Dive Brief:
- HistoSonics said Monday it has filed for de novo authorization of its Edison Histotripsy System for the treatment of kidney tumors.
- The submission to the Food and Drug Administration is based on an ongoing kidney cancer trial that completed enrollment last year.
- HistoSonics raised $250 million in October to expand beyond liver cancer, enabling the company to advance its kidney cancer program and start a benign prostatic hyperplasia study.
Dive Insight:
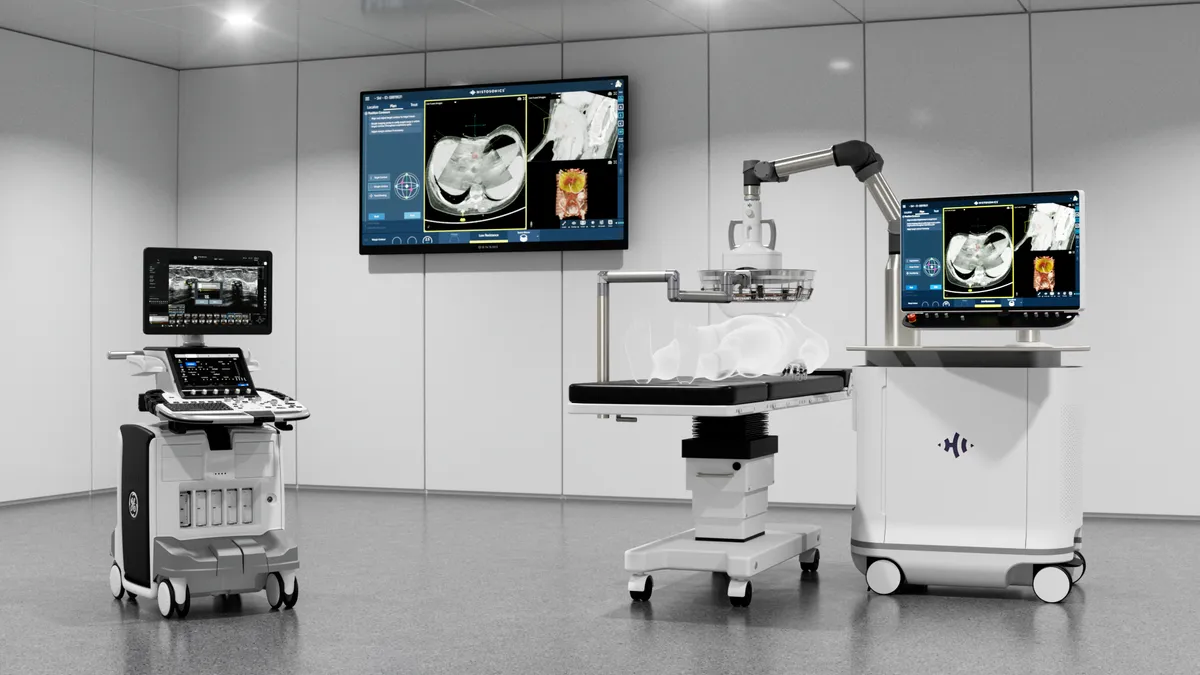
The Edison system delivers pulsed sound waves that cause bubbles to quickly form and collapse. Guided by real-time imaging, the histotripsy system is designed to provide precise and predictable destruction of the target tissue while preserving organ function. The noninvasive approach eliminates the incisions, radiation and thermal injury associated with other methods of removing and destroying tissue.
HistoSonics received de novo authorization for the product in the treatment of liver cancer in 2023. The company started a pivotal kidney cancer trial the next year. HistoSonics is evaluating the percentage of targeted tumors that are successfully eliminated after a single histotripsy session. The trial “continues to generate promising insights,” according to HistoSonics.
De novo authorization could significantly expand HistoSonics’ addressable market. In the U.S., physicians will diagnose about 80,000 new cases of kidney cancer this year, the American Cancer Society estimates. Physicians are estimated to diagnose about 42,000 new liver cancer cases in the U.S. this year.
Kidney cancer patients have multiple treatment options, including surgical removal of the tumor and destruction of cancer cells using thermal ablation. However, the procedures are invasive and can cause bleeding, infection and unintended damage to surrounding tissue, HistoSonics said.
The planned expansion into kidney cancer is part of a broader push into new indications. HistoSonics is running trials in pancreatic tumors and benign prostatic hyperplasia and sees opportunities to apply the technology to a range of benign and malignant conditions.
Investors including K5 Global, Bezos Expeditions and Wellington Management backed HistoSonics’ vision last year, buying a majority stake in a deal that valued the company at $2.25 billion and participating in a $250 million financing round. HistoSonics raised the money to expand into new indications and global markets.