Editor’s note: This article is the second in a series about the market for continuous glucose monitors. Read the first story here.
In a year marked by the launch of new diabetes devices, Abbott and Dexcom announced plans for new sensors intended for people who don’t take insulin.
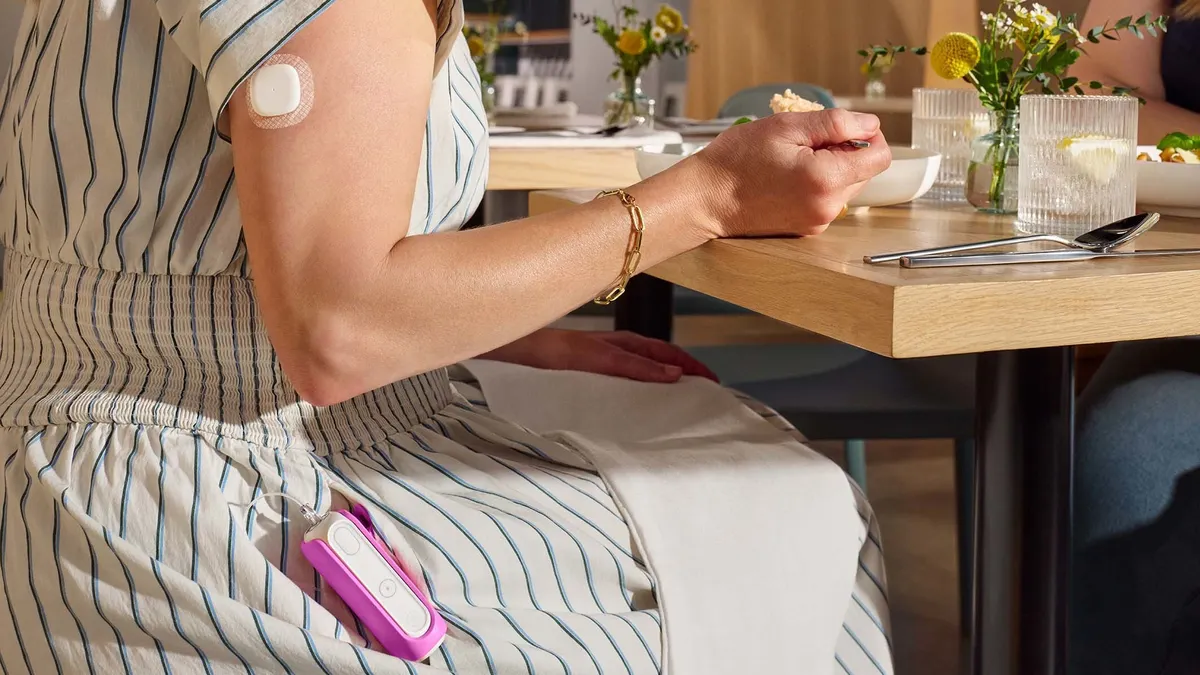
Abbott Laboratories debuted a new wearable in July designed to help people without diabetes track glucose spikes and lows. The company, which makes continuous glucose monitors to help people manage their diabetes, quietly rolled out a new website for the consumer-facing device called Lingo, which is currently available in the U.K.
Abbott is billing the device as “Your personal metabolic coach for optimal wellbeing,” as part of a subscription package costing 150 pounds ($190) per month. Users receive two sensors, similar to Abbott’s Freestyle Libre CGMs, but the resulting data is presented in a different way through an app, with a focus on counting glucose spikes throughout the day.
Abbott CEO Robert Ford has said he expects the consumer devices to contribute to the company’s goal of $10 billion in sales for its Libre business by 2028.
After testing Lingo in the U.K., Abbott plans to roll it out to other markets, including filing in the U.S. at the end of the year, Ford said in a July 20 earnings call.
“I don’t expect a big contribution right now from a financial perspective early on,” Ford said, “but I absolutely expect this to be a significant contributor over time for us.”
Competitor Dexcom is also looking for ways to extend its sensor technology to a broader group of people. The company announced plans in June to develop a new CGM targeted at people with Type 2 diabetes who don’t take insulin.
“We envision that there will be multiple iterations and generations of this product focused on adults who are not on insulin,” Dexcom Chief Commercial Officer Teri Lawver said in an interview.
Both companies are laying out plans for a broader category of devices focused on metabolic health, with hopes of sustaining future growth. Whether they will succeed is yet to be proven, experts said.
Libre to Lingo
“When we were developing the FreeStyle Libre technology, we always imagined it as a platform that had broader applicability,” Marc Taub, divisional vice president of technical operations for Abbott’s diabetes business, said in an interview.
The company’s Libre CGMs are designed and intended for people with diabetes, but Abbott is taking the underlying technology from the platform and developing it into a new line of products that are appropriate for the health and wellness markets, Taub said.
“The sensor, the chemistry, that fundamentally is the same, but the product needs to be different for those different people across the consumer segments,” he added.
Flush with cash from COVID-19 testing, Abbott first announced plans for a category of Lingo “biowearables” at the Consumer Electronics Showcase in 2022. Ford outlined a concept for a line of devices that can track glucose, lactate, and ketones.
The current Lingo device only measures blood glucose, but Ford said the company may add “a pipeline of different analytes” in the future. Abbott already has experience with this; it is building a CGM that can measure glucose and ketones to help people with diabetes avoid diabetic ketoacidosis, a serious complication where the body produces too many ketones.
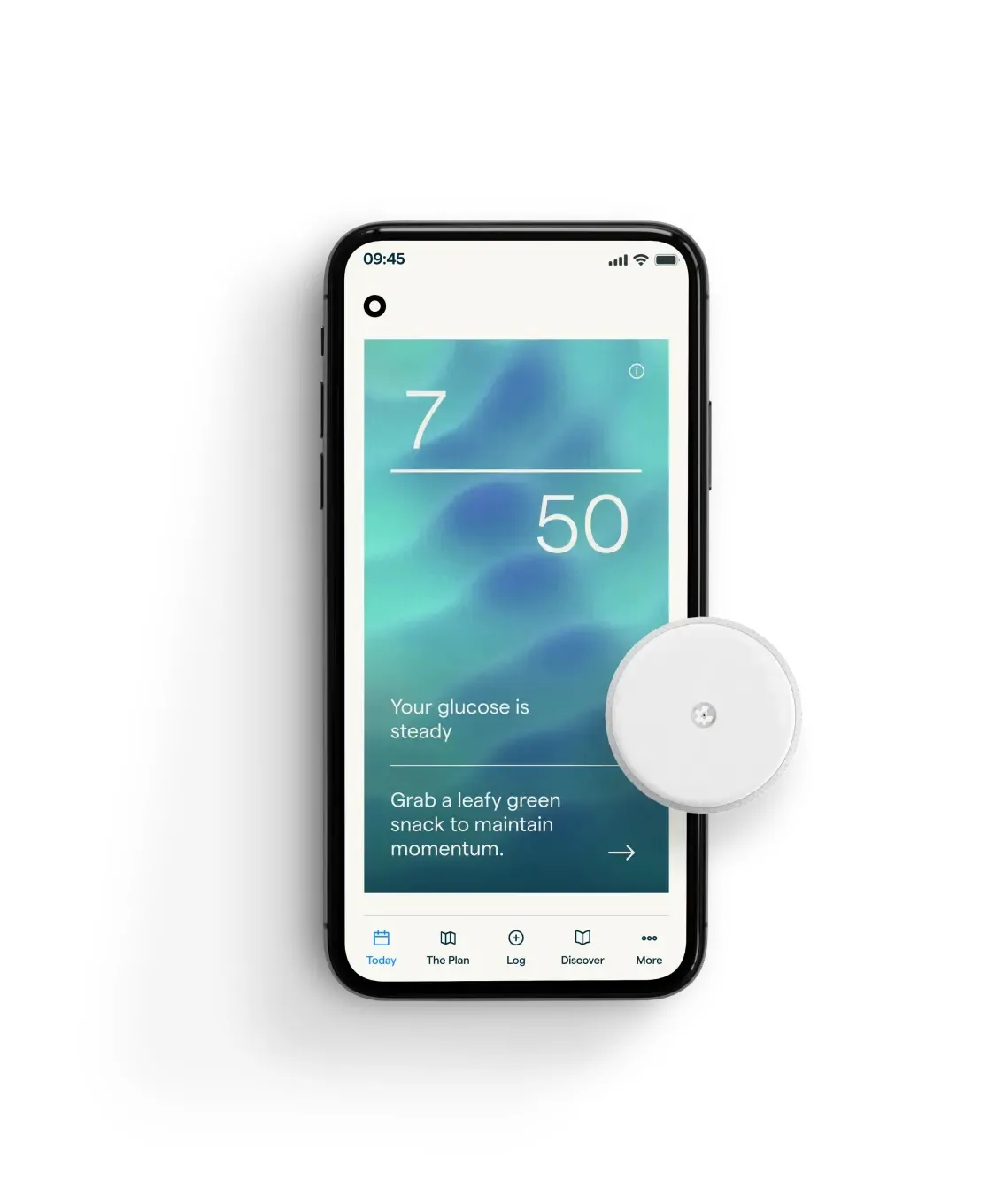
One key change between Abbott’s CGMs and its consumer wearables is how blood glucose information is presented. For example, time in range, a measure used in CGMs to help people with diabetes track how often their blood glucose levels are in a recommended range, might not be as relevant for people interested in athletic performance or general wellness, Taub said.
Lingo tracks how often a person’s blood sugar spikes during the day, which is measured as a “Lingo count.”
“We believe that the simplicity of this Lingo count is really key to modifying behavior,” Ford said.
Capturing the broader Type 2 market
Dexcom, meanwhile, has talked about building a new CGM targeted at the roughly 25 million people in the U.S. with Type 2 diabetes who don’t take insulin. It would be a departure from Dexcom’s current line of CGMs, which are predominantly used by people with Type 1 diabetes, according to data from UBS Securities. Typically, insurers don’t cover the devices for people who don’t take insulin.
Details about the planned device are sparse, but Dexcom has said it will be based on a 15-day version of its G7 CGM, and will include a cash-pay option. It will also include some software differences from Dexcom’s other devices, such as offering more curated weekly reports, and reducing alarms and alerts that would be unnecessary for most of these users, Lawver said.
“We know that the biggest needs of this population are one, how do I optimize my exercise and nutrition choices? And number two, how do I prevent the progression of disease?” she added.
The company is working on collecting evidence that these patients benefit from using CGMs, too, including real-world data presented at the American Diabetes Association Scientific Sessions.
Dexcom hasn’t announced plans for any consumer-specific devices yet, but CEO Kevin Sayer has spoken openly about the value he sees for CGMs in conditions like prediabetes.
“When you wear a glucose sensor, you can see the effect of what goes on in your life and what foods, what timing of foods is not good for you,” Sayer said in an interview. “I wear sensors all the time.”
Dexcom has also hinted at plans for a broader consumer focus. Earlier this year, the company changed its mission statement from “empowering people to take control of diabetes” to “empowering people to take control of health.”
“Importantly, this new product also provides a glimpse into our future where we expect to utilize software to build tailored experiences and serve much larger populations,” Sayer said in a July earnings call.
Source: International Data Corporation, U.S. Consumer Healthcare Survey and U.S. Healthcare Provider Technology and Connected Health Survey, January 2022.
“The higher response rate of consumers reporting using wearables for managing their chronic conditions versus providers investing in this technology to use with their patients is also why medical device manufacturers are using direct-to-consumer marketing approaches.”

Lynne Dunbrack
Group vice president of public sector for IDC
Consumer demand
Insurance doesn’t cover CGMs for people without diabetes, but that hasn’t stopped people from purchasing the devices. On TikTok, a person dubbed the “blood sugar king” shares their blood sugar readings while eating Chipotle burritos and hot wings. Ads for wellness programs that incorporate CGMs — such as Nutrisense — come up as top results in a Google search.
Currently, there’s little evidence that wearing a CGM offers health benefits for people without diabetes. A study in 2019 of medical students without diabetes who wore the devices found that the changes in their blood glucose range were slight. It gets to the point that they “get bored of the information,” one of the study’s co-authors told JAMA.
But that hasn’t deterred interest.
“I’ve seen people do it just because they want to know. People like monitoring their biometrics,” said Dr. Marilyn Tan, an endocrinologist at Stanford Healthcare, pointing to Apple’s arrhythmia-detecting feature in smartwatches as another example. She added that she sees a lot of interest in these types of wearables from venture capitalists and people who work in tech.
“On the other hand, if your blood sugar is completely normal, I’m not sure how much more information that gives you,” she said. “If it gives you information that helps you to guide your choices to make healthier lifestyle choices, that's great.”
Admittedly, some of those people wear CGMs but don’t scan their devices to retrieve the data, “and then it’s kind of pointless,” she added.
That said, Tan does see a benefit in people using the CGMs to better understand how their blood sugar responds to different foods and said she finds them “extremely helpful” for patients with prediabetes, even though it would be an off-label use of the device.
“It’s really eye opening. Things that are not intuitive can be revealed. And it really helps in my mind with personalized medicine because you can really tailor your diet toward foods that don't spike your blood sugar,” Tan said.
She said insurance coverage has gotten “tremendously better” over the last few years, and a couple of her patients with prediabetes have been able to get their CGMs covered. Most of these patients still pay out-of-pocket, but the cost — while substantial — has gotten more attainable at about $75 to $100 per month with a coupon, she said.
Medical to ‘wellness’
The idea of turning a medical device into a consumer product is not new. Scales, pulse oximeters and thermometers have all gone through this process, said Natasha Bond, president of ERI Group, a Colorado-based firm that manufactures and develops medical devices.
It’s much easier to turn a medical device into a consumer product than the other way around.
“I have helped multiple companies go in both directions,” Bond said. “Trying to help consumer products become medical devices is a very, very difficult challenge. It requires a top-to-bottom wholesale rethink of the entire way they do business, because medical device regulation is onerous and pervasive and hits every element of the business.”
For example, Apple is developing a proof-of-concept for a light-based blood glucose monitor, according to a report earlier this year from Bloomberg. The technology would be intended to help notify people that they might have prediabetes, but it has a long way to go, including shrinking down the prototype to a size that could be embedded into an Apple Watch, and working with regulators, according to the report.
For a medtech company looking to sell a consumer version of its device, that firm could use the same technology, execute a separate marketing plan “and magic up another revenue stream,” Bond said. “That’s almost free money, right? I think the question though is how far are these different companies actually going to go down that route?”
It also raises some questions that are not well answered by current regulations, she added. For example, what happens if someone continues to use a consumer device after they are diagnosed with diabetes? Or what happens if people share their data with their physician, who doesn’t know it came from a consumer device?
“There’s a risk there,” Bond said.
Future of the business
Abbott hasn’t yet shared sales expectations for its Lingo devices.
“I would say that they do envision it being a big growth driver to come and something quite novel, but I don't know that that comes immediately,” BTIG analyst Marie Thibault said in an interview.
“I think they think it will be big. I think that's yet to be proven,” William Blair analyst Margaret Kaczor said.
Abbott took a similar direction in 2020 when it debuted a device geared at athletes, called Libre Sense, which can be bought over-the-counter to monitor blood glucose. Abbott hasn’t broken out sales of the device in its earnings from the rest of its diabetes segment, and Thibault estimated it wasn't a big revenue contributor compared to its core business.
That said, she sees value in the devices for understanding how different factors, including food, sleep and exercise, affect glucose control.
“I myself monitor my glucose as sort of a consumer that wants to be aware,” Thibault said. “I think there is a demand among the consumer population for these sorts of wearables. And Abbott is seeing that and realizing they have a lot of access to consumers already through other parts of their businesses.”
Despite rising interest in glucose monitoring, the focus for medtech companies will likely remain on people with diabetes. These firms have long relationships with people whose devices and medications are a part of their lives, Bond said.
But if other companies are offering glucose monitoring to a broader group of people, “Why on earth would they stay out of that competition?”
Correction: This article was updated with information about Abbott’s plans for Lingo in the U.S. Abbott intends to file the device with regulators by the end of the year.