Dive Brief:
-
A clinical trial has linked use of Tandem Diabetes Care’s artificial pancreas in children with Type 1 diabetes to significant improvements in glucose control.
-
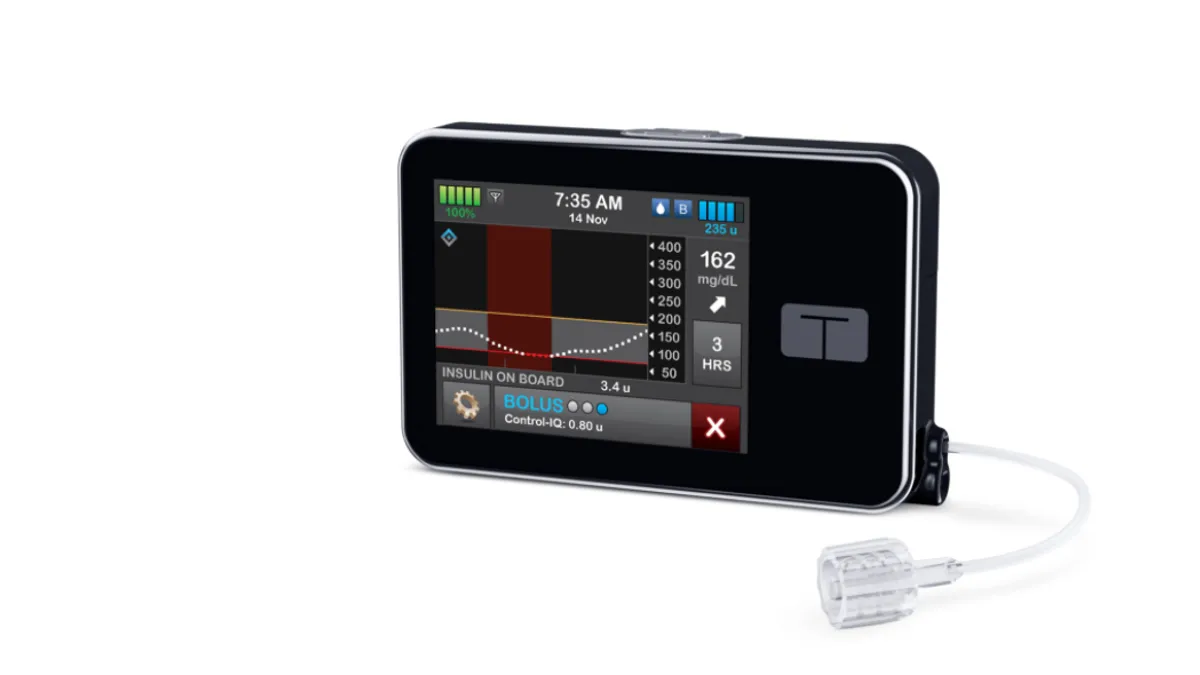
A paper partially funded by Tandem published in New England Journal of Medicine Thursday shows children treated with Tandem’s closed-loop insulin delivery system spent more time in the target glucose range than their peers who received Dexcom G6 sensors and insulin alone. The closed-loop system also used G6 sensors.
- The publication comes weeks after Tandem received FDA clearance to use its insulin dosing algorithm in conjunction with a pump in children aged 6 years and up, giving it evidence to support its targeting of the new population.
Dive Insight:
Most cases of Type 1 diabetes are diagnosed between the ages of 5 and 14. That makes the young demographic a key target market for developers of Type 1 diabetes devices such as Tandem, which became the second company after Medtronic to win FDA approval for a closed-loop system in kids aged under 14 earlier this year.
Tandem said it received the expanded label for its t:slim X2 pump with Control-IQ technology after running a clinical trial that found time in range increased to 67% in the artificial pancreas group.
The NEJM paper provides a closer look at the study. In the 78 children treated with the closed-loop system, time in range increased from 53% at baseline to an average of 67% over the 16-week trial. Time in range in the 23-subject control cohort rose from 51% to 55%. The two interventions performed comparably in terms of keeping the children out of hypoglycemia.
Children with a range of characteristics appeared to benefit from the Tandem device. Analyses of the responses of subjects of different ages, sexes, body-mass indices and household incomes found the Tandem closed-loop system consistently delivered better time in range regardless of the baseline characteristics.
The advantage of the closed-loop system was most pronounced at night-time. From midnight to 6 a.m, children with the closed-loop system were in the target range 80% of the time. That compared to 54% of the time in the control cohort.
Tandem’s device was associated with a numerically higher rate of adverse events than the control treatment — 19% versus 9% — but no children in either cohort suffered severe hypoglycemia or diabetic ketoacidosis. Cases of hyperglycemia or hyperketosis, without diabetic ketoacidosis, due to “an insulin pump problem” accounted for most of the difference between the two arms. All the cases were “related to problems with the pump infusion set, except for one in the closed-loop group that was related to an issue involving the insulin pump battery,” the researchers wrote.
Tandem now faces the challenge of leveraging the data to grow sales of its device. Late last month, Tandem reinstated its original full-year sales guidance after finding demand for its t:slim X2 insulin pump had survived the disruption caused by the COVID-19 pandemic. At that time, Tandem CEO John Sheridan said some endocrinologists had been prescribing the algorithm off-label prior to the FDA decision but the regulatory status cleared the sales team to “market openly."