
The company aims to help surgeons achieve better results for patients and hospitals.
By Susan Kelly • Sept. 8, 2023 -
Sterigenics’ $408M ethylene oxide settlement removes ‘most problematic’ lawsuits: analysts
Remaining lawsuits pose less of a threat to the medical device sterilizer, and tighter emissions standards seem achievable, KeyBanc analysts wrote.
By Nick Paul Taylor • Sept. 8, 2023 -
Bayer faces Essure lawsuit after UK court clears 200 women to claim for damages
Bayer has vowed to defend itself “vigorously” against claims that could cost the company more than $12 million in damages.
By Nick Paul Taylor • Sept. 8, 2023 -
Zimmer Biomet promotes 3 executives
The appointments come less than a month after the company elevated Ivan Tornos to the CEO position.
By Susan Kelly • Sept. 7, 2023 -
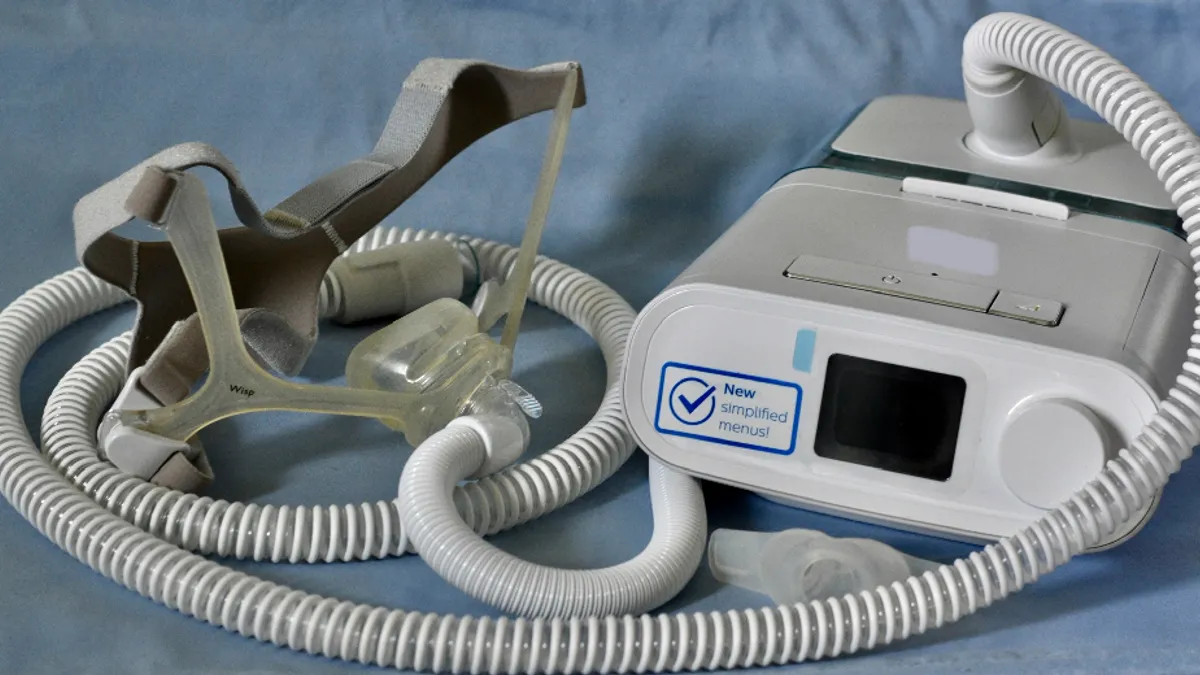
Philips reaches settlement in US respiratory device litigation
The company will pay more than $479 million to resolve all economic loss claims. The settlement does not include injury or medical monitoring claims.
By Elise Reuter • Sept. 7, 2023 -
FDA proposes 3 guidances to improve 510(k) clearance process
The agency has made recommendations for selecting predicate devices, using clinical data and conducting performance testing for implants.
By Nick Paul Taylor • Sept. 7, 2023 -
Boston Scientific gets FDA nod for latest device in fast-growing Watchman franchise
Analysts at RBC Capital Markets called the Watchman FLX Pro approval an “incremental positive” for Boston Scientific.
By Nick Paul Taylor • Sept. 7, 2023 -
Synchron brain-computer interface implanted in first 6 US patients
The device is intended to give people with severe paralysis the ability to use their thoughts to perform everyday functions, such as online communications, hands-free.
By Susan Kelly • Sept. 6, 2023 -
Abbott to buy smart insulin pen cap maker Bigfoot
The planned acquisition could shift patients interested in smart pens to Abbott’s Libre CGMs, an analyst wrote.
By Elise Reuter • Sept. 6, 2023 -
Illumina names Agilent’s Jacob Thaysen as its next CEO
The appointment comes less than three months after former CEO Francis deSouza stepped down following a proxy battle.
By Susan Kelly • Sept. 5, 2023 -
Medtronic sued for allegedly sharing ‘treasure trove’ of diabetes patient data with Google
The plaintiff said disclosures to Google are “particularly problematic” because his use of Gmail meant his personal information was “automatically linked to his real identity.”
By Nick Paul Taylor • Sept. 5, 2023 -
Noah Medical lung biopsy robot exceeds expectations in first-in-human trial
The startup presented early data on its FDA-cleared system at a meeting of pulmonologists in Chicago.
By Susan Kelly • Sept. 1, 2023 -
Spine volumes to grow 6.5% next year as post-pandemic boost continues: analysts
Use of robotic systems in spine surgeries also is on the rise, according to the latest poll by BTIG.
By Nick Paul Taylor • Sept. 1, 2023 -
Q&A
ZimVie’s Rebecca Whitney sizes up the competition in cervical disc implants
The company is bringing new products to market as it faces more challengers in cervical disc replacement and tethering for scoliosis.
By Elise Reuter • Sept. 1, 2023 -
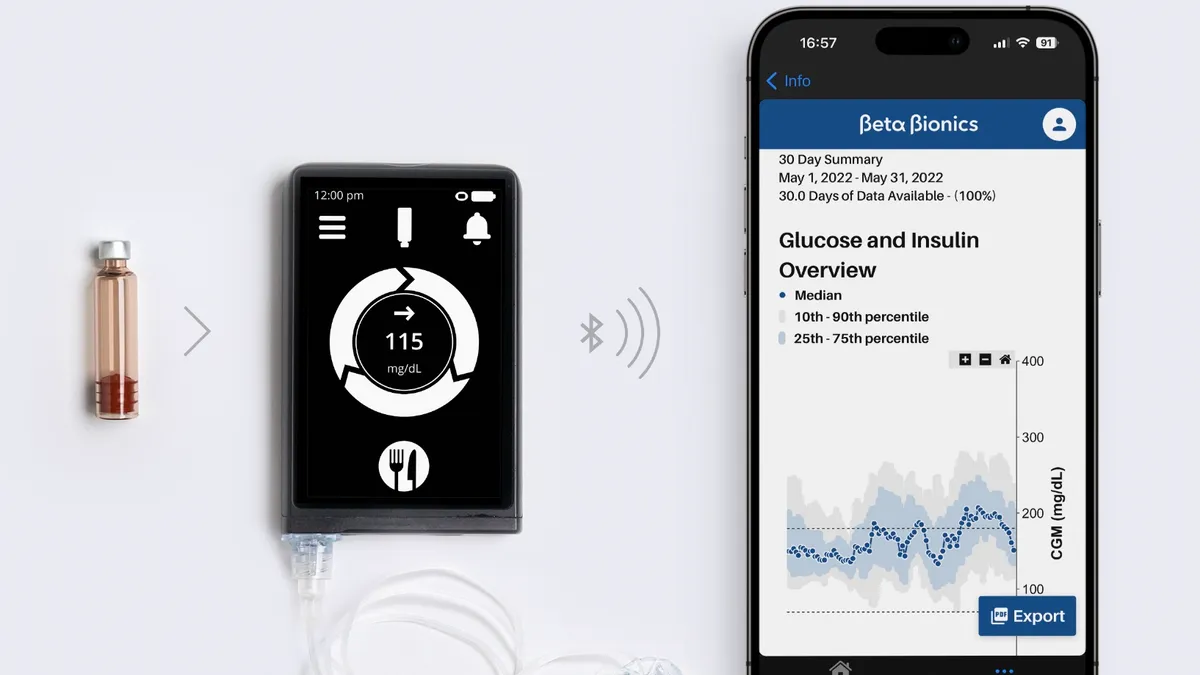
Beta Bionics raises $100M to challenge Medtronic, Tandem for automated insulin dosing market
The company’s iLet Bionic Pancreas automates insulin dosing without requiring mealtime carb counting.
By Nick Paul Taylor • Sept. 1, 2023 -
Abbott study shows benefits of OCT imaging in stent placement
Optical coherence tomography improved doctors’ ability to place stents, but the study did not meet its primary goal for reducing a composite of deaths, heart attacks and repeat procedures.
By Susan Kelly • Aug. 31, 2023 -
UK regulators name 3 approved bodies to ease device certification bottleneck
A MHRA leader hailed the action as “almost doubling capacity for medical device assessment in the U.K.”
By Nick Paul Taylor • Aug. 31, 2023 -
Hamilton Medical’s urgent ventilator notice deemed Class I recall by FDA
The company contacted customers in June after learning that long-term use of the devices could cause them to stop providing active ventilation.
By Nick Paul Taylor • Aug. 31, 2023