Dive Brief:
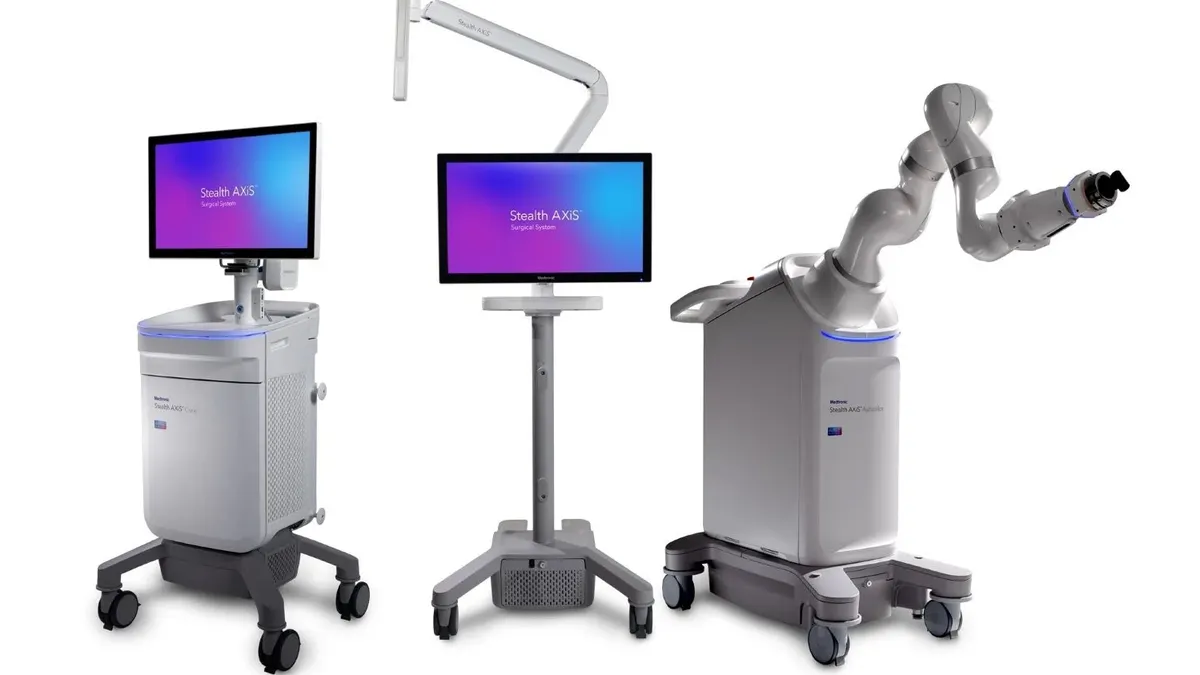
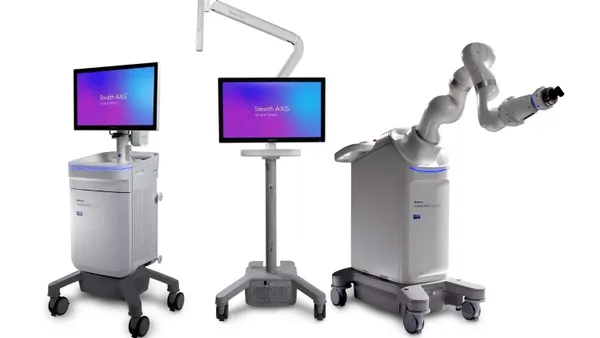
- Medtronic has received a CE mark for its Stealth AXiS surgical system in spine and cranial procedures, the company said Tuesday.
- The system, which was cleared in the U.S. earlier this year, builds on capabilities Medtronic acquired in its $1.64 billion Mazor Robotics takeover in 2018 by combining surgical planning, navigation and robotics.
- Medtronic has identified Stealth AXiS as a product that can help expand its share of the $15 billion cranial and spinal technologies market.
Dive Insight:
The Food and Drug Administration cleared Stealth AXiS for use in the U.S. in spine surgeries in February and expanded the label to cover cranial and ear, nose and throat procedures in March. GE HealthCare has integrated intraoperative ultrasound technology into the system to give physicians real-time images that could enable surgical teams to recognize and respond to mid-procedure changes in anatomy.
Surgeons can use the system to visualize anatomic motion, surgical adjustments and patient alignment in real time during spine surgery. The technology eliminates the need for repeated imaging to see changes during the procedure.
Applied to cranial procedures, the system has artificial intelligence-enabled tractography for mapping the brain. Medtronic’s goal is to give surgeons more information to make decisions before and during brain surgeries.
Early evidence that physicians are embracing the surgical system could emerge when Medtronic reports fourth-quarter results in June. CFO Thierry Piéton said on a February earnings call that the Stealth AXiS system could add to sales in the quarter.
Receiving the CE mark positions Medtronic to launch Stealth AXiS in the European Union and other parts of the world that accept the certification. International markets, including the EU, accounted for half of the $9 billion in quarterly revenues that Medtronic reported in its latest financial results.
Securing a CE mark under the EU’s Medical Device Regulation underscores Medtronic’s “commitment to bringing meaningful innovation into a highly diverse healthcare landscape,” Oli Prosperi, the company’s vice president, international commercial, cranial and spinal technologies, said in a statement.