FDA: Page 61
-
Dive Awards
Regulatory Disruption of the Year: HHS lab developed test policy
The surprise move to no longer require FDA premarket review for laboratory developed tests spurred backlash across a spectrum of public health experts and industry.
By Greg Slabodkin • Dec. 9, 2020 -
Hospitals prepare to reveal prices online come Jan. 1
The rule requires hospitals to prepare a list of pricing information for 300 "shoppable services," such as total knee replacements. However, experts question if the data will benefit consumers.
By Samantha Liss • Dec. 8, 2020 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
ACA defender Becerra tapped for Biden's HHS chief
The California attorney general has voiced opposition to provider consolidation in his state, alleging anticompetitive practices by regional powerhouse Sutter Health and denying a hospital system merger.
By Shannon Muchmore • Dec. 7, 2020 -
Endologix AAA graft to go before FDA panel over potential life-threatening leak risk
The agency also released postmarketing data reinforcing an increased likelihood of blood leaking into the aneurysm for patients with the company's AFX endovascular grafts.
By Susan Kelly • Dec. 7, 2020 -

 Retrieved from Quest Diagnostics, PRNewswire on May 29, 2020
Retrieved from Quest Diagnostics, PRNewswire on May 29, 2020
Quest gets first FDA nod for at-home collection coronavirus-flu combo test
The lab giant is using Roche’s diagnostic and instrument, which received an emergency use authorization in September, with its own process for testing samples collected by consumers.
By Nick Paul Taylor • Dec. 7, 2020 -
Siemens COVID-19 test gets high marks on FDA sensitivity list, beating Abbott, BD and Roche
A PerkinElmer test still holds the top position among coronavirus diagnostics least likely to return false negatives.
By Nick Paul Taylor • Dec. 4, 2020 -
CDRH headed for 'reset' in 2021 after COVID-19 derailed priorities, Shuren says
After a "massive increase" in work due to the pandemic, the device center anticipates continuing to manage coronavirus projects while also focusing on MDUFA V and new programs.
By Ricky Zipp • Dec. 3, 2020 -
CMS adds hip replacements, other surgeries to ASC list in final rule
A representative from Stryker applauded the changes and said the rule will encourage providers to consider the shift to ambulatory surgery centers.
By Ricky Zipp • Updated Dec. 4, 2020 -
EU underscores remote notified body audit policy amid pandemic
MedTech Europe has pressed the Commission to expand such reviews to the incoming MDR and IVDR regs, but a new Q&A notes the scope is limited to the directives and devices considered "clinically necessary" during the crisis.
By Nick Paul Taylor • Dec. 3, 2020 -
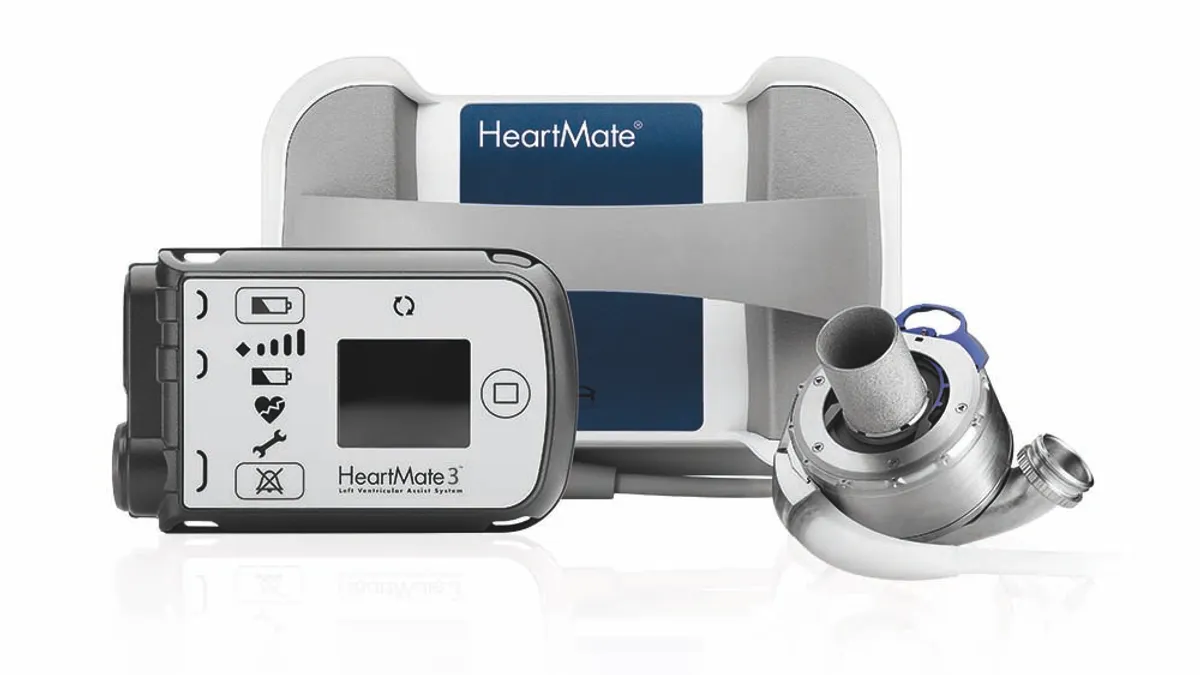
CMS finalizes expanded artificial heart, ventricular assist device coverage
Medicare will cover artificial hearts outside of clinical studies and align VAD coverage criteria with medical practice. Cowen called the decision good for Abbott and Medtronic.
By Nick Paul Taylor • Dec. 2, 2020 -
CMS makes some telehealth coverage permanent, finalizes specialty rate cuts
Areas getting pay slashed include thoracic and cardiac surgery and interventional radiology, Credit Suisse analysts said. Surgeon groups decried the changes.
By Rebecca Pifer Parduhn • Dec. 2, 2020 -
Digital therapeutic De Novos climb with FDA nod to IBS treatment
Despite a series of first-of-their-kind authorizations for such tech, total 2020 De Novo OKs currently match a 2019 low in recent years.
By Maria Rachal • Dec. 1, 2020 -
iRhythm gains UK support for EKG system while awaiting key final rule from CMS
The ambulatory cardiac monitoring system is one of just six medtech products recommended this year in guidance from the U.K. National Institute for Health and Care Excellence. The focus is on individuals with suspected arrhythmias.
By Maria Rachal • Dec. 1, 2020 -
CMS expands ability for hospital-level care at home ahead of holiday virus spread
Six health systems received waivers to treat more than 60 acute conditions under the new program, which also tweaks earlier changes allowing ambulatory surgical centers to provide greater inpatient care.
By Shannon Muchmore • Nov. 30, 2020 -
IVDR notified body count inches up to 5 with TÜV Rheinland designation
The medtech notified bodies trade group urged swift action on potentially delaying the start date for the In Vitro Diagnostic Regulation if designations don't more than double by year's end.
By Nick Paul Taylor • Updated Nov. 30, 2020 -
Breast cancer tech among FDA's latest breakthrough nods
Over the past month, designations for speedier review have also ranged from a migraine treatment to tissue regeneration tech for spinal cord injuries.
By Susan Kelly • Nov. 24, 2020 -
Medical device makers get more flexibility with value-based care in Stark, anti-kickback final rules
The changes include two safe harbor pathways and "allow you to do things that otherwise might violate the kickback statute or the Stark Law. So, that is always welcome in the industry," one lawyer said.
By Ricky Zipp • Nov. 24, 2020 -
In last-minute push, Trump admin rolls back Stark, anti-kickback rules
The final regs are slated to take effect Jan. 19, one day before President-elect Joe Biden is inaugurated. The new administration could change them but experts called that unlikely.
By Rebecca Pifer Parduhn • Nov. 23, 2020 -
NEJM commentary calls for 510(k)s for OTC hearing aids after senators pan FDA delay to rule
While critics of the delay acknowledged FDA's coronavirus-related workload this year, the agency has failed to come through with the draft regulation required by a 2017 law.
By Nick Paul Taylor • Nov. 23, 2020 -
MedTech Europe backs proposed EU Health Union but flags potential hangups
The trade group fears moving responsibility for administering the IVDR and MDR expert panels to the European Medicines Agency could slow deployment.
By Nick Paul Taylor • Nov. 20, 2020 -
Notified bodies report early success with remote audits but challenges remain
While there have been benefits to going remote during the pandemic, 57% of those polled by a European trade group say they take longer than on-site assessments and most encounter problems due to poor network connections.
By Nick Paul Taylor • Nov. 20, 2020 -
HHS piloting Cue's NBA-tested rapid COVID-19 diagnostic in 5 states
Unlike lab-based molecular tests, which can take two to three days to turn around, Cue Health's point-of-care molecular test generates results in about 20 minutes.
By Maria Rachal • Nov. 19, 2020 -

 Master Sgt. Hecht, Matt. (2020). [Photograph]. Retrieved from Flickr.
Master Sgt. Hecht, Matt. (2020). [Photograph]. Retrieved from Flickr.
Health insurers, labs point fingers over COVID-19 test coverage and prices
An AHIP study of claims data is the latest volley in a debate about who ultimately foots the bill for coronavirus diagnostics. ACLA blames "murky" tri-agency guidance for coverage denials and high out-of-pocket costs.
By Nick Paul Taylor , Greg Slabodkin • Nov. 19, 2020 -
FDA OKs first fully at-home COVID-19 test as US cases surge
An emergency use authorization to biotech Lucira Health comes as some experts say the tests could help significantly boost capacity. Others say that's a long way off, and call for more guidance from regulators on proper use.
By Greg Slabodkin • Nov. 18, 2020 -

 "White House Press Briefing". Retrieved from The White House.
"White House Press Briefing". Retrieved from The White House.
In shift, FDA ordered to provide 'timely' EUA reviews for COVID-19 lab developed tests
Testing czar Brett Giroir has reversed FDA's decision to no longer review emergency use authorization requests for LDTs so that university labs can secure EUAs and get liability protection under the PREP Act.
By Greg Slabodkin • Nov. 17, 2020