Dive Brief:
- Roche's Genentech over the weekend released additional results from a Phase 2 study for its Port Delivery System for its eye drug ranibizumab in people with wet age-related macular degeneration at the annual meeting of the American Academy of Ophthalmology in Chicago.
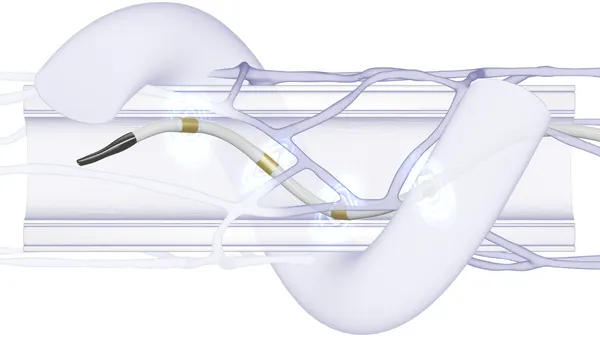
- The data showed that vision and anatomical outcomes in wet AMD patients who received the device, and were dosed every six months or longer, were comparable to ranibizumab dosed every four weeks. The PDS device, which is surgically implanted into the eye, is refillable and about the size of a grain of rice.
- Genentech sees new long-acting delivery systems as a way to address under-treatment and improve clinical outcomes for wet AMD patients. Ranibizumab, as an active drug, is currently sold under the brand name Lucentis.
Dive Insight:
Age-related macular degeneration (AMD) is a leading cause of blindness in people age 60 and older, affecting about 11 million Americans, according to Genentech. The disease affects the part of the eye that provides central vision. About 1.1 million Americans have wet AMD, an advanced form of the disease that can cause rapid, severe vision loss.
Lucentis (ranibizumab), developed by Genentech, came to market in 2006 and proved a disruptor for wet AMD treatment. Lucentis is a vascular endothelial growth factor (VEGF) inhibitor designed to bind to and inhibit VEGF-A, a protein thought to play a role in blood vessel formation.
Regeneron Pharmaceuticals brought competition to the wet AMD treatment market with the introduction of Eylea (aflibercept), which also works by inhibiting VEGF. Eylea's dosing, as a 2 mg once-monthly treatment, with an option to switch to a regimen of once every two months after the first three months of treatment, has helped Regeneron expand its market share.
Regulators recommend wet AMD patients treated with the currently marketed Lucentis get a 0.5 mg injection every 28 days.
The PDS continuously delivers a special formulation of ranibizumab over time. The formulation is not FDA-approved and is different from the approved ranibizumab intravitreal injection.
In July, Genentech released data from its Ladder study showing that a majority of wet AMD patients who received the device went at least six months before needing a refill of the drug.
Patients in the high-dose PDS group achieved similar visual outcomes as ranibizumab 0.5 mg dosed every four weeks, the company said. Based on results from the Ladder study, the pivotal Phase III Archway clinical trial and the Portal label extension study were initiated in September. The studies will evaluate the safety and effectiveness of the PDS 100 mg/mL concentration in patients with wet AMD at a fixed dosing interval of 24 weeks.