Medical Devices: Page 7
-
Novocure wins FDA approval to treat pancreatic cancer with electric fields
Winning approval to target the 15,000 U.S. patients with locally advanced pancreatic cancer is the first step in a broader expansion in the tumor type.
By Nick Paul Taylor • Feb. 13, 2026 -
Neurent raises $74M to commercialize treatment for nasal condition
The device creates lesions to disrupt nerve signals that drive mucus production.
By Nick Paul Taylor • Feb. 12, 2026 -
 Explore the Trendline➔
Explore the Trendline➔
 Permission granted by Boston Scientific
Permission granted by Boston Scientific Trendline
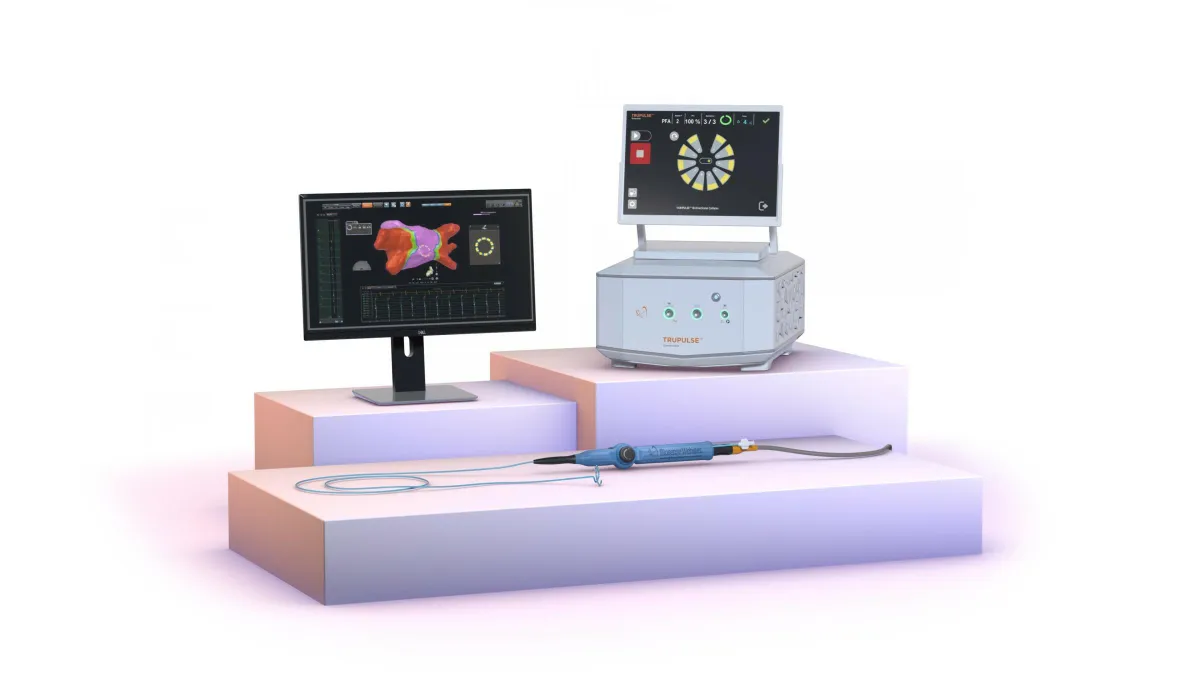
TrendlineNew medical devices are reshaping the medtech industry
From pulsed field ablation devices to glucose sensors and surgical robotics, new medical technologies are transforming patient care and how people manage their health.
By MedTech Dive staff -
AdvaMed names Melissa Torres as new leader for technology and regulatory affairs
Torres joins the medtech lobby from the FDA’s Center for Devices and Radiological Health, where she was associate director for international affairs.
By Susan Kelly • Feb. 12, 2026 -
Zimmer plans US salesforce reorganization
Zimmer Biomet expects the shift of its U.S. salesforce to a more specialized, direct approach will help it better compete in the orthopedics market.
By Elise Reuter • Feb. 11, 2026 -
Edwards focuses on earlier TAVR adoption
The heart valve specialist reported a second straight quarter of double-digit TAVR sales growth and credited recent study results with driving a “sense of urgency” for treating patients.
By Susan Kelly • Feb. 11, 2026 -
Baxter cuts roughly 90 jobs at IV solutions plant that recovered from Hurricane Helene
Hospital demand for IV solutions in the U.S. is lower than before Hurricane Helene, the company said.
By Elise Reuter • Feb. 10, 2026 -
4 takeaways from the 2026 AF Symposium
Johnson & Johnson, Boston Scientific and Abbott were among the companies showcasing new data on atrial fibrillation treatments, including pulsed field ablation catheters.
By Susan Kelly • Feb. 10, 2026 -
Stryker starts limited release of Mako RPS handheld knee robot
Surgeons interested in robotic technology but wanting a simpler system than existing platforms are the target market for the new device.
By Nick Paul Taylor • Feb. 10, 2026 -
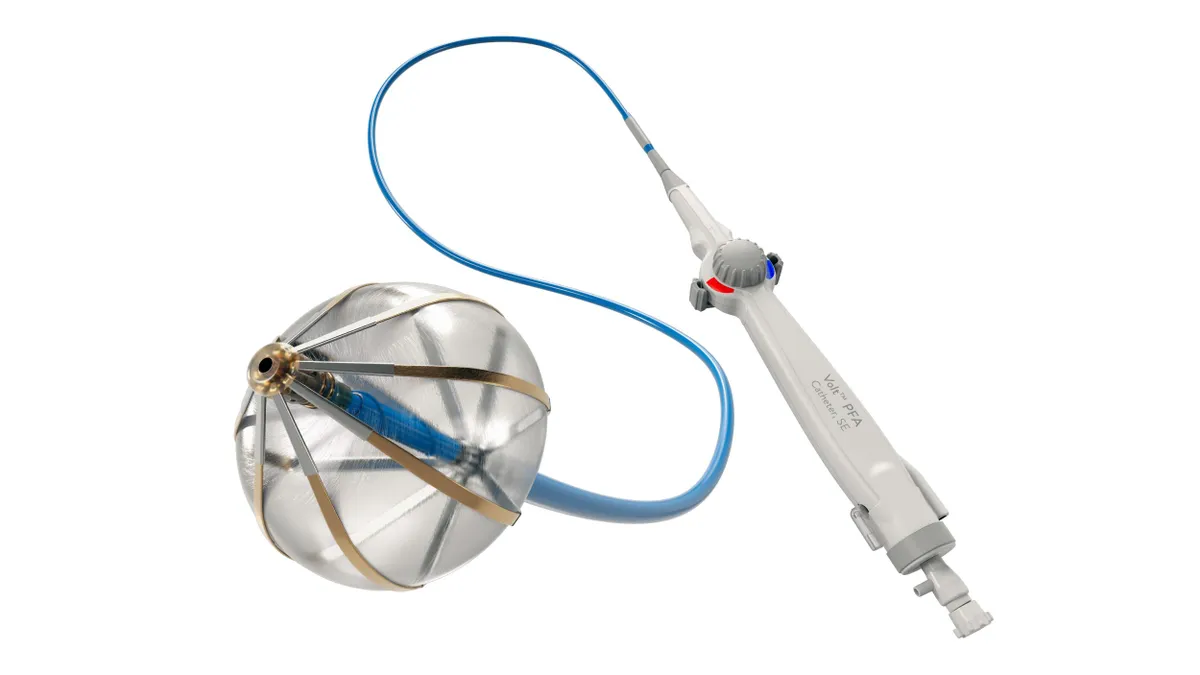
Abbott details Volt, TactiFlex data; Pulse Biosciences’ results shine
Abbott and Pulse released their findings at the AF Symposium that wrapped this weekend in Boston.
By Susan Kelly • Feb. 9, 2026 -
FDA breakthrough program starts FY2026 at steady pace
Orthopedics was the most active area over the second half of 2025, with the FDA issuing 13 breakthrough designations to devices in the therapeutic area.
By Nick Paul Taylor • Feb. 9, 2026 -
J&J recalls coil systems used in aneurysm treatment
Healthcare providers are advised to stop using the devices after a failure to detach was linked with four serious injuries and one death.
By Susan Kelly • Feb. 6, 2026 -
GE HealthCare expects smaller tariff impact in 2026
After taking a $245 million hit from the levies in 2025, CFO James Saccaro said the company plans more actions to mitigate their effects in 2026, including shifting its supply chain.
By Elise Reuter • Feb. 5, 2026 -
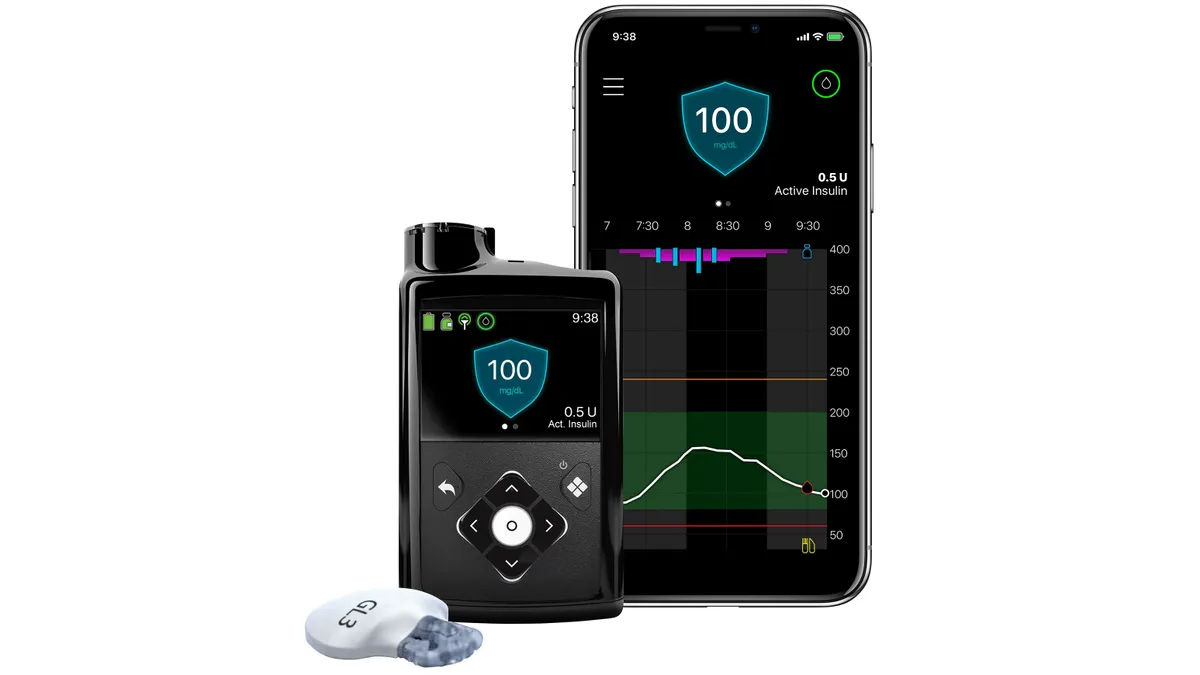
Medtronic wins expanded FDA label for 780G insulin pump
The 510(k) decision clears the device for use with ultra-rapid-acting insulins from Eli Lilly and Novo Nordisk.
By Nick Paul Taylor • Feb. 5, 2026 -
Prices rise at surgery centers acquired by Optum: study
The full financial impact of the price hikes could exceed $67 million each year in two dozen markets, according to the new study published in Health Affairs.
By Rebecca Pifer Parduhn • Feb. 4, 2026 -
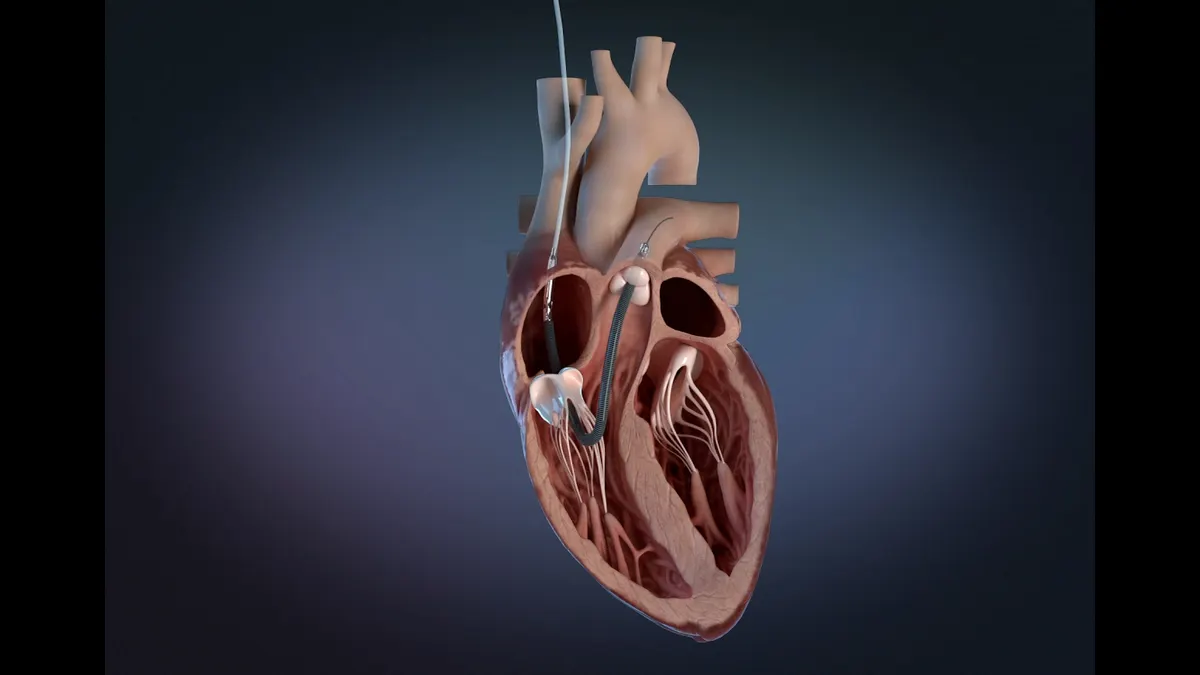
J&J corrects Impella heart pumps over issue linked to 22 serious injuries
The urgent correction notice is the latest in a series of actions related to the safety of Impella devices.
By Nick Paul Taylor • Feb. 4, 2026 -
Genetic testing takes greater share of Medicare Part B test spending: OIG
Spending on the tests, which analyze genetic material from both people and pathogens, rose 20% year over year in 2024, the analysis found.
By Susan Kelly • Feb. 4, 2026 -
Abbott receives FDA warning letter over Freestyle Libre CGMs
Abbott needs to conduct more performance testing to ensure its FreeStyle Libre devices are accurate, FDA inspectors found.
By Elise Reuter • Feb. 3, 2026 -
Medtronic to acquire CathWorks for up to $585M
The purchase will give Medtronic a larger presence in the cath lab, with a tool to help diagnose and treat coronary artery disease.
By Elise Reuter • Feb. 3, 2026 -
STAAR names interim co-CEOs amid search for permanent leader
The company is looking for a new top executive after former CEO Stephen Farrell left when STAAR shareholders rejected a buyout offer from Alcon.
By Nick Paul Taylor • Feb. 3, 2026 -
Beta Bionics receives FDA warning letter
The letter, which the FDA has not yet published, does not affect Beta Bionics’ ability to market, manufacture or distribute products, the company said.
By Elise Reuter • Feb. 2, 2026 -
Lawsuit challenges ethylene oxide exemptions for device sterilizers
The lawsuit questions the Trump administration’s authority to make sweeping environmental exemptions without evidence.
By Elise Reuter • Feb. 2, 2026 -
Stryker starts first cases with handheld ortho robot
Stryker expects the robot, called Mako RPS, to bring in new customers, including more ambulatory surgery centers.
By Elise Reuter • Jan. 30, 2026 -
Senseonics wins CE mark for one-year CGM
The company is hiring sales reps and transitioning staff from its former partner to support launches in Germany, Italy, Spain and Sweden.
By Nick Paul Taylor • Jan. 30, 2026 -
Top medtech trends to watch in 2026
From M&A to surgical robotics and user fee negotiations, the medical device industry has a busy year ahead. Check out MedTech Dive’s roundup of the top medtech trends to watch in 2026.
By Ricky Zipp • Jan. 29, 2026 -
CDRH on track for review timelines despite staff cuts
The Center for Devices and Radiological Health said it is on track to meet review times specified by user fee agreements, despite “resource challenges.”
By Elise Reuter • Jan. 29, 2026