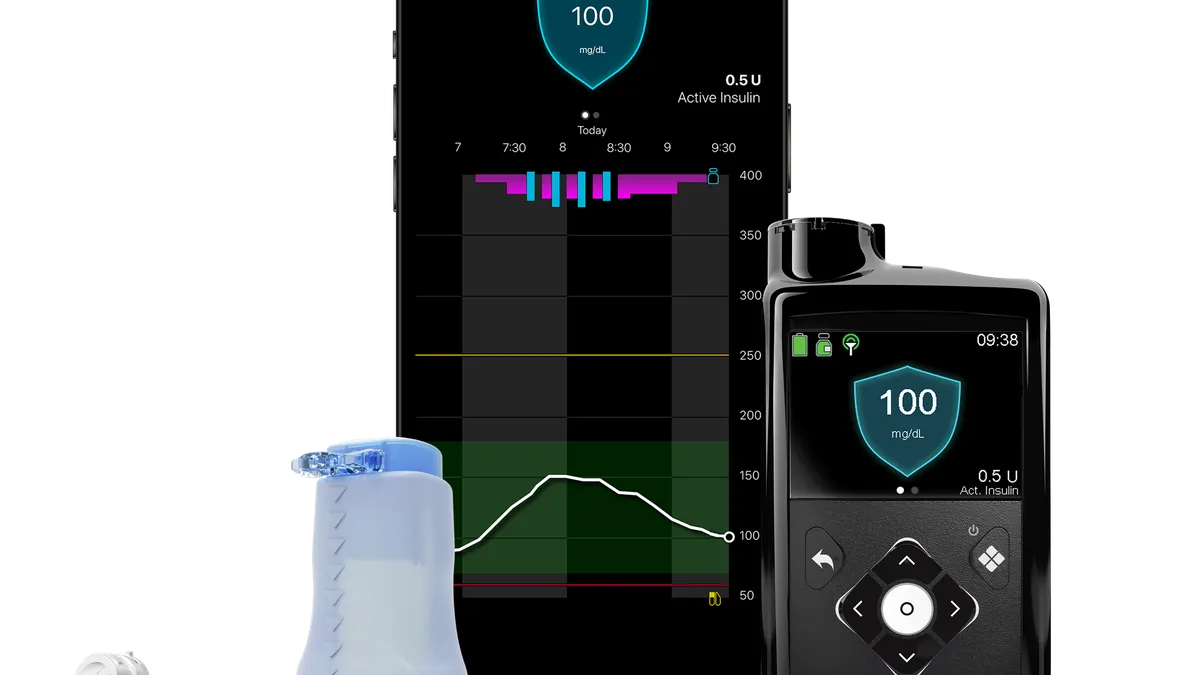
Dive Brief:
- Medtronic has received Medicare and Medicare Advantage coverage for its recently approved MiniMed 780G insulin pump.
- The company said on Thursday that it has begun processing orders and will start shipping devices to patients with Type 1 diabetes who meet the eligibility criteria over “the next few weeks.”
- The coverage clears a barrier to patient access for a pump that is central to Medtronic’s plans to kickstart growth at its diabetes business after a tough period defined by regulatory problems.
Dive Insight:
The U.S. Food and Drug Administration approved the 780G pump in April. Medtronic had aimed to bring the device to the U.S. market sooner, but its plans were delayed by a warning letter, hindering its ability to compete with Insulet and Tandem Diabetes Care in the insulin pump market.
With the device now approved for use, Medtronic is working to ensure patients in the U.S. can access the pump. The Centers for Medicare & Medicaid Services delivered a boost to that effort last week by agreeing to cover MiniMed 780G for all eligible Medicare and Medicare Advantage beneficiaries. An order form for the device is now live on Medtronic’s website.
Securing Medicare coverage positions Medtronic to access more patients and build on the progress of its launch to date.
The “780G is a very important step in the turnaround of the U.S. business. It's going to take us time to rebuild the installed base,” Que Dallara, president of Medtronic’s diabetes unit, said at a June investor event. “We have approximately 10,000 unique [healthcare providers] already prescribing 780G in the first 30 days. Our CGM attachment, which historically in the U.S. has been very low because we've had fingerstick requirements in our sensor, is very high.”
Medtronic has yet to discuss the financial side of the launch. The company is scheduled to release its first quarter financial results, which will cover the first two months of the MiniMed 780G launch, next month.