FDA
-
Philips secures FDA clearance for AI-enabled CT system
The company aims to increase its share of the premium CT device market through 2028.
By Nick Paul Taylor • April 17, 2026 -
FDA, medtech industry near MDUFA VI agreement
The FDA said it intends to hire “substantial numbers” of medical device review staff as part of the agreement for the next medical device user fee amendments.
By Elise Reuter • April 9, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Courtesy of Intuitive Surgical
Courtesy of Intuitive Surgical Trendline
TrendlineTop 5 stories from MedTech Dive
From the top medtech trends to watch in 2026 to haphazard layoffs at the Food and Drug Administration and the evolving use of AI in the medtech sector, here is a collection of top stories from MedTech Dive.
By MedTech Dive staff -
CDRH targets hospital readmissions with home device innovation challenge
The center plans to pick nine devices to help support patients and caregivers after a hospital stay.
By Elise Reuter • April 9, 2026 -
UK steps up push to align medtech regulations with the US
Health regulators on both sides of the Atlantic are assessing alignment opportunities and exploring mutual recognition mechanisms.
By Nick Paul Taylor • April 7, 2026 -
VDyne secures FDA nod to start pivotal trial for tricuspid valve
With approval to study the transcatheter replacement device in patients with severe tricuspid regurgitation, VDyne is advancing a treatment to compete with Edwards Lifesciences’ Evoque system.
By Susan Kelly • April 6, 2026 -
Deep Dive
‘Cracks show’ as CDRH staff contend with heavy workloads
Former device leaders said a culture of “fear and anxiety” has driven experienced staff from the Food and Drug Administration. An overburdened workforce has affected communications from the agency.
By Elise Reuter , Shaun Lucas • April 1, 2026 -
Distalmotion targets ASC robotic gynecology programs with FDA filing
The company is seeking clearance in additional gynecological indications to support outpatient use of its surgical robot.
By Nick Paul Taylor • April 1, 2026 -
Medtronic wins FDA clearance for robot in cranial, ENT surgeries
The surgical navigation and robotics system is part of the company’s push to expand its share of the $15 billion cranial and spinal technologies market.
By Nick Paul Taylor • March 30, 2026 -
Philips wins FDA clearance for imaging device developed with Edwards
The system uses AI to optimize the placement of Edwards’ mitral valve repair device.
By Nick Paul Taylor • March 27, 2026 -
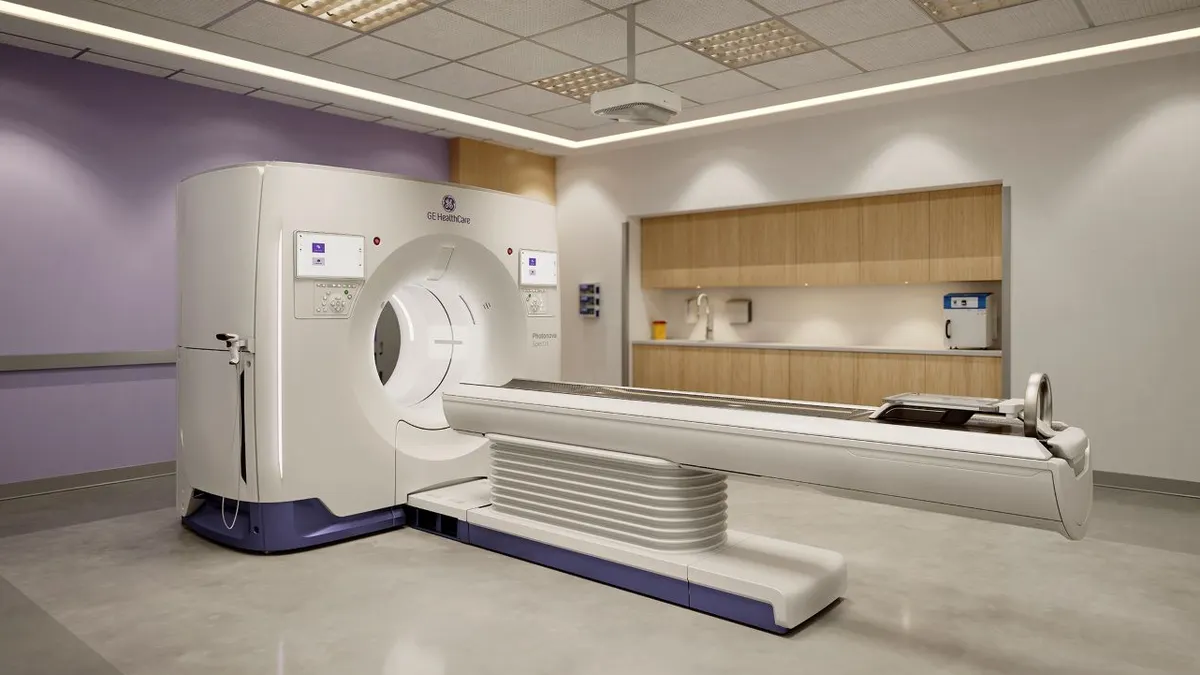
GE HealthCare wins FDA nod for photon-counting CT technology
The 510(k) decision clears the company to challenge Siemens Healthineers for an emerging part of the CT imaging sector.
By Nick Paul Taylor • March 24, 2026 -
Intuitive Surgical recalls stapler reloads over issue linked to patient death
The recall follows reports of incomplete staple lines in procedures using the company’s da Vinci surgical robots.
By Nick Paul Taylor • March 19, 2026 -
JenaValve snags FDA approval in aortic regurgitation
The approval comes two months after Edwards Lifesciences scuttled plans to buy JenaValve for $945 million amid opposition from the Federal Trade Commission.
By Susan Kelly • March 18, 2026 -
FDA to unify agency’s ‘fragmented’ safety surveillance system
Combining the disparate databases used to detect potential issues with drugs, vaccines and other products into a single dashboard will enhance their utility and cut costs, the agency said.
By Kristin Jensen • March 12, 2026 -
Advita Ortho receives warning letter related to shoulder system
Advita Ortho was formed after orthopedic implant maker Exactech sold its assets in bankruptcy.
By Elise Reuter • March 12, 2026 -
Medtech industry calls for FDA staffing transparency in MDUFA talks
Stakeholders also discussed changing international user fees and the FDA’s spending trigger for the program.
By Elise Reuter • March 5, 2026 -
Abbott wins FDA approval for updated heart failure monitoring device
The company redesigned the reader to fit in a carry-on suitcase and be easier to use day to day.
By Nick Paul Taylor • March 2, 2026 -
FDA warns Beta Bionics on unreported complaints, insulin pump changes
The Food and Drug Administration warning letter detailed unreported low blood sugar and a software change to correct for delayed glucose readings.
By Elise Reuter • Feb. 26, 2026 -
FDA posts early alert for safety issue with Impella heart pump devices
The cassettes have an increased risk of purge leaks that can lead to the loss of hemodynamic support.
By Nick Paul Taylor • Feb. 26, 2026 -
Medline addresses bed fire risk linked to death
The FDA said hand pendants and cords may overheat, causing the devices to spark, burn, melt, smoke and catch fire.
By Nick Paul Taylor • Feb. 18, 2026 -
HHS elevates officials into Kennedy’s inner circle in advance of midterms
The HHS secretary has four new senior advisors to help him “move faster and go further” on his Make America Healthy Again agenda. The appointments preempt the GOP’s midterm messaging push.
By Rebecca Pifer Parduhn • Feb. 13, 2026 -
Novocure wins FDA approval to treat pancreatic cancer with electric fields
Winning approval to target the 15,000 U.S. patients with locally advanced pancreatic cancer is the first step in a broader expansion in the tumor type.
By Nick Paul Taylor • Feb. 13, 2026 -
FDA breakthrough program starts FY2026 at steady pace
Orthopedics was the most active area over the second half of 2025, with the FDA issuing 13 breakthrough designations to devices in the therapeutic area.
By Nick Paul Taylor • Feb. 9, 2026 -
J&J recalls coil systems used in aneurysm treatment
Healthcare providers are advised to stop using the devices after a failure to detach was linked with four serious injuries and one death.
By Susan Kelly • Feb. 6, 2026 -
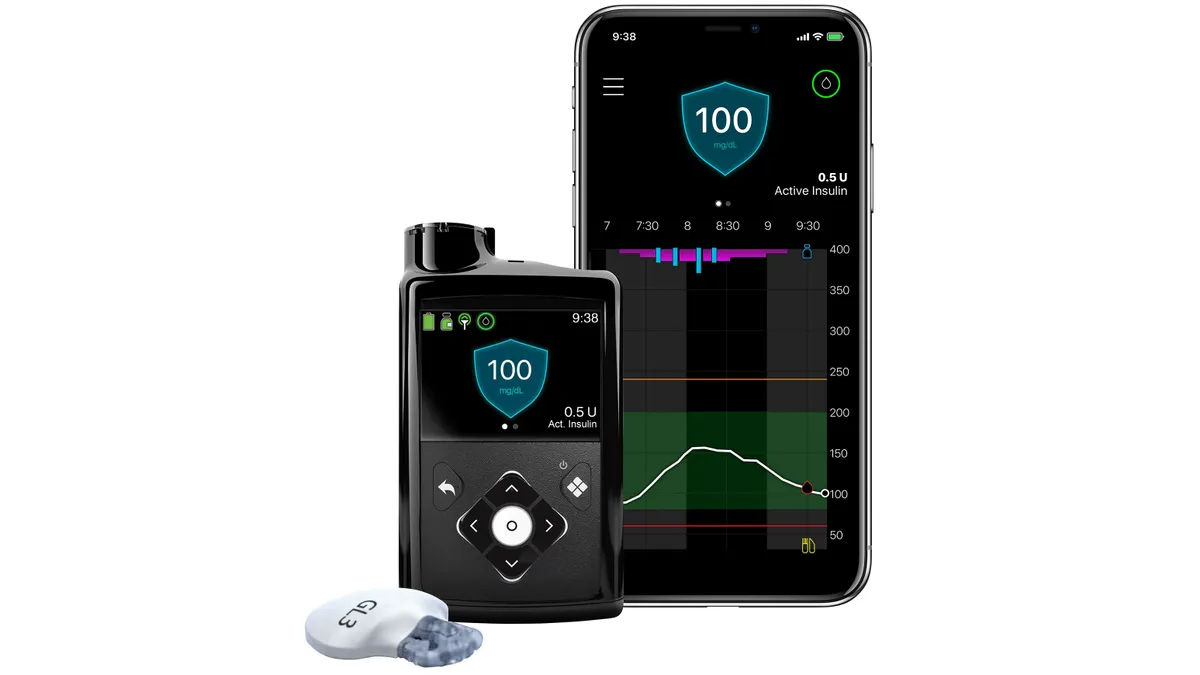
Medtronic wins expanded FDA label for 780G insulin pump
The 510(k) decision clears the device for use with ultra-rapid-acting insulins from Eli Lilly and Novo Nordisk.
By Nick Paul Taylor • Feb. 5, 2026 -
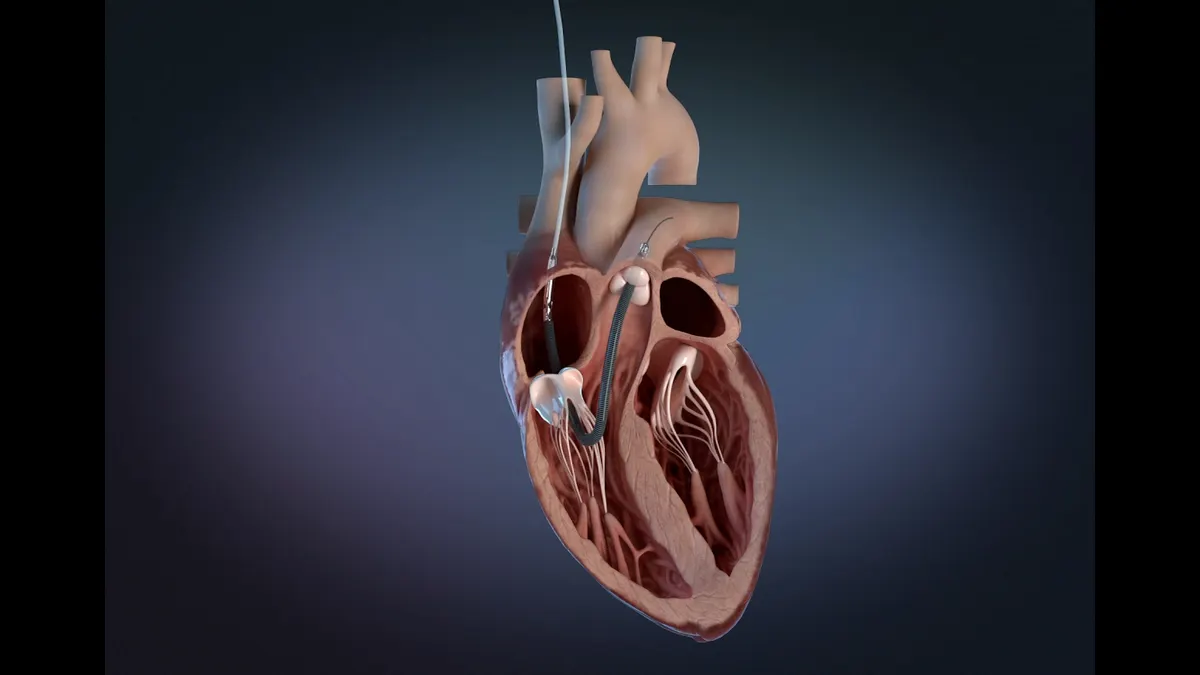
J&J corrects Impella heart pumps over issue linked to 22 serious injuries
The urgent correction notice is the latest in a series of actions related to the safety of Impella devices.
By Nick Paul Taylor • Feb. 4, 2026