FDA: Page 62
-
FDA draft guidance expands electromagnetic compatibility advice for medtechs
A new document builds on a very brief final guidance from 2016, as the agency looks to gather the right information on how devices function in intended electromagnetic environments.
By Nick Paul Taylor • Nov. 17, 2020 -
EU regulators provide 7 rules for classifying diagnostics under IVDR
The European Commission's Medical Device Coordination Group placed IVDs into different risk categories based on how they will be used. Only one class can be self-certified.
By Nick Paul Taylor • Nov. 16, 2020 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -

 "State Public Health Laboratory in Exton Tests for COVID-19" by Governor Tom Wolf is licensed under CC BY 2.0
"State Public Health Laboratory in Exton Tests for COVID-19" by Governor Tom Wolf is licensed under CC BY 2.0
Labs face COVID-19 surges, warn of test result delays as CMS pay cuts loom
The American Clinical Laboratory Association sounded an alarm over nationwide capacity constraints, which may increase average time to results.
By Maria Rachal • Nov. 13, 2020 -
BD's Alaris infusion pumps flagged for cybersecurity vulnerability
The Department of Homeland Security alert scored the issue 6.5 out of 10 and said a successful attack that exploited the weakness could force operators to manually program the pumps.
By Nick Paul Taylor • Nov. 13, 2020 -
3 ways Biden's COVID-19 approach could impact medtech
A new administration — regardless of who's in power — means device makers can anticipate shifts at key government agencies like the FDA.
By Maria Rachal , Greg Slabodkin • Nov. 12, 2020 -
Qiagen seeks EUA for COVID-19 antigen test aiming for positive results in 2 minutes
The company claims 90% sensitivity, whereas other antigen tests sold by Abbott, Roche and Quidel have reported sensitivities of around 97%.
By Nick Paul Taylor • Nov. 12, 2020 -
Remote trials, US-Israel device partnership among pitches in FDA, HHS funding bills
The Senate appropriations committee released a series of budget proposals for fiscal 2021. FDA's device center is slated for a 5% funding increase, making it one of the only centers set to receive less than requested.
By Nick Paul Taylor • Nov. 11, 2020 -
UK fleshes out post-Brexit approach to regional medical device marking
The Medicines and Healthcare products Regulatory Agency further clarified conformity marking as medtechs prepare for the transition period to end.
By Nick Paul Taylor • Nov. 11, 2020 -
Top picks for who could run HHS, CMS under Biden
Former Obama adviser and ACA architect Ezekiel Emanuel as well as high-profile state officials like New Mexico Governor Michelle Lujan Grisham are among those considered likely advisers.
By Rebecca Pifer Parduhn • Nov. 10, 2020 -

 "White House Press Briefing". Retrieved from The White House.
"White House Press Briefing". Retrieved from The White House.
Abbott COVID-19 tests at center of squabble between Trump administration, states
HHS testing czar Brett Giroir on Monday said the government is halting the antigen test shipments to eight states until those already sent are distributed and used. At least one state pushed back on his assertions.
By Greg Slabodkin • Nov. 10, 2020 -
FDA greenlights digital therapeutic for Apple Watch to quell PTSD nightmares
The device, from Minneapolis-based startup Nightware, monitors heart rate and body movement data to best deliver vibrations that interrupt a nightmare without waking the person sleeping.
By Susan Kelly • Nov. 9, 2020 -

 "200323-Z-IB607-0016" by New Jersey National Guard is licensed under CC BY-ND 2.0
"200323-Z-IB607-0016" by New Jersey National Guard is licensed under CC BY-ND 2.0
GenScript wins 1st FDA nod to test for antibodies that could neutralize COVID-19
The Hong Kong-listed biotech's offering differs from earlier EUAs for serology tests from Abbott, Roche and Siemens Healthineers that only screen for antibodies that do not necessarily cut viral infection.
By Susan Kelly • Nov. 9, 2020 -

 The image by Gage Skidmore is licensed under CC BY-SA 2.0
The image by Gage Skidmore is licensed under CC BY-SA 2.0
Biden transition team names COVID-19 advisers, seeks dramatic testing scale-up with federal approach
The president-elect calls for doubling drive-through test sites and increasing capacity by "orders of magnitude" through investing in rapid at-home tests. The transition team has already called in help from former FDA and BARDA heads.
By Greg Slabodkin • Nov. 9, 2020 -
Even if Biden wins, divided Congress stifles chance for more progressive health policies
Results of the election are not final and may be uncertain for weeks, but the most likely scenario points to mostly incremental change, a positive for many parts of the healthcare sector.
By Shannon Muchmore • Nov. 5, 2020 -
FDA warns of COVID-19 antigen test false positives as report flags Quidel on accuracy
The agency alert, prompted by reports from nursing homes and other settings, comes a day after data emerged raising concerns about the ability of a Quidel test to detect asymptomatic cases.
By Nick Paul Taylor • Nov. 4, 2020 -
MedPAC pans CMS idea to lean on commercial payers for Medicare coverage decisions
The advisory group warned against threats to transparency and rigor in response to a proposed rule defining the term "reasonable and necessary" and adding a faster coverage pathway for FDA-designated breakthrough devices.
By Susan Kelly • Nov. 3, 2020 -
CMS finalizes rejection of Baxter, Outset dialysis device add-on payments
The broader, now final, ESRD rule is meant to support use of equipment and supplies for at-home dialysis treatment.
By Nick Paul Taylor • Updated Nov. 3, 2020 -
Duke researchers pitch CDS tool to keep electives going during COVID-19 surges
JAMA Network Open authors found that using predictive modeling to develop a clinical decision support tool could help determine patient length of stay and use of a ventilator in procedures like knee and hip replacements.
By Ron Shinkman • Nov. 2, 2020 -
CMS competitive bidding process fails to drive expected savings, sparking rethink
While Needham analysts contend one action removes an overhang for Inogen, Invacare and ResMed, those at Jefferies see a "major negative" in the agency's determination to lower rates outlined in a proposed rule.
By Nick Paul Taylor • Oct. 29, 2020 -
Neovasc angina device fails to win FDA panel backing, stock tumbles 43%
By a vote of 17-1, experts on FDA's circulatory system devices panel said the available data did not provide reasonable assurance of effectiveness, though most thought the Reducer system was safe for patients.
By Susan Kelly • Oct. 28, 2020 -


Yujin Kim / MedTech Dive, original photo courtesy of U.S. Food and Drug Administration

MDUFA V talks kick off as FDA grapples with onslaught of COVID-19 submissions
Medtech industry groups broadly expressed a desire to maintain the status quo after FDA Commissioner Stephen Hahn described the strain on agency workers under MDUFA IV as unsustainable.
By Maria Rachal • Oct. 28, 2020 -
USPSTF proposes lowering colorectal cancer screening age in boost to Exact Sciences' Cologuard
A federal task force, which has recommended the stool-based kit, now advises screening starting at age 45 after modeling found the change would avert one additional death per 1,000 adults.
By Nick Paul Taylor • Oct. 28, 2020 -
Neovasc refractory angina treatment faces FDA panel review
The agency's ultimate approval decision on the Canadian company's CE-marked device has important financial implications for the medtech, which saw its revenue plunge and operating loss deepen in the second quarter.
By Susan Kelly • Oct. 27, 2020 -
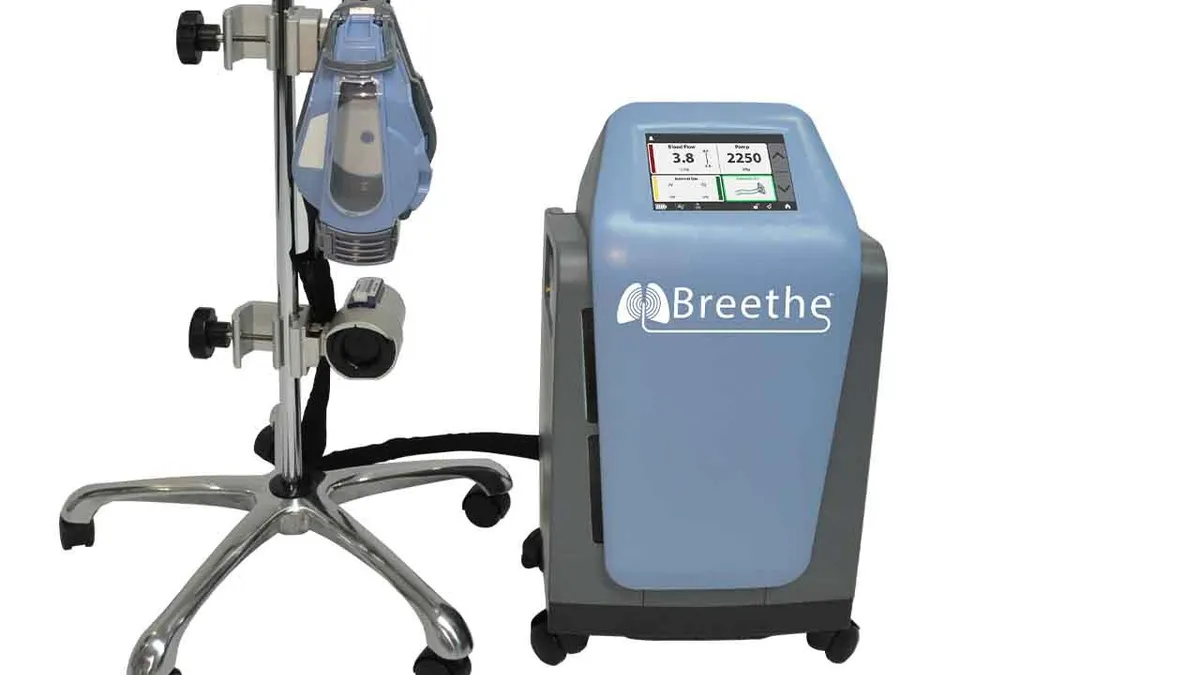
 Retrieved from Abiomed/BusinessWire on April 30, 2020
Retrieved from Abiomed/BusinessWire on April 30, 2020
FDA grants 510(k) to Abiomed's artificial lung, teeing up use in COVID-19
While LivaNova and Medtronic already sell ECMO devices, Abiomed is focusing on a relatively compact design to differentiate the device from the pack.
By Nick Paul Taylor • Oct. 27, 2020 -
FDA, Philips warn of data bias in AI, machine learning devices
The comments come 18 months after FDA unveiled a yet-to-be-finalized framework for modifying AI/ML-based software as a medical device using real-world learning and adaptation.
By Greg Slabodkin • Oct. 26, 2020