Medtronic's second run at bringing a renal denervation treatment to market in the U.S. reaches an important milestone next month. And competitors are likely watching intently.
Six years after its device for treating resistant hypertension failed to demonstrate effectiveness in a closely eyed clinical study, the medtech giant is set to unveil new data on its so-called Symplicity system on March 29 in a late-breaking trial at the American College of Cardiology meeting in Chicago.
Outgoing Medtronic CEO Omar Ishrak in January highlighted forthcoming renal denervation tech as a key product in the company’s pipeline in a presentation at the J.P. Morgan Healthcare Conference. Calling it “a market that we have to create,” Ishrak noted that the company began work to develop the treatment about 10 years ago and has stuck with it despite the 2014 clinical trial setback. “This has not been easy,” Ishrak told investors.
If results of the SPYRAL trial are favorable, the company will move a big step closer to being rewarded for its efforts.
Resistant hypertension is blood pressure that remains elevated above target levels despite the use of medications prescribed to control it. With about 12% to 15% of the population treated for high blood pressure struggling with the resistant form, according to the American Heart Association, a novel treatment could be a game changer.
“We expect positive pivotal trial results to serve as a significant catalyst for [Medtronic] shares since we think it would increase the probability that [Medtronic] is first to market in the U.S. with a renal denervation system,” Needham analyst Mike Matson said in a report Friday to clients.
Matson thinks Medtronic may have a potential $1.7 billion market on its hands. That's based on the belief that 5% of an estimated 42 million U.S. adults with uncontrolled primary hypertension and 4.6 million with resistant hypertension would be eligible for renal denervation, at around $7,500 per device.
Medtronic could enter the market as early as 2021, Matson said, and company management has said it's already working with Medicare and private payers to obtain coverage.
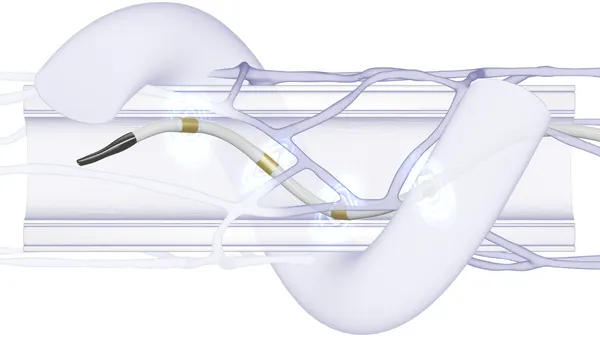
The minimally invasive renal denervation procedure works by targeting specific nerves near the kidneys with radio frequency ablation to regulate overactivity. Medtronic picked up the original technology in its 2011 acquisition of Ardian for $800 million. Although not approved in the U.S., Japan or Canada, the device is sold in more than 50 countries, according to the company.
In addition to the pivotal trial to be unveiled at ACC, last week the company announced it will begin enrolling patients in a pilot study evaluating the safety and efficacy of the system using a simplified procedural approach with a reduced number of radio frequency ablations. The single-arm study will enroll 50 patients at up to 15 sites in the U.S. and Europe.
Needham's Matson said that while Medtronic is on track to become the first company to the U.S. market with renal denervation, competitors Abbott and Boston Scientific are likely to restart development of their own programs, which were "shelved" in the wake of Medtronic’s earlier failed study. Several smaller companies, including Ablative Solutions and Recor Medical, are also working on renal denervation systems, the analyst wrote.