Dive Brief:
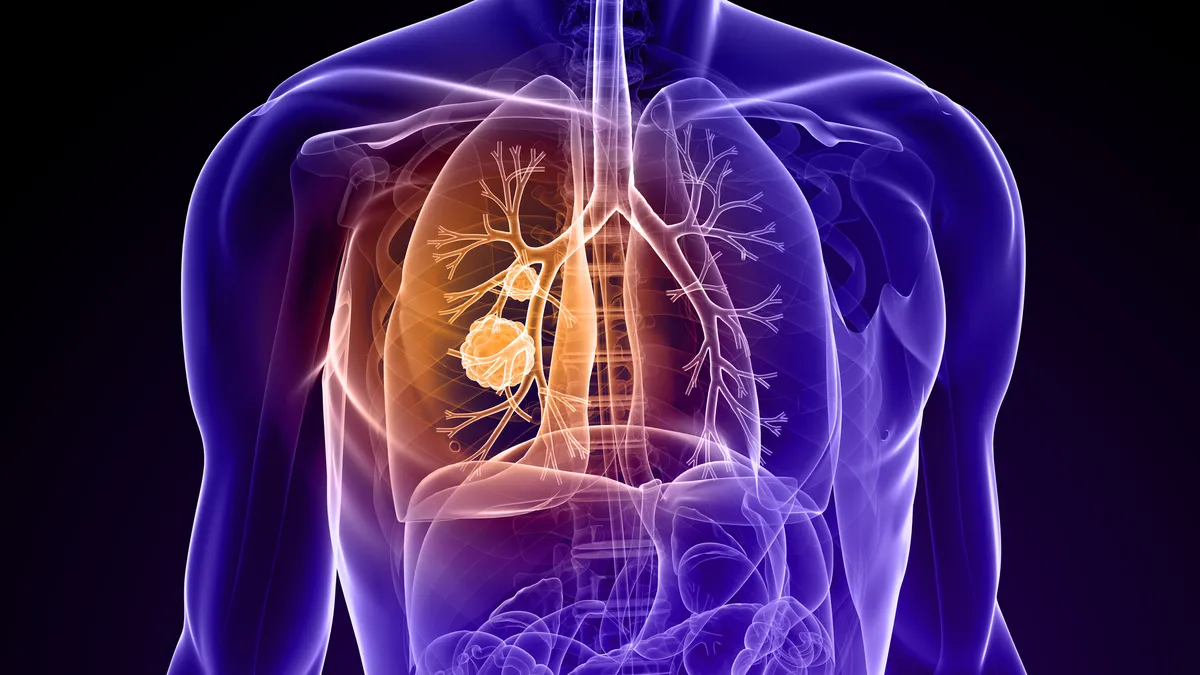
- Swiss biotech Novocure said Thursday its electrical field medical device, paired with immunotherapy, helped people with advanced lung cancer live significantly longer than those receiving immunotherapy alone in a Phase 3 trial.
- The results, disclosed in a press release Thursday, didn’t include specific details, leaving the treatment’s effect size unclear.
- Novocure says its devices, called “tumor treating fields therapy,” slow the growth of cancer by disrupting tumor cell division, migration and repair mechanisms as well as stimulating an immune response. It’s already gained FDA approval for electrical field products that treat brain cancer and a rare lung tumor.
Dive Insight:
Novocure’s treatment was first approved in 2011 for the brain cancer glioblastoma under the FDA’s standard device treatment pathway, and in 2019 for malignant mesothelioma under the regulator’s “humanitarian use” program. Adding a clearance in non-small cell lung cancer could substantially expand its use, as annual diagnoses for that condition number more than 200,000 in the U.S., compared with 13,000 glioblastoma and 3,000 mesothelioma cases.
An approval could also help improve Novocure’s financial outlook. The company didn’t make a profit in 2021 and was posting losses in 2022. In 2021, it had a loss of $58 million on $535 million in revenue, and through the first nine months of 2022 lost another $55 million on $409 million in revenue.
Novocure’s Phase 3 trial enrolled 276 patients with cancer that had spread to at least one other area of the body and who had progressed on platinum-based chemotherapy. Patients and physicians knew who was receiving the tumor-treating fields therapy on top of immunotherapy or docetaxel. The trial’s primary goal was to see whether patients receiving a combination of tumor-treating fields and immunotherapy or docetaxel would live longer than those who got either immunotherapy or docetaxel alone.
Novocure’s therapy also didn’t definitively beat docetaxel on measures of survival. Still, the company’s shares rose 52% in early trading Thursday as Novocure announced plans to ask the Food and Drug Administration to approve the device in the second half of 2023, as well as seek a CE mark in the European Union.
In its statement, the company said the therapy demonstrated a “statistically significant and clinically meaningful improvement in overall survival.” Novocure said it was holding detailed data for an upcoming medical meeting, leaving important information, such as the magnitude of its survival benefit or the type of immunotherapy patients were receiving, unclear.
Those details are important. Two immunotherapies for non-small cell lung cancer — Bristol Myers Squibb’s Opdivo and Roche’s Tecentriq — beat docetaxel on overall survival when evaluated as monotherapies in clinical testing. Furthermore, docetaxel is frequently used when these patients have already received an immunotherapy like Merck & Co.’s Keytruda, which is a commonly prescribed as a first-line treatment.
“We think showing a trend though not hitting [statistical significance] could generate some questions,” Jefferies analyst Michael Yee, who covers Novocure’s China-based partner Zai Labs, wrote in a Jan. 5 note to clients.
Shares jumped 60% from Wednesday’s close at $70.53, rising $42.36 to $112.98 in midday trading on Friday.