Dive Brief:
- Johnson & Johnson’s venture capital arm is leading an $85 million investment in HistoSonics ahead of the planned commercialization of its liver cancer treatment.
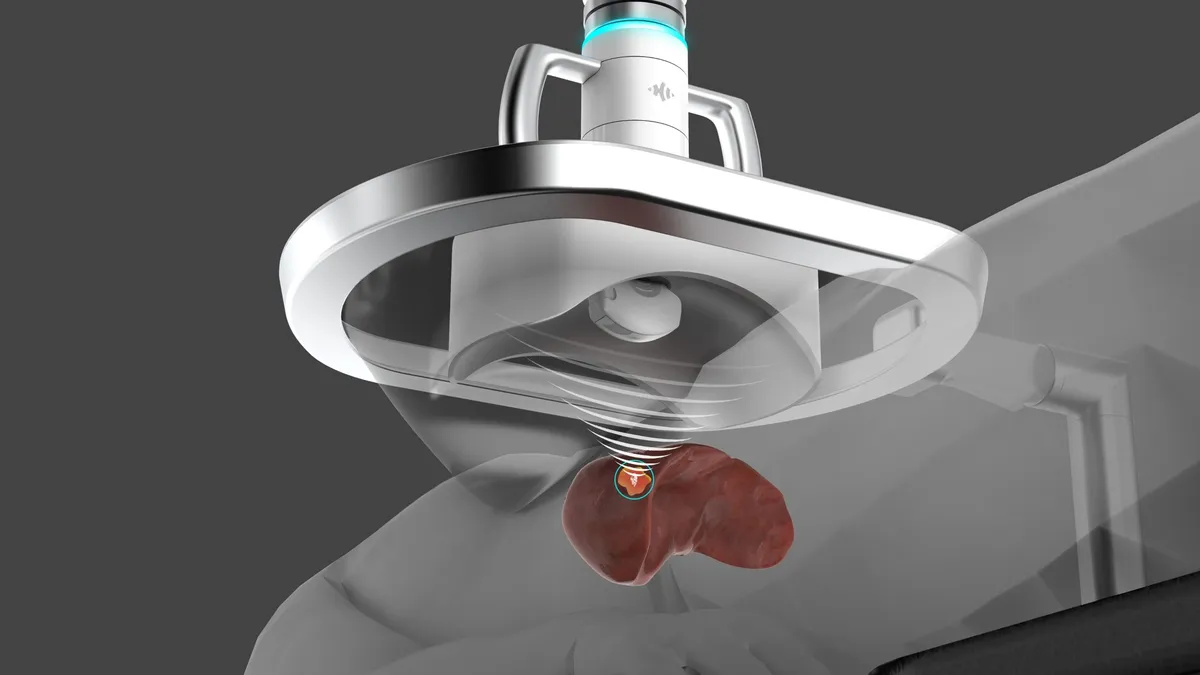
- HistoSonics is developing Edison, a device that uses imaging and sensing technology to deliver focused ultrasound to destroy and liquify unwanted tissue and tumors without the need for incisions.
- Having recently submitted clinical data to the Food and Drug Administration, HistoSonics expects to secure market authorization in the U.S. next year.
Dive Insight:
HistoSonics is built on the discovery that pulsed sound waves can induce bubble clouds in human tissue. As the clouds form and collapse in microseconds, they unleash mechanical forces that are strong enough to destroy tissue. The body then absorbs the debris.
The method, known as histotripsy, has applications in cancer, neurological diseases and cardiovascular diseases. HistoSonics initially tested its technology in patients with benign prostatic hyperplasia but later identified liver cancer as its lead indication, resulting in the initiation of U.S. and European #HOPE4LIVER trials in people with primary and metastatic liver tumors last year.
HistoSonics met the patient enrollment target required by regulators earlier this year and has sent data from the trials to the FDA. The company expects to win authorization in the U.S. via the De Novo route next year.
In anticipation of commercialization, HistoSonics has secured additional funding. Johnson & Johnson Innovation - JJDC, the strategic venture capital arm of J&J, led the $85 million round with assists from existing investors such as Venture Investors, Lumira Ventures, Yonjin Venture and the State of Wisconsin Investment Board. HistoSonics also secured a $15 million expansion of its debt facility.
The cash will support commercialization of Edison, plus additional clinical trials. As HistoSonics executes its plans, it will be able to draw on the experience of Silk Road Medical CEO Erica Rogers, who recently joined the company’s board of directors.