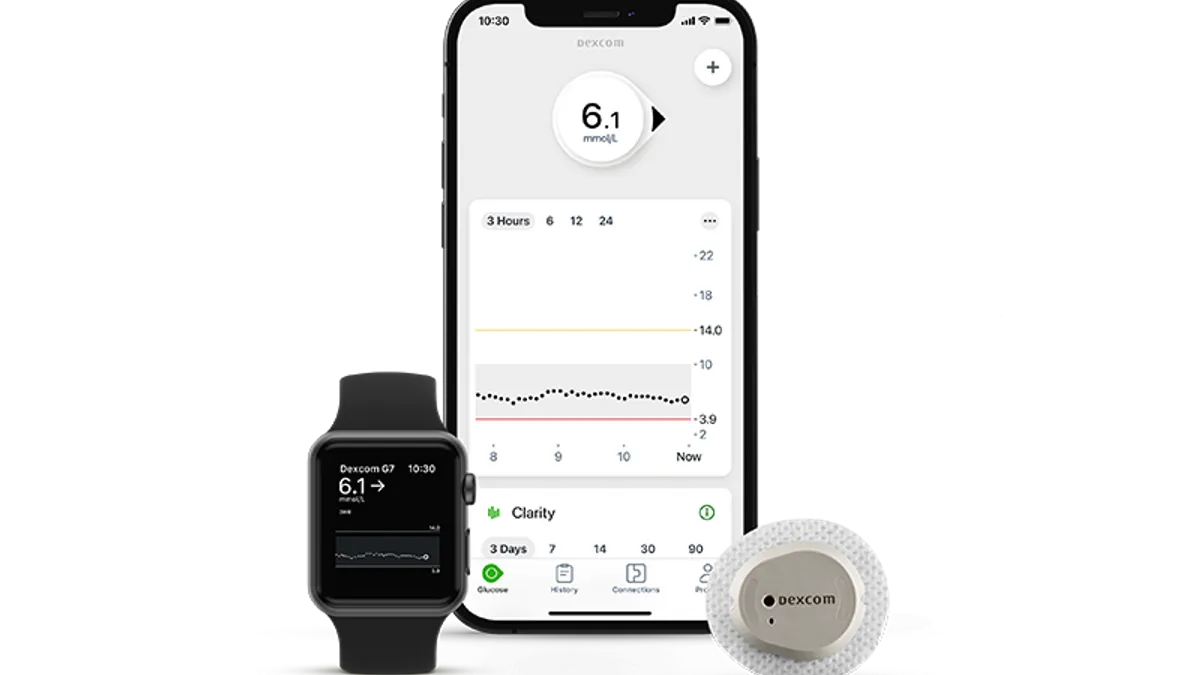
Dive Brief:
- Dexcom received clearance from the Food and Drug Administration for its G7 continuous glucose monitor, the company’s newest device that it expects will become a driver of growth.
- The CGM is Dexcom’s smallest and most accurate device, the company claims. The FDA cleared the device to be used in patients with Type 1 and Type 2 diabetes, as well as those with gestational diabetes.
- Dexcom is working on securing insurance coverage for the new device, and plans to launch it in the U.S. in 2023. The company said it will offer cash pay options as it transitions coverage to the G7.
Dive Insight:
Dexcom’s stock rose 3.8% on Thursday afternoon to $122.34 in response to the news that it could now sell its flagship CGM in the U.S. The device received a CE Mark in March, authorizing it to be marketed in Europe, but the regulatory process in the U.S. was delayed when the FDA requested a software update during its review of the device.
“We view G7 as the definitive form factor for Dexcom that we have been waiting on over a decade covering the stock, and we believe will generate new patient demand,” William Blair analyst Margaret Kaczor wrote in a research note on Thursday.
The FDA clearance for gestational diabetes came as a surprise because the device had previously been contraindicated for people with gestational diabetes, wrote Kaczor.
“Though this segment of the population was referenced as a target for DexCom at its 2020 analyst day, it was encouraging to see the broad label in advance of large clinical trial support,” she wrote.
The next step is to ensure reimbursement for patients with gestational diabetes “before any meaningful sales can occur,” J.P. Morgan analyst Robbie Marcus wrote in a research note. Gestational diabetes develops in about 5% to 10% of pregnancies.
Dexcom is working to expand coverage for the G7, but said it plans to offer cash pay options to “facilitate immediate access” to the device.
Kaczor wrote that her team expects government and private insurers to cover the G7 “relatively quickly,” with commercial insurers covering it initially as durable medical equipment and later under pharmacy coverage, a path many companies are take for diabetes devices.