FDA: Page 2
-
Grail files for FDA approval of multi-cancer early detection test
Premarket approval could help the company secure coverage from commercial payers and Medicare.
By Nick Paul Taylor • Feb. 2, 2026 -
Top medtech trends to watch in 2026
From M&A to surgical robotics and user fee negotiations, the medical device industry has a busy year ahead. Check out MedTech Dive’s roundup of the top medtech trends to watch in 2026.
By Ricky Zipp • Jan. 29, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Courtesy of Intuitive Surgical
Courtesy of Intuitive Surgical Trendline
TrendlineTop 5 stories from MedTech Dive
From the top medtech trends to watch in 2026 to haphazard layoffs at the Food and Drug Administration and the evolving use of AI in the medtech sector, here is a collection of top stories from MedTech Dive.
By MedTech Dive staff -
CDRH on track for review timelines despite staff cuts
The Center for Devices and Radiological Health said it is on track to meet review times specified by user fee agreements, despite “resource challenges.”
By Elise Reuter • Jan. 29, 2026 -
FDA guidance eases wearables oversight. But experts have questions about what’s next.
Experts say two new guidances — issued without the usual public comment period — leave questions about how patients will navigate a growing pool of wearables.
By Elise Reuter • Jan. 27, 2026 -
Integra recalls wound and burn devices amid reports of serious injuries
Packaging failures could lead to breaches in the sterile barrier and patient infections.
By Nick Paul Taylor • Jan. 23, 2026 -
AdvaMed leaders on tariffs, MDUFA and wearables
AdvaMed CEO Scott Whitaker and Mick Farrell, new board chair of the trade group, weighed in on important issues for the medtech industry one year into the Trump administration.
By Elise Reuter • Jan. 22, 2026 -
Boston Scientific recalls stent over issue linked to 3 deaths
The company said the deaths were associated with off-label or investigational use of the device.
By Nick Paul Taylor • Jan. 20, 2026 -
Deep Dive
5 topics to watch as MDUFA negotiations restart
The next Medical Device User Fee Amendments, which will set how much the FDA can raise from the industry in fees over a five-year period, are expected to go to Congress this year.
By Elise Reuter • Jan. 14, 2026 -
FDA exempts more wearable, AI features from oversight
In a pair of guidance documents released Tuesday, the regulator clarified the types of wellness features and clinical decision support tools that don’t fall under medical device oversight.
By Elise Reuter • Jan. 8, 2026 -
Deep Dive
4 medtech topics to watch in 2026
From insurance coverage questions to M&A and tariffs, here are the top storylines to watch in the medical device space in 2026.
By Ricky Zipp • Jan. 8, 2026 -
Stereotaxis wins FDA approval for robotically navigated ablation catheter
Stereotaxis’ CEO said the company has been “hampered clinically, commercially and strategically” by its prior dependence on a J&J catheter.
By Nick Paul Taylor • Jan. 7, 2026 -
Edwards receives FDA approval for mitral valve replacement system
The Sapien M3 device is the first approved mitral regurgitation treatment to use a transseptal approach, Edwards said.
By Elise Reuter • Dec. 23, 2025 -
FDA posts Class I recall notice about Medtronic heart vent catheters
The company received three complaints about patients who had perforation injuries linked to the devices.
By Nick Paul Taylor • Dec. 23, 2025 -
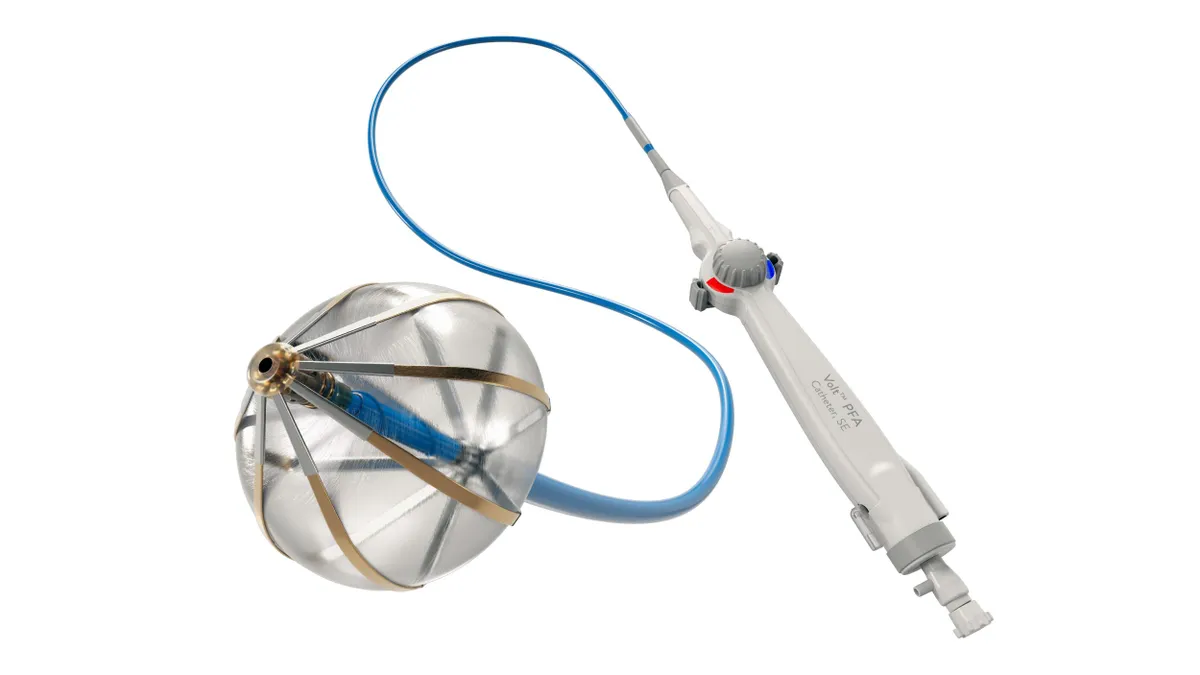
Abbott receives FDA approval for Volt PFA system
Abbott can now compete with Medtronic, Boston Scientific and J&J in the U.S. for the fast-growing pulsed field ablation market.
By Elise Reuter • Dec. 22, 2025 -
Abbott receives clearance for heart delivery device in premature babies
Physician feedback has informed a delivery device designed to make procedures safer and easier.
By Nick Paul Taylor • Dec. 22, 2025 -
FDA gets mixed feedback on performance monitoring for AI
Medtech industry groups said the FDA should use existing regulatory and quality tools to monitor performance, while medical groups said device manufacturers should be responsible for monitoring AI.
By Elise Reuter • Dec. 16, 2025 -
FDA needs more staff, authority to oversee device recalls, watchdog finds
“FDA's challenges — such as insufficient staffing — can create inefficiencies in the process and potentially put lives at risk,” the U.S. Government Accountability Office said.
By Elise Reuter • Dec. 15, 2025 -
FDA pilots allowing digital health devices access to CMS payment program
Companies can ask the FDA to waive premarket authorization and investigational device requirements while they collect real-world data in a CMS program.
By Nick Paul Taylor • Dec. 10, 2025 -
Deep Dive
What medtech firms can learn from Whoop’s warning letter
An FDA warning letter about the wearable company’s blood pressure feature raises questions about the blurring lines between medtech and wellness.
By Elise Reuter • Dec. 8, 2025 -
FDA advisory panel votes unanimously against J&J heart shunt
Panelists focused on study data that showed the implant did not improve outcomes in heart failure patients.
By Susan Kelly • Dec. 5, 2025 -
Olympus reinforces advice over device issue linked to 113 serious injuries
The company received complaints that the ligation loop can become unintentionally anchored in place.
By Nick Paul Taylor • Dec. 4, 2025 -
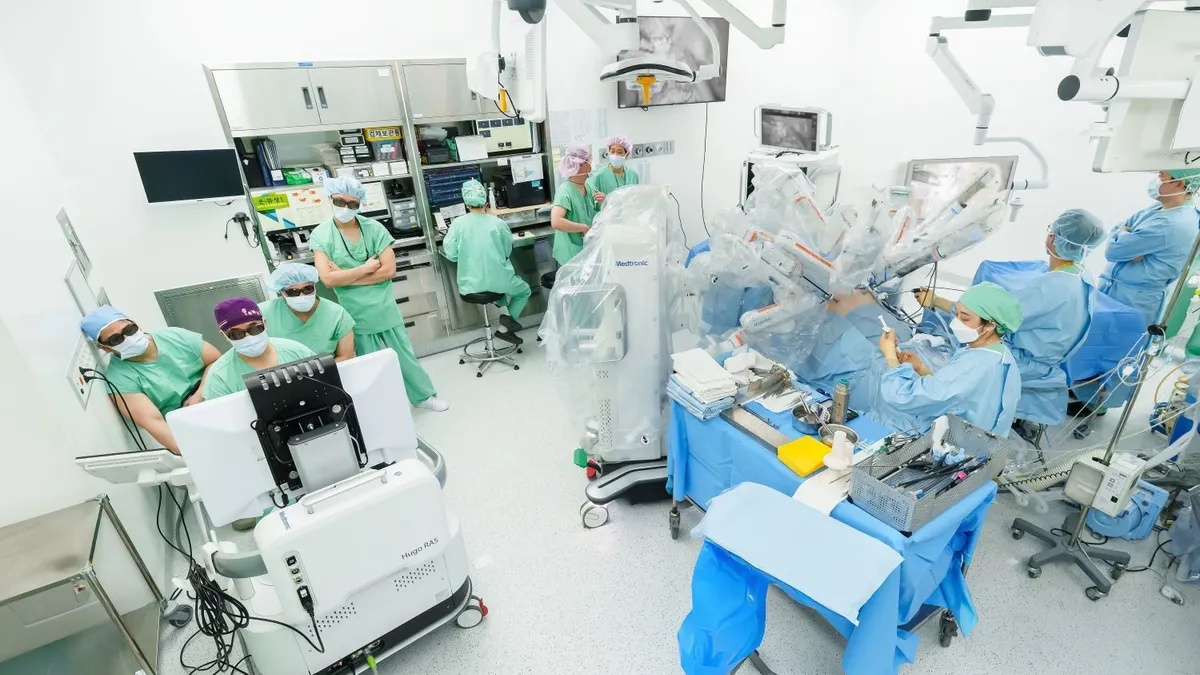
Medtronic’s Hugo surgical robot earns FDA clearance
With the U.S. authorization for urologic procedures, “there is now choice” for hospitals that want to expand their robotic programs, a company executive said.
By Susan Kelly • Dec. 4, 2025 -
Richard Pazdur, FDA drug czar, to retire from agency
Weeks after being named head of CDER, Pazdur has submitted paperwork to step down at the end of the month, exacerbating what’s already been a turbulent year at the FDA.
By Ben Fidler • Dec. 2, 2025 -
Cleveland Diagnostics wins FDA nod for prostate cancer test
The IsoPSA test is intended to help assess whether patients with elevated PSA levels should go on to receive a biopsy procedure.
By Susan Kelly • Dec. 2, 2025 -
FDA flags risk from dropped BD Alaris pumps after 2 injury reports
BD told customers to immediately remove from use any devices they drop or severely jar.
By Nick Paul Taylor • Dec. 2, 2025